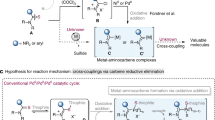
Abstract
A palladium-catalyzed Suzuki cross-coupling of benzyl ammonium triflates via activation of a Csp3–N bond to construct a Csp3–Csp2 bond is described. Various boronic acids can be coupled with a range of benzylamine-derived quaternary ammonium salts in 1:1 molar ratio by the bench-stable N-heterocyclic carbene palladium complex, SIPr-PdCl2-Py to afford diarylmethane derivatives. This reaction exhibits a wide substrate scope and functional group tolerance. Direct arylation of benzylamine in a one-pot process and the gram-scale reaction can be performed successfully.



Similar content being viewed by others
References
B. Liegault, J.L. Renaud, C. Bruneau, Chem. Soc. Rev. 37, 290 (2008)
S. Messaoudi, A. Hamze, O. Provot, B. Treguier, J. Bignon, J.M. Liu, J. Wdzieczak-Bakala, S. Thoret, J. Dubois, J.D. Brion, M. Alami, ChemMedChem 6, 488 (2011)
W.F. McCalmont, T.N. Heady, J.R. Patterson, M.A. Lindenmuth, D.M. Haverstick, L.S. Gray, T.L. Macdonald, Bioorg. Med. Chem. Lett. 14, 3691 (2004)
C. Wang, B. Wang, S. Hou, L. Xue, Z. Kang, J. Du, Y. Li, X. Liu, Q. Wang, C. Zhang, Eur. J. Med. Chem. 163, 787 (2019)
W. Cai, J. Wu, W. Liu, Y. Xie, Y. Liu, S. Zhang, W. Xu, L. Tang, J. Wang, G. Zhao, Molecules 2, 23 (2018)
P. Taslimi, H.E. Aslan, Y. Demir, N. Oztaskin, A. Maras, I. Gulcin, S. Beydemir, S. Goksu, Int. J. Biol. Macromol. 119, 857 (2018)
B. Li, Z. Chao, C. Li, Z. Gu, J. Am. Chem. Soc. 140, 9400 (2018)
X. Li, Y. Feng, L. Lin, G. Zou, J. Org. Chem. 77, 10991 (2012)
R.J. Tang, T. Milcent, B. Crousse, J. Org. Chem. 83, 14001 (2018)
S. Guduguntla, V. Hornillos, R. Tessier, M. Fananas-Mastral, B.L. Feringa, Org. Lett. 18, 252 (2016)
J. Zhang, G. Lu, J. Xu, H. Sun, Q. Shen, Org. Lett. 18, 2860 (2016)
B. Li, S.L. Fang, D.Y. Huang, B.F. Shi, Org. Lett. 19, 3950 (2017)
B.R.P. Reddy, S. Chowdhury, A. Auffrant, C. Gosmini, Adv. Synth. Catal. 360, 3026 (2018)
S. Ogoshi, H. Nakashima, K. Shimonaka, H. Kurosawa, J. Am. Chem. Soc. 123, 8626 (2001)
K. Mertins, I. Iovel, J. Kischel, A. Zapf, M. Beller, Angew. Chem. Int. Ed. 44, 238 (2004)
Q. Qin, Y. Xie, P.E. Floreancig, Chem. Sci. 9, 8528 (2018)
M. Sai, Adv. Synth. Catal. 360, 4330 (2018)
T. Mahdi, D.W. Stephan, Angew. Chem. Int. Ed. 54, 8511 (2015)
I.P. Beletskaya, F. Alonso, V. Tyurin, Coord. Chem. Rev. 385, 137 (2019)
B. Zeynizadeh, H. Mousavi, F. Sepehraddin, Res. Chem. Intermed. 46, 3361 (2020)
F. Chahdoura, C. Pradel, M. Gomez, Adv. Synth. Catal. 355, 3648 (2013)
C. Yue, Q. Xing, P. Sun, Z. Zhao, H. Lv, F. Li, Nat. Commun. 12, 1875 (2021)
A. Suzuki, Angew. Chem. Int. Ed. 50, 6722 (2011)
H. Doucet, Eur. J. Org. Chem. 12, 2013 (2008)
R. Martin, S.L. Buchwald, Acc. Chem. Res. 41, 1461 (2008)
E. Wenkert, A.L. Han, C.J. Jenny, J. Am. Chem. Soc. 14, 975 (1988)
J.T. Reeves, Org. Lett. 12, 4388 (2010)
M. Guisan-Ceinos, V. Martin-Heras, M. Tortosa, J. Am. Chem. Soc. 139, 8448 (2017)
L.G. Xie, Z.X. Wang, Angew. Chem. Int. Ed. 50, 4901 (2011)
D. Wu, J.L. Tao, Z.X. Wang, Org. Chem. Front. 2, 265 (2015)
