Abstract
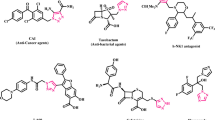
Cellulose-immobilized Cu(II) Schiff base complex [Cell-AP-Cu(II)] has been designed and synthesized by multistep approach by the reaction of Schiff base functionalized cellulose (Cell-AP) with Cu(OAc)2 in ethanol under reflux condition. The [Cell-AP-Cu(II)] complex was characterized by FT-IR, TGA, XRD, SEM, EDS, and BET/BJH techniques. The functionality and composition of the catalyst was confirmed by IR and EDX analysis, respectively. The catalytic effectiveness of [Cell-AP-Cu (II)] is investigated for Chan–Lam coupling of phenylboronic acids and secondary amines. A range of functionalized derivatives have been synthesized with high yield in short reaction time. The plausible mechanism illustrates the formation of derivatives. The peculiar advantages of the proposed protocol are high TONs (up to 873) and TOFs (up to 873), minimal catalyst loading (0.11 mol %), recyclability of the catalyst (up to 5 times) and moderate reaction conditions.











Similar content being viewed by others
Availability of data and materials
Data are available as electronic supplementary material.
References
A.P. Dysin, A.R. Egorov, O. Khubiev, R. Golubev, A.A. Kirichuk, V.N. Khrustalev, N.N. Lobanov, V.V. Rubanik, A.G. Tskhovrebov, A.S. Kritchenkov, Catalysts 13, 203 (2023)
Y. Rangraz, M.M. Heravi, Appl. Organomet. Chem. 37, e6819 (2023)
M.J. Buskes, M.-J. Blanco, Molecules 25, 3493 (2020)
J. Chen, J. Li, Z. Dong, Adv. Synth. Catal. 362, 3311 (2020)
F. Abedinifar, M. Mahdavi, E. Babazadeh Rezaei, M. Asadi, B. Larijani, Curr. Org. Synth. 19, 16 (2022)
K. Kumari, S. Kumar, K.N. Singh, M.G.B. Drew, N. Singh, New J. Chem. 44, 12143 (2020)
D.S. Raghuvanshi, A.K. Gupta, K.N. Singh, Org. Lett. 14, 4326 (2012)
I. Munir, A.F. Zahoor, N. Rasool, S.A.R. Naqvi, K.M. Zia, R. Ahmad, Mol. Divers. 23, 215 (2019)
S.M. Hashemian, T. Farhadi, M. Ganjparvar, Drug Des. Dev. Ther. 12, 1759 (2018)
C. Bailly, P.-E. Hecquet, M. Kouach, X. Thuru, J.-F. Goossens, Bioorg. Med. Chem. 28, 115463 (2020)
D.Y. Mladentsev, E.N. Kuznetsova, M.N. Skvortsova, R.R. Dashkin, Org. Process Res. Dev. 26, 2311 (2022)
F. Kratz, U. Beyer, T. Roth, M.T. Schütte, A. Unold, H.H. Fiebig, C. Unger, Arch. Pharm. (Weinheim). 331, 47 (1998)
R. Agrawal, R. Shyam Bahare, P. Jain, S. Narayan Dikshit, S. Ganguly, Mini Rev. Med. Chem. 12, 1345 (2012)
D. Feedman, Endocr. Rev. 7, 409 (1986)
B. Seifinoferest, A. Tanbakouchian, B. Larijani, M. Mahdavi, Asian. J. Org. Chem. 10, 1319 (2021)
T.M.A. Al-Shboul, S.S. Al-Tarawneh, T.S. Ababneh, T.M.A. Jazzazi, Separations 9, 55 (2022)
B.S. Takale, F.-Y. Kong, R.R. Thakore, Organics 3, 1 (2022)
F. Monnier, M. Taillefer, Angew. Chemie Int. Ed. 48, 6954 (2009)
I.P. Beletskaya, A.V. Cheprakov, Coord. Chem. Rev. 248, 2337 (2004)
M.M. Heravi, Z. Kheilkordi, V. Zadsirjan, M. Heydari, M. Malmir, J. Organomet. Chem. 861, 17 (2018)
R. Dorel, C.P. Grugel, A.M. Haydl, Angew. Chemie Int. Ed. 58, 17118 (2019)
P.A. Forero-Cortés, A.M. Haydl, Org. Process Res. Dev. 23, 1478 (2019)
D.H.R. Barton, J.-P. Finet, J. Khamsi, Tetrahedron Lett. 29, 1115 (1988)
