Abstract
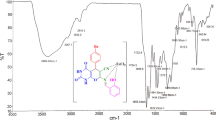
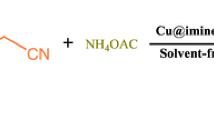
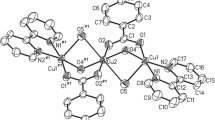
Nano-Cu-(4-chloro-3-nitrophenyl-salicylaldimine-pyranopyrimidine dione)(Cl)2 {nano-[Cu-4C3NSP](Cl)2} as a new Schiff base complex and catalyst was designed and successfully tested as a catalyst on the reaction of 7-amino-5-(4-chloro-3-nitrophenyl)-2,4-dioxo-2,3,4,5-tetrahydro-1H-pyrano[2,3-d]pyrimidine-6-carbonitrile with salicylaldehyde and Cu(Cl)2·2H2O. Nano-[Cu-4C3NSP](Cl)2 was fully characterized using Fourier transform infrared spectroscopy, energy-dispersive X-ray spectroscopy, thermal gravimetric analysis, mass spectroscopy, field emission scanning electron microscopy, SEM–EDS elemental mapping and transmission electron microscopy. {Nano-[Cu-4C3NSP](Cl)2} was successfully used as an efficient catalyst for the preparation of pyrimido[4,5-b]quinoline derivatives in high yields (73–95%).
Graphical abstract












Similar content being viewed by others
References:
I. Ali, M.N. Lone, Z.A. Al-Othman, A. Al-Warthan, M.M. Sanagi, Curr. Drug. Targ. 16, 711 (2015)
K. Saleem, W.A. Wani, A. Haque, M.N. Lone, M.-F. Hsieh, M.A. Jairajpuri, I. Ali, Future Med. Chem. 5, 135 (2013)
I. Ali, W.A. Wani, A. Haque, K. Saleem, Future Med. Chem. 5, 961 (2013)
A. Drabczynska, C.E. Muller, A. Schiedel, B. Schumacher, J.K. Wojciechowska, A. Fruzinski, W. Zobnina, O. Yuzlenko, K. Kiec-Kononowicz, Bioorg. Med. Chem. 15, 6956 (2007)
J.A. Valderrama, P. Colonelli, D. Vasquez, M.F. Gonzalez, J.A. Rodríguez, C. Theoduloz, Bioorg. Med. Chem. 16, 10172 (2008)
V. Nair, G. Chi, Q. Shu, J. Julander, D.F. Smee, Bioorg. Med. Chem. Lett. 19, 1425 (2009)
A.B.A. El-Gazzar, H.N. Hafez, G.A.M. Nawwar, Eur. J. Med. Chem. 44, 1427 (2009)
A.A. Joshi, C.L. Viswanathan, Agents Med. Chem. 5, 105 (2006)
S.I. Alqasoumi, A.M. Al-Taweel, A.M. Alafeefy, E. Noaman, M.M. Ghorab, Eur. J. Med. Chem. 45, 738 (2010)
V. Alagarsamy, Pharmazie 59, 753 (2004)
V. Alagarsamy, R. Venkatesaperumal, S. Vijayakumar, T. Angayarkanni, P. Pounammal, S. Senthilganesh, S. Kandeeban, Pharmazie 57, 306 (2002)
F. Jalili, M. Zarei, M.A. Zolfigol, S. Rostamnia, A.R. Moosavi-Zare, Microporous Mesoporous Mater. 294, 109865 (2020)
S.M. Ebrahimi, A.K. Abbas, H. Shahbazi-Alavi, J. Safaei-Ghomi, A. Bakhtiari, R. Teymuri, Res. Chem. Intermed. 47, 3963 (2021)
A.R. Moosavi-Zare, M.A. Zolfigol, M. Zarei, A. Zare, V. Khakyzadeh, J. Mol. Liq. 211, 373 (2015)
A.R. Moosavi-Zare, H. Afshar-Hezarkhani, M.M. Rezaei, Polycycl. Aromat. Compd. 40, 150 (2020)
A.R. Moosavi-Zare, H. Afshar-Hezarkhani, Org. Prep. Proced. Int. 52, 410 (2020)
