Abstract
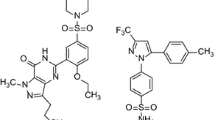
Sulfonamides have enormous importance in biological sciences. In the present investigation, the fundamental energetic group, hydrazide-based sulfonamide, has been incorporated into a naturally occurring phenolic monoterpenoid, carvacrol (5-isopropyl-2-methylphenol) moiety, with the aim of combining the active groups in order to generate more potent antimicrobial and anti-oxidant agents. Series of hydrazide-based sulfonamide derivatives (IV a–g) have been synthesized and characterized by modern sophisticated techniques such as 1H NMR, 13C NMR and LC–MS. The newly synthesized derivatives were screened for their antimicrobial activities against three fungal (Aspergillus niger, A. flavus and A. fumigatus) and three bacterial (Escherichia coli, Staphylococcus aureus and Bacillus subtilis) species, of which compounds IVc and IVd were found to have good antifungal activity and compounds IVd and IVg exhibited decent antibacterial activities. antioxidant activity was performed by DPPH radical scavenging assay. Compound IVd has been found to be more active in comparison to standard ascorbic acid and compounds IVa, IVb, and IVc demonstrated excellent antioxidant activity.
Graphical Abstract






Similar content being viewed by others
References
N. Aggarwal, R. Kumar, P. Dureja, J.M. Khurana, Synthesis of novel nalidixic acid-based 1, 3, 4-thiadiazole and 1, 3, 4-oxadiazole derivatives as potent antibacterial agents. Chem. Biol. Drug Des. 79, 384–397 (2012)
A.M. Suzan, Wamidh H. Talib, Mohammad S. Mustafa, Mohammad S. Mubarak, Murad A. AlDamen, Synthesis, characterization, and antimicrobial activity of Schiff bases derived from benzaldehydes and 3,30-diaminodipropylamine. Arab. J. Chem. 8, 850–857 (2015)
Ü.Ö. Özdemir, P. Güvenç, E. Şahin, F. Hamurcu, Synthesis, characterization and antibacterial activity of new sulfonamide derivatives and their nickel(II), cobalt(II) complexes. Inorg. Chim. Acta 362, 2613–2618 (2009)
E.H. Ali, F.I. Nassar, A.M. Badawi, S.A. Afify, Physical properties and biological applications of novel substituted biphenyl-sulfonamides. Int. J. Genet. Mol. Biol. 2, 78–91 (2010)
X. Zhang, Y. Lin, L. Liu, C. Lin, Study on the synthesis of sulfonamide derivatives and their interaction with bovine serum albumin. Luminescence 30, 269–279 (2015)
A. Alsughayer, A.Z.A. Elassar, S. Mustafa, F. Al Sagheer, Synthesis, structure analysis and antibacterial activity of new potent sulfonamide derivatives. J. Biomater. Nanobiotechnol. 2, 143 (2011)
F. Zani, P. Vicini, Antimicrobial activity of some 1, 2-benzisothiazoles having a benzenesulfonamide moiety. Arch. Pharm. 331, 219–223 (1998)
Ö. Güzel, A. Innocenti, A. Scozzafava, A. Salman, C.T. Supuran, Carbonic anhydrase inhibitors. Phenacetyl-, pyridylacetyl-and thienylacetyl-substituted aromatic sulfonamides act as potent and selective isoform VII inhibitors. Bioorg. Med. Chem. Lett. 19, 3170–3173 (2009)
A. Scozzafava, T. Owa, A. Mastrolorenzo, C.T. Supuran, Anticancer and antiviral sulfonamides. Curr. Med. Chem. 10, 925–953 (2003)
T.H. Maren, Relations between structure and biological activity of sulfonamides. Annu. Rev. Pharmacol. Toxicol. 16, 309–327 (1976)
N.S. El-Sayed, E.R. El-Bendary, S.M. El-Ashry, M.M. El-Kerdawy, Synthesis and antitumor activity of new sulfonamide derivatives of thiadiazolo [3, 2-a] pyrimidines. Eur. J. Med. Chem. 46, 3714–3720 (2011)