P. Lopez-Alvarado, C. Avendano, J.C. Menendez, J. Org. Chem. 60, 5678 (1995)
A. C. Patrick Lam* , C. Clark, S. Saubern, J. Adams, K. Averill, D. Chan, Synlett 2000, 0674 (2000).
D.W. Old, M.C. Harris, S.L. Buchwald, Org. Lett. 2, 1403 (2000)
J. Qiao, P. Lam, Synthesis (Stuttg). 2011, 829 (2011)
M.J. West, J.W.B. Fyfe, J.C. Vantourout, A.J.B. Watson, Chem. Rev. 119, 12491 (2019)
A. Vijayan, D.N. Rao, K.V. Radhakrishnan, P.Y.S. Lam, P. Das, Synthesis (Stuttg). 53, 805 (2021)
X. Jia and J. He, Appl. Organomet. Chem. 36, (2022).
S. Raju, P. E. Sheridan, A. K. Hauer, A. E. Garrett, D. E. McConnell, J. A. Thornton, S. L. Stokes, and J. P. Emerson, Chem. Biodivers. 19, (2022).
H. Chen, C. Wei, G.-L. Pang, C. Liang, D.-L. Mo, X.-P. Ma, Org. Lett. 24, 6013 (2022)
C. Li, K. Zhang, H. Ma, S. Wu, Y. Huang, Y. Duan, Y. Luo, J. Yan, and G. Yang, Chem. A Eur. J. 28, (2022).
Y. Wang, T. Meng, S. Su, L. Han, N. Zhu, T. Jia, Adv. Synth. Catal. 364, 2040 (2022)
W. Shi, Y. Shi, M. Xü, G. Zou, X.-Y. Wu, Mol. Catal. 528, 112472 (2022)
T. Meng, L.A. Wells, T. Wang, J. Wang, S. Zhang, J. Wang, M.C. Kozlowski, T. Jia, J. Am. Chem. Soc. 144, 12476 (2022)
Y. Li, Z. Cao, Z. Wang, L. Xu, Y. Wei, Org. Lett. 24, 6554 (2022)
J. Valencia, O.A. Sánchez-Velasco, J. Saavedra-Olavarría, P. Hermosilla-Ibáñez, E.G. Pérez, D. Insuasty, Molecules 27, 8345 (2022)
H.-Y. Bi, Q.-Y. Wu, X.-M. Zhou, H.-J. Xu, C. Liang, D.-L. Mo, X.-P. Ma, Org. Lett. 24, 4675 (2022)
N. Ghasemi, A. Yavari, S. Bahadorikhalili, A. Moazzam, S. Hosseini, B. Larijani, A. Iraji, S. Moradi, M. Mahdavi, Inorganics 10, 231 (2022)
N. Ghasemi, A. Moazzam, S. Bahadorikhalili, A. Yavari, S. Hosseini, M.H. Sayahi, B. Larijani, H. Hamedifar, S. Ansari, M. Mahdavi, Catal. Lett. 153, 805 (2023)
S.P. Vibhute, P.M. Mhaldar, D.S. Gaikwad, R.V. Shejwal, D.M. Pore, Monatshefte Für Chemie Chem. Mon. 151, 87 (2020)
C. Sharma, N. Sharma, S. Sharma, S. Sharma, S. Paul, Curr. Res. Green Sustain. Chem. 4, 100133 (2021)
A. Clerc, V. Bénéteau, P. Pale, S. Chassaing, ChemCatChem 12, 2060 (2020)
A. Muñoz, P. Leo, G. Orcajo, F. Martínez, G. Calleja, ChemCatChem 11, 3376 (2019)
S. Naderi, R. Sandaroos, S. Peiman, B. Maleki, J. Phys. Chem. Solids 180, 111459 (2023)
A.A. Ismail, S.M. Lateef, Chem. Methodol. 6, 1007 (2022)
M.I. Obaid, W.A. Jaafar, Chem Methodol 6(6), 457 (2022)
B. Maleki, R. Sandaroos, S. Naderi, S. Peiman, J. Organomet. Chem. 990, 122666 (2023)
B. Maleki, R. Sandaroos, S. Peiman, Heliyon 9, e15041 (2023)
R. Sandaroos, B. Maleki, S. Naderi, S. Peiman, Inorg. Chem. Commun. 148, 110294 (2023)
H. Alinezhad, M. Tajbakhsh, B. Maleki, F.P. Oushibi, Polycycl. Aromat. Compd. 40, 1495 (2020)
B. Maleki, S.S. Ashrafi, R. Tayebee, RSC Adv. 4, 41521 (2014)
B. Maleki, G.E. Kahoo, R. Tayebee, Org. Prep. Proced. Int. 47, 461 (2015)
B. Jiang, C. Chen, Z. Liang, S. He, Y. Kuang, J. Song, R. Mi, G. Chen, M. Jiao, L. Hu, Adv. Funct. Mater. 30, 1906307 (2020)
T. Yamada, W. Teranishi, K. Park, J. Jiang, T. Tachikawa, S. Furusato, H. Sajiki, ChemCatChem 12, 4052 (2020)
S. Wang, A. Lu, L. Zhang, Prog. Polym. Sci. 53, 169 (2016)
G. Yang, H. Kong, Y. Chen, B. Liu, D. Zhu, L. Guo, G. Wei, Carbohydr. Polym. 279, 118947 (2022)