A. Khazaei, F. Gohari-Ghalil, M. Tavasoli, M. Rezaei-Gohar, A.R. Moosavi-Zare, Chem. Methodol. 4, 543 (2020)
A.R. Moosavi-Zare, M.A. Zolfigol, F. Derakhshan-Panah, S. Balalaie, Mol. Catal. 449, 142 (2018)
A. Khazaei, A.R. Moosavi-Zare, H. Afshar-Hezarkhani, V. Khakyzadeh, Eurasian Chem. Commun. 2, 27 (2020)
A.R. Moosavi-Zare, H. Afshar-Hezarkhani, Eurasian Chem. Commun. 2, 465 (2020)
A.R. Moosavi-Zare, H. Goudarziafshar, Z. Jalilian, F. Hosseinabadi, Chem. Methodol. 6, 571 (2022)
A.R. Salih, Z.A.K. Al-Messri, Eurasian Chem. Commun. 3, 533 (2021)
A. Zare, M. Dianat, M.M. Eskandari, New J. Chem. 44, 4736 (2020)
F. Shirini, M.S.N. Langarudi, N. Daneshvar, M. Mashhadinezhad, N. Nabinia, J. Mol. Liq. 243, 302 (2017)
S.C. Azimi, Iran. J. Catal. 4, 113 (2014)
F. Shirini, M.S.N. Langarudi, N. Daneshvar, N. Jamasbi, M. Irankhah-Khanghah, J. Mol. Struct. 1161, 366 (2018)
K. Mohammadi, F. Shirini, A. Yahyazadeh, RSC Adv. 5, 23586 (2015)
A. Gholami, M. Mokhtary, M. Nikpassand, Appl. Organomet. Chem. 34, e6007 (2020)
A. Zare, M. Barzegar, Res. Chem. Intermed. 46, 3727 (2020)
S. Esmaili, A.R. Moosavi-Zare, A. Khazaei, RSC Adv. 12, 5386 (2022)
A. Zare, N. Lotfifar, M. Dianat, J. Mol. Struct. 1211, 128030 (2020)
F. Osanlou, F. Nemati, S. Sabaqian, Res. Chem. Intermed. 43, 2159 (2017)
Z. Karami, M.M. Khodaei, Res. Chem. Intermed. 48, 1773 (2022)
M.M. Ghorab, A.A.Y. Hassan, Phosphorus Sulfur Silicon Relat. Elem. 141, 251 (1998)
G.L. Anderson, J.L. Shim, A.D. Broom, J. Org. Chem. 41, 1095 (1976)
W. J. Coates, Eur. Pat., 351058, 1990, Chem. Abstr. 113, 40711 (1990)
D. Heber, C. Heers, U. Ravens, Pharmazie 48, 537 (1993)
A.R. Moosavi-Zare, H. Goudarziafshar, Z. Jalilian, Appl. Organometal. Chem. 33, e4584 (2019)
A. Khazaei, H.A.A. Nik, A.R. Moosavi-Zare, J. Chin. Chem. Soc. 62, 675 (2015)
H. Goudarziafshar, A.R. Moosavi-Zare, E. Khazael, Polycycl. Aromat. Compd. 42, 3606 (2022)
H. Goudarziafshar, A.R. Moosavi-Zare, F. Hosseinabadi, Z. Jalilian, Res. Chem. Intermed. 48, 1423 (2022)
A.R. Moosavi-Zare, H. Goudarziafshar, M. Yadollahi, Z. Jalilian, Polycycl. Aromat. Comd. (2022)
A.R. Moosavi-Zare, H. Goudarziafshar, P. Fashi, Res. Chem. Intermed. 46, 5567 (2020)
Funding
Funding was provided by Sayyed Jamaleddin Asadabadi University.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
There are no conflicts to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Moosavi-Zare, A.R., Goudarziafshar, H. & Bahrami, Z. Nano-[Cu-4C3NSP](Cl)2 as a new catalyst for the preparation of pyrimido[4,5-b]quinoline derivatives. Res Chem Intermed 49, 507–523 (2023). https://doi.org/10.1007/s11164-022-04901-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-022-04901-8