Abstract
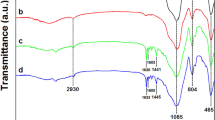
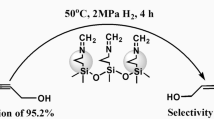
In this work, the catalytic activity of a palladium(II) isatin Schiff base complex immobilized into mesoporous silica KIT-6 (Pd-isatin Schiff base@KIT-6) was studied for the oxidation of different alcohols with tert-butyl hydroperoxide (TBHP, 70% aqueous solution) as an oxidant under solvent-free conditions. To find the suitable reaction conditions, the effect of essential factors including the solvent, temperature, catalyst amount and kind of oxidant on the oxidation of benzyl alcohol was explored. The results showed that in this catalytic system, the corresponding aldehydes and ketones were obtained with high to excellent yields at 50 °C without the formation of carboxylic acids as by-products. The catalyst was easily recovered by simple filtration and reused in five subsequent reaction cycles without any significant loss in the catalytic activity. Moreover, the comparison of the Fourier transform infrared (FT-IR) spectrum, X-ray diffraction (XRD) pattern, scanning and transmission electron microscopy (SEM and TEM) images of the used catalyst with a fresh one showed that the structure of the Pd-isatin Schiff base@KIT-6 catalyst remained intact after five times of reuse.







Similar content being viewed by others
Availability of data and material
Not applicable.
Code availability
Not applicable.
References
P. Wang, Z. Dong, Y. Lei, Y. Du, H. Li, H. Yang, Y. Nie, J. Ma, J. Porous Mater. 20, 277 (2013)
E.J. García-Suárez, M. Tristany, A.B. García, V. Collière, K. Philippot, Microporous Mesoporous Mater. 153, 155 (2012)
Bandna, N. Aggarwal, P. Das, Tetrahedron Lett. 52, 4954 (2011)
J.F. Hu, Y.P. Wang, M. Han, Y.M. Zhou, X.Q. Jiang, P.P. Sun, Catal Sci. Technol. 2, 2332 (2012)
M. Asadniaye Fardjahromi, M. Moghadam, S. Tangestaninejad, V. Mirkhani, I. Mohammadpoor-Baltork, J. Iran. Chem. Soc. 14, 1317 (2017)
S.Y. Oh, H.W. Kim, J.M. Park, H.S. Park, C. Yoon, J. Hazard. Mater. 168, 346 (2009)
A. Guðmundsson, K.E. Schlipköter, J.-E. Bäckvall, Angew. Chem. 132, 5441 (2020)
M. Hatefi Ardakani, S. Saeednia, M. Sabet, Silicon 11, 1775 (2019)
N. Chantarasiri, V. Ruangpornvisuti, N. Muangsin, H. Detsen, T. Mananunsap, C. Batiya, N. Chaichit, J. Mol. Struc. 701, 93 (2004)
H. Hosseini Monfared, R. Bikas, P. Mayer, Inorg. Chim. Acta 363, 2574 (2010)
H. Sakurai, H. Sano, T. Takino, H. Yasui, J. Inorg. Biochem. 80, 99 (2000)
M. Sarkheil, M. Lashanizadegan, M. Ghiasi, J. Mol. Struct. 1179, 278 (2019)
K.L. Reddy, A.M. Kumar, A. Dhir, V. Krishnan, J. Fluoresc. 26, 2041 (2016)
R. Pandey, L. Reddy, S. Ishihara, A. Dhir, V. Krishnan, RSC. Adv. 3, 21365 (2013)
K.L. Reddy, A.M. Kumar, A. Dhir, V. Krishnan, Int. J. Spectrosc. 2018, 1321427 (2018)
J. Shen, J. Zhang, Y. Zuo, L. Wang, X. Sun, J. Li, W. Han, R. He, J. Hazard. Mater. 163, 1199 (2009)
H.R. Mardani, M. Ziari, Res. Chem. Intermed. 44, 6605 (2018)
H.R. Saadati-Moshtaghin, F. Mohammadi Zonoz, M.M. Amini, J. Solid State Chem. 260, 16 (2018)
R.J. Kalbasi, N. Mosaddegh, C. R. Chim. 15, 988 (2012)
A. Rezaeifard, M. Jafarpour, A. Naeimi, M. Salimi, Inorg. Chem. Commun. 15, 230 (2012)