Y.-D. Dong, H. Zhang, G.-J. Zhong, G. Yao, B. Lai, Chem. Eng. J. 405, 126980 (2021)
P.S. Pharande, G.S. Rashinkar, D.M. Pore, Res. Chem. Intermed. 47, 4457 (2021)
L. Zhang, S. Long, H. Jiao, Z. Liu, P. Zhang, A. Lei, W. Gong, X. Pei, RSC Adv. 12, 18676 (2022)
A. Anžlovar, E. Žagar, Nanomaterials 12, 1837 (2022)
S. Kamel, TA Khattab Cellulose 28, 4545 (2021)
J. Rahimi, R. Taheri-Ledari, A. Maleki, Curr. Org. Synth. 17, 288 (2020)
H. Ahmad, J. Clust. Sci. 33, 421 (2022)
F. Liu, X. Liu, Q. Fu, Cellulose 30, 953 (2023)
M. Boroumand Jazi, M. Arshadi, M.J. Amiri, A. Gil, J. Colloid Interface Sci. 422, 16 (2014)
W. Gindl-altmutter, M. Bacher, M. Edler, T. Griesser, Int. J. Biol. Macromol. 106, 1288 (2017)
X. Qi, H. Yoon, S.H. Lee, J. Yoon, S.J. Kim, J. Ind. Eng. Chem. 14, 136 (2008)
N. Safajoo, B. Bi, F. Mirjalili, A. Bamoniri, RSC Adv. 9, 1278 (2019)
C.J. Fui, T.X. Ting, M.S. Sarjadi, Z. Amin, S.M. Sarkar, B. Musta, M. Rahman, ACS Omega 6, 6766 (2021)
M.M. Vadiyar, S.C. Bhise, S.S. Kolekar, J.-Y. Chang, K.S. Ghule, A.V. Ghule, J. Mater. Chem. A 4, 3504 (2016)
M. Diakité, A. Paul, C. Jäger, J. Pielert, J. Mumme, Bioresour. Technol. 150, 98 (2013)
J.B. Chen, K. Natte, N.Y.T. Man, S.G. Stewart, X.F. Wu, Tetrahedron Lett. 46, 4843 (2015)
A. Khosravi, J. Mokhtari, M.R. Naimi-Jamal, S. Tahmasebi, L. Panahi, RSC Adv. 7, 46022 (2017)
Y. Han, M. Zhang, Y.-Q. Zhang, Z.-H. Zhang, Green Chem. 20, 4891 (2018)
M. Sarmah, A. Dewan, P.K. Boruah, M.R. Das, U. Bora, Appl. Organomet. Chem. 34, e5554 (2020)
N. Seyedi, M. Shahabi Nejad, K. Saidi, and H. Sheibani, Appl. Organomet. Chem. 34, (2020).
X. Jia, P. Peng, Org. Biomol. Chem. 16, 8984 (2018)
C. Zhang, H. Zhu, K. Gang, M. Tao, N. Ma, W. Zhang, React. Funct. Polym. 160, 104831 (2021)
S. Sharma, M. Kaur, C. Sharma, A. Choudhary, S. Paul, ACS Omega 6, 19529 (2021)
N. Anbu, A. Dhakshinamoorthy, J. Ind. Eng. Chem. 65, 120 (2018)
N. Seyedi, M. Zahedifar, Appl. Organomet. Chem. 35, e6364 (2021)
T. Garnier, R. Sakly, M. Danel, S. Chassaing, P. Pale, Synthesis (Stuttg). 49, 1223 (2016)
S.M. Islam, R.C. Dey, A.S. Roy, S. Paul, S. Mondal, Transit. Met. Chem. 39, 961 (2014)
Z.-H. Li, L.-P. Xue, L. Wang, S.-T. Zhang, B.-T. Zhao, Inorg. Chem. Commun. 27, 119 (2013)
Acknowledgements
One of the authors, Pharande P. S., is grateful to the Council of Scientific and Industrial Research (CSIR), New Delhi, Government of India for the award of the Senior Research Fellowship (File no. 09/816(0044)/2019-EMR-I).
Funding
No funds, grants, or other support were received.
Author information
Authors and Affiliations
Contributions
All authors contributed to design, material preparation, data collection. The first draft of the manuscript was written by P. S. Pharande. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
The authors declare that the research process did not involve any human or animal experiments.
Conflict of interest
The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Pharande, P.S., Mhaldar, P.M., Lohar, T.R. et al. A selective heterogeneous cellulose supported Schiff base Cu(II) catalyst for Chan–Evans–Lam coupling. Res Chem Intermed 49, 4541–4560 (2023). https://doi.org/10.1007/s11164-023-05089-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-023-05089-1