A.K. Tiwari, A.K. Mishra, A. Bajpai, P. Mishra, R.K. Sharma, V.K. Pandey, V.K. Singh, Synthesis and pharmacological study of novel pyrido-quinazolone analogues as anti-fungal, antibacterial, and anticancer agents. Bioorg. Med. Chem. Lett. 16, 4581–4585 (2006)
S. Yotphan, L. Sumunnee, D. Beukeaw, C. Buathongjan, V. Reutrakul, Iodine-catalyzed expeditious synthesis of sulfonamides from sulfonyl hydrazides and amines. Org. Biomol. Chem. 14, 590–597 (2016)
R.J. Lu, J.A. Tucker, T. Zinevitch, O. Kirichenko, V. Konoplev, S. Kuznetsova, S. Sviridov, J. Pickens, S. Tandel, E. Brahmachary, Y. Yang, Design and synthesis of human immunodeficiency virus entry inhibitors: sulfonamide as an isostere for the α-ketoamide group. J. Med. Chem. 50, 6535–6544 (2007)
A. Camargo-Ordoñez, C. Moreno-Reyes, F. Olazarán-Santibáñez, S. Martínez-Hernández, V. Bocanegra-García, G. Rivera, Efficient synthesis of sulfonamide derivatives on solid supports catalyzed using solvent-free and microwave-assisted methods. Quim. Nova 34, 787–791 (2011)
P.B. Molinoff, R.W. Ruddon, Goodman and Gilman’s, the Pharmacological Basis of Therapeutics (Pergamon, New York, 1996)
D.S. Doğruer, Ş. Urlu, T. Önkol, B. Özçelik, M.F. Şahin, Synthesis of some pyridazine derivatives carrying urea, thiourea, and sulfonamide moieties and their antimicrobial activity. Turk. J. Chem. 34, 57–65 (2010)
A. Kamal, M.N.A. Khan, K.S. Reddy, K. Rohini, Synthesis of a new class of 2-anilino substituted nicotinyl arylsulfonylhydrazides as potential anticancer and antibacterial agents. Bioorg. Med. Chem. 15, 1004–1013 (2007)
J.D. Rajput, S.D. Bagul, S.K. Tadavi, P.S. Karandikar, R.S. Bendre, Design, synthesis and biological evaluation of novel class diindolyl methanes (DIMs) derived from naturally occurring phenolic monoterpenoids. Med. Chem. (2016)
R.S. Bendre, J.D. Rajput, S.D. Bagul, P.S. Karandikar, Outlooks on medicinal properties of eugenol and its synthetic derivatives. Nat. Prod. Chem. Res. 4, 212 (2016). doi:10.4172/2329-6836.1000212
J. Agric. Food. Chem. 2016; 64(26): 5295–300
V.N. Mahire, V.E. Patel, A.B. Chaudhari, V.V. Gite, P.P. Mahulikar, Silane@ TiO 2 nanoparticles-driven expeditious synthesis of biologically active benzo [4, 5] imidazo [1,2-a] chromeno [4, 3-d] pyrimidin-6-one scaffolds: a green approach. J. Chem. Sci. (2016); 1–9
S. Burda, W. Oleszek, anti-oxidant and antiradical activities of flavonoids. J. Agric. Food Chem. 49, 2774–2779 (2001)
M. Özyanik, S. Demirci, H. Bektaş, N. Demirbas, A. Demirbas, Ş.A. Karaoğlu, Preparation and antimicrobial activity evaluation of some quinoline derivatives containing an azole nucleus. Turk. J. Chem. 36, 233–246 (2012)
Y. Sawada, T. Yanai, H. Nakagawa, Y. Tsukamoto, S. Yokoi, M. Yanagi, T. Toya, H. Sugizaki, Y. Kato, H. Shirakura, T. Watanabe, Synthesis and insecticidal activity of benzoheterocyclic analogues of N′-benzoyl-N-(tert-butyl) benzohydrazide: Part 1. Design of benzoheterocyclic analogues. Pest Manag. Sci. 59, 25–35 (2003)
X.H. Liu, Y.X. Shi, Y. Ma, G.R. He, W.L. Dong, C.Y. Zhang, B.L. Wang, S.H. Wang, B.J. Li, Z.M. Li, Synthesis of some N,N′-diacylhydrazine derivatives with radical-scavenging and antifungal activity. Chem. Biol. Drug Des. 73, 320–327 (2009)
G.R. Carlson, T.S. Dhadialla, R. Hunter, R.K. Jansson, C.S. Jany, Z. Lidert, R.A. Slawecki