L.Y. Zhu, Z. Lou, J. Lin, W. Zheng, C. Zhang, J.D. Lou, Res. Chem. Intermed. 39, 4287 (2013)
J. Zhao, W.Y. Hernández, W. Zhou, Y. Yang, E.I. Vovk, M. Capron, V. Ordomsky, ChemCatChem 12, 238 (2020)
S. Sobhani, F. Zarifi, Chin. J. Catal. 36, 555 (2015)
S. Sobhani, Z. Mesbah Falatooni, S. Asadi, M. Honarmand, Catal. Lett. 146, 255 (2016)
A. Kumar, K. Kumar, V. Krishnan, Mater. Lett. 245, 45 (2019)
B. Qi, Y. Wang, L.L. Lou, L. Huang, Y. Yang, S. Liu, J. Mol. Catal. A: Chem. 370, 95 (2013)
H.M.A. Hassan, E.M. Saad, M.S. Soltan, M.A. Betiha, I.S. Butler, S.I. Mostafa, Appl. Catal. A: Gen. 488, 148 (2014)
S. Keshipour, N. Kalam Khalteh, J. Iran. Chem. Soc. 13, 1977 (2016)
K.A. De Castro, H. Rhee, J. Incl. Phenom. Macrocycl. Chem. 82, 13 (2015)
S. Rezaei, A. Ghorbani-Choghamarani, R. Badri, Appl. Organometal. Chem. 30, 985 (2016)
H. Veisi, R. Azadbakht, F. Saeidifar, M.R. Abdi, Catal. Lett. 147, 976 (2017)
Z. Pahlevanneshan, M. Moghadam, V. Mirkhani, S. Tangestaninejad, I. Mohammadpoor-Baltork, A.R. Khosropour, Appl. Organometal. Chem. 29, 346 (2015)
M. Nikoorazm, A. Ghorbani-Choghamarani, A. Jabbari, J. Porous Mater. 23, 967 (2016)
F. Sadegh, O. Bagheri, M. Moghadam, V. Mirkhani, S. Tangestaninejad, I. Mohammadpoor-Baltork, J. Organomet. Chem. 759, 46 (2014)
S. Chirra, S. Siliveri, A.K. Adepu, S. Goskula, S.R. Gujjula, V. Narayanan, J. Porous Mater. 26, 1667 (2019)
L. Zhang, P. Li, J. Yang, M. Wang, L. Wang, ChemPlusChem 79, 217 (2014)
S. Pakvojoud, M. Hatefi Ardakani, S. Saeednia, E. Heydari-Bafrooei, J. Sulfur Chem. 41, 561 (2020)
F. Kleitz, T.W. Kim, R. Ryoo, Bull. Korean Chem. Soc. 26, 1653 (2005)
M.J. Schultz, M.S. Sigman, Tetrahedron 62, 8227 (2006)
W. Zierkiewicz, T. Privalov, Organometallics 24, 6019 (2005)
B.A. Steinhoff, S.R. Fix, S.S. Stahl, J. Am. Chem. Soc. 124, 766 (2002)
A. Rezaeifard, P. Farshid, M. Jafarpour, G. Kardan Moghadam, RSC Adv. 4, 9189 (2014)
S.M. Islam, A.S. Roy, P. Mondal, S. Paul, N. Salam, Inorg. Chem. Commun. 24, 170 (2012)
P.B. Bhat, F. Inam, B.R. Bhat, A.C.S. Comb, Sci. 16, 397 (2014)
V. Mahdavi, M. Mardani, Res. Chem. Intermed. 41, 8907 (2015)
X. Li, Z. Zhou, Y. Zhao, D. Ramella, Y. Luan, Appl Organometal Chem. (2020). https://doi.org/10.1002/aoc.5445
Acknowledgements
The support received for this work from Vali-e-Asr University of Rafsanjan is gratefully acknowledged. The corresponding author also appreciates Professor Majid Moghadam from University of Isfahan, Dr. Zahra Pakdin-Parizi from Razavi Hospital of Mashhad, Dr. Jamileh Seyedyazdi, and Dr. Parisa Salarizadeh from Vali-e-Asr University of Rafsanjan for their valuable helps.
Funding
This research did not receive any specific grant from funding agencies.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interes
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Mousavi, D., Ardakani, M.H., Saeednia, S. et al. Solvent-free selective oxidation of alcohols with tert-butyl hydroperoxide catalyzed by palladium(II) isatin Schiff base complex supported into three-dimensional mesoporous silica KIT-6. Res Chem Intermed 47, 1105–1121 (2021). https://doi.org/10.1007/s11164-020-04319-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-020-04319-0