Abstract
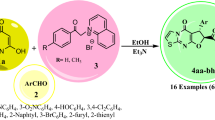
Considering benzimidazole as a privileged structure for developing probes of impressive pharmacological potentials, some new coumarin and pyrazole conjugates of benzimidazoles were synthesized with a sulphur linkage. Thus, 3-(2-oxo-2H-chromen-3-yl)-1-phenyl-1H-pyrazole-4-carbaldehyde (3) was prepared starting from simple salicylaldehyde which has been used as an important synthon and incorporated in a series of structural manipulations to obtain various of pharmacophoric motif conjugates 9(a–b) and 13(a–f), in fair yields. We have developed facile and stepwise methods for the synthesis of all these compounds in various approaches, which also involves sub-sequences. All synthesized compounds were well characterized by IR, 1HNMR, 13CNMR and mass spectra data.
Graphical Abstract











Similar content being viewed by others
References
D. Srikrishna, G. Chandraiah, P.K. Dubey, Mini-Rev. Med. Chem. 18, 113 (2018)
I.A. Kim, J.H. Shin, I.H. Kim, H.G. Wu, E.K. Chie, S.W. Ha, C.I. Park, G.D. Kao, Clin. Cancer Res. 12, 940 (2006)
W. Tang, H. Hioki, K. Harad, M. Kubo, Y. Fukuyama, J. Nat. Prod. 70, 2010 (2007)
T.H. Stefanova, N.J. Nikolova, R.A. Toshkova, H.O. Neychev, J. Exp. Ther. Oncol. 6, 107 (2007)
L. Taneja, A.K. Sharma, R.D. Singh, J. Lumin. 63, 203 (1995)
M. Yamaji, Y. Hakoda, H. Okamoto, F. Tani, Photochem. Photobiol. Sci. 16, 555 (2017)
V. Kumar, K. Kaur, G.K. Gupta, A. Sharma, Eur. J. Med. Chem. 69, 735 (2013)
H.A. Saad, N.A. Osman, A.H. Moustafa, Molecules 16, 10187 (2011)
R. Gondru, J. Banothu, R.K. Thatipamula, S.K.A. Hussain, R. Bavantula, RSC Adv. 5, 33562 (2015)
S. Viveka, P. Dinesha, G.K. Shama, S. Nagaraja, S. Ballav, S. Kerkar, Eur. J. Med. Chem. 101, 442 (2015)
N. Harikrishna, A.M. Isloor, K. Ananda, A. Obaid, H.K. Fun, RSC Adv. 5, 43648 (2015)
S. Dinesha, B. Viveka, K. Priya, K.S.R. Pai, S. Naveen, N.K. Lokanath, G.K. Nagaraja, Eur. J. Med. Chem. 104, 25 (2015)
S. Viveka, P. Dinesha, S. Shama, N.K. Naveen, Lokanath, G.K. Nagaraja, RSC Adv. 5, 94786 (2015)
R. Sridhar, P.T. Perumal, S. Etti, G. Shanmugam, M.N. Ponnuswamy, V.R. Prabavathy, N. Mathivanan, Bio. Org. Med. Chem. Lett. 14, 6035 (2004)
H. Song, Y. Liu, L. Xiong, Y. Li, N. Yang, Q. Wang, J. Agric. Food Chem. 60, 1470 (2012)
H.F. Rizk, M.A. El-Badawi, S.A. Ibrahim, M.A. El-Borai, Arab. J. Chem. 4, 37 (2011)
F. Wang, H. Duan, D. Xing, G. Yang, J. Fluoresc. 27, 1721 (2017)
J.R. Kumar, J.L. Jawahar, D.P. Pathak, Eur. J. Chem. 3, 278 (2006)
K.C. Nicolaou, A.J. Roecker, R.V. Summeren, R. Hughes, J.A. Pfefferkorn, N. Winssinger, Bioorg. Med. Chem. 11, 465 (2003)
A.T. Mavrova, D.I. Vuchev, K.K. Anichina, J.A. Tsenov, M.S. Kondeva, M.K. Micheva, Bioorg. Med. Chem. 13, 5550 (2005)
H. Al-Muhaimeed, J. Int. Med. Res. 25, 175 (1997)
S. Demirayak, A.C. Karaburun, U.A. Mohsen, Eur. J. Med. Chem. 37, 255 (2002)
P. Hao, S. Zhang, W.H. Sun, Q. Shi, S. Adewuyi, X. Lu, P. Li, Organometallics 26, 2439 (2007)
D.D. Rajadhyaksha, D.W. Rangnekar, J. Chem. Technol. Biotechnol. 36, 300 (1986)
J.A. Asensio, P. Gomez-Romero, Fuel Cells 5, 336 (2005)
P.C. Tway, L.J.C. Love, J. Phys. Chem. 26, 5223 (1982)
B. Xu, Y. Li, P. Song, F. Ma, M. Sun, Sci Rep. 7, 45688 (2017)
D.S. Strand, D. Kim, D.A. Peura, Gut and Liver 11, 27 (2017)
A. Mejia, W.K. Kraft, Expert Rev. Clin. Pharmacol. 2, 295 (2009)
C. Viegas-Junior, A. Danuello, V. Bolzani, E.J. Barreiro, C.A. Fraga, Curr. Med. Chem. 14, 1829 (2007)
G.L. Liu, Y. Hu, X.H. Chen, G.X. Wang, F. Ling, Bioorg. Med. Chem. Lett. 26, 5039 (2016)
R.K. Arora, N. Kaur, Y. Bansal, G. Bansal, Acta Pharma. Sin. B 4, 368 (2014)
R. Kenchappa, Y.D. Bodke, A. Chandrashekar, M.A. Sindhea, S.K. Peethambar, Arab. J. Chem. 10, S3895 (2017)
R.A.A. Khaled, A.A. Mohamed, H.A.H. Elshemy, N.M. Kahk, D.M. El-Amir, Lett. Drug. Des. Disc. 14, 773 (2017)
S.A. Galal, A.S. Abdelsamie, S.A. Shouman, Y.M. Attia, H.I. Ali, A. Tabll, R. El-Shenawy, Y.S. El-Abd, M.M. Ali, A.E. Mahmoud, A.H. Abdel-Halim, A.A. Fyiad, A.S. Girgis, H.I. El-Diwani, Eur. J. Med. Chem. 134, 392 (2017)
B. Mahesh, G. Mohan, S. Santhisudha, Ch. Sridevi, N.V. Reddy, T. Vijaya, C.S. Reddy, Chem. Heterocycl. Compd. 53, 173 (2017)
D. Srikrishna, S. Tasqeeruddin, P.K. Dubey, Lett. Org. Chem. 11, 556 (2014)
D. Srikrishna, P.K. Dubey, Tetrahedron Lett. 55, 6561 (2014)
D. Srikrishna, A. Mallika, P.K. Dubey, Indian J. Chem. 56B, 96 (2017)
D. Srikrishna, P.K. Dubey, New J. Chem. 41, 5168 (2017)
Acknowledgements
The authors are thankful to the authorities of Jawaharlal Nehru Technological University Hyderabad for providing laboratory facilities. They are grateful to Department of Science and Technology, Govt. of India, New Delhi, for financial support to DS in the form of INSPIRE Fellowship.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Srikrishna, D., Dubey, P.K. Synthesis of novel substituted 3-(4-((1H-benzo[d]imidazol-2-ylthio)methyl)-1-phenyl-1H-pyrazol-3-yl)-2H-chromen-2-ones: various approaches. Res Chem Intermed 44, 4455–4468 (2018). https://doi.org/10.1007/s11164-018-3397-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-018-3397-x