D.Y. Wang, M. Kawahata, Z.K. Yang, K. Miyamoto, S. Komagawa, K. Yamaguchi, C. Wang, M. Uchiyama, Nat. Commun. 7, 12937 (2016)
S.B. Blakey, D.W.C. MacMillan, J. Am. Chem. Soc. 125, 6046 (2003)
P. Maity, D.M. Shacklady-McAtee, G.P. Yap, E.R. Sirianni, M.P. Watson, J. Am. Chem. Soc. 135, 280 (2013)
D.M. Shacklady-McAtee, K.M. Roberts, C.H. Basch, Y.G. Song, M.P. Watson, Tetrahedron 70, 4257 (2014)
T. Wang, S. Yang, S. Xu, C. Han, G. Guo, J. Zhao, RSC Adv. 7, 15805 (2017)
Z. Zhang, H. Wang, N. Qiu, Y. Kong, W. Zeng, Y. Zhang, J. Zhao, J. Org. Chem. 83, 8710 (2018)
M.Y. Takeshi Uemura, C. Naoto, Angew. Chem. Int. Ed. 55, 3162 (2016)
S. Yu, S. Liu, Y. Lan, B. Wan, X. Li, J. Am. Chem. Soc. 137, 1623 (2015)
M.G. Toni Moragas, M. Ruben, Angew. Chem. Int. Ed. 55, 5053 (2016)
J. Hu, H. Sun, W. Cai, X. Pu, Y. Zhang, Z. Shi, J. Org. Chem. 81, 14 (2016)
D.Y. Wang, Z.K. Yang, C. Wang, A. Zhang, M. Uchiyama, Angew. Chem. Int. Ed. 57, 3641 (2018)
C.H. Basch, J. Liao, J. Xu, J.J. Piane, M.P. Watson, J. Am. Chem. Soc. 139, 5313 (2017)
J. Liao, W. Guan, B.P. Boscoe, J.W. Tucker, J.W. Tomlin, M.R. Garnsey, M.P. Watson, Org. Lett. 20, 3030 (2018)
T. Wang, J. Guo, X. Wang, H. Guo, D. Jia, H. Wang, L. Liu, RSC Adv. 9, 5738 (2019)
E. Peris, Chem. Rev. 118, 9988 (2018)
G.C. Fortman, S.P. Nolan, Chem. Soc. Rev. 40, 5151 (2011)
C. Valente, S. Çalimsiz, K.H. Hoi, D. Mallik, M. Sayah, M.G. Organ, Angew. Chem. Int. Ed. 51, 3314 (2012)
Q. Chen, F. Gao, H. Tang, M. Yao, Q. Zhao, Y. Shi, Y. Dang, C. Cao, ACS Catal. 9, 3730 (2019)
H. Tang, M. Liu, M. Zhu, B. Cui, Y. Shi, C. Cao, Tetrahedron 98, 132431 (2021)
Q. Chen, S. Wu, S. Yan, C. Li, H. Abduhulam, Y. Shi, Y. Dang, C. Cao, ACS Catal. 10, 8168 (2020)
M. Organ, G. Chass, D.C. Fang, A. Hopkinson, C. Valente, Synthesis 17, 2776 (2008)
J. Luo, B. Hu, W. Wu, M. Hu, T.L. Liu, Angew. Chem. Int. Ed. 60, 6107 (2021)
R.J. Procter, J.J. Dunsford, P.J. Rushworth, D.G. Hulcoop, R.A. Layfield, M.J. Ingleson, Chemistry 23, 15889 (2017)
A. Vasilopoulos, S.L. Zultanski, S.S. Stahl, J. Am. Chem. Soc. 139, 7705 (2017)
P. Zhang, J. Xu, Y. Gao, X. Li, G. Tang, Y. Zhao, Synlett 25, 2928 (2014)
S. Yoon, M.C. Hong, H. Rhee, J. Org. Chem. 79, 4206 (2014)
L.K.G. Ackerman, L.L. Anka-Lufford, M. Naodovic, D.J. Weix, Chem. Sci. 6, 1115 (2015)
V. Ramakrishna, N. DastagiriReddy, Dalton Trans. 46, 8598 (2017)
G. Wu, S. Xu, Y. Deng, C. Wu, X. Zhao, W. Ji, Y. Zhang, J. Wang, Tetrahedron 72, 8022 (2016)
M. Kuriyama, M. Shinozawa, N. Hamaguchi, S. Matsuo, O. Onomura, J. Org. Chem. 79, 5921 (2014)
F. Lima, M.A. Kabeshov, D.N. Tran, C. Battilocchio, J. Sedelmeier, G. Sedelmeier, B. Schenkel, S.V. Ley, Angew. Chem. Int. Ed. 55, 14085 (2016)
Acknowledgements
We are grateful to the National Natural Science Foundation of China (21571087) for the financial support.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
None.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Zhong, C., Tang, H., Cui, B. et al. Pd-NHC catalyzed Suzuki cross-coupling of benzyl ammonium salts. Res Chem Intermed 48, 4017–4032 (2022). https://doi.org/10.1007/s11164-022-04795-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-022-04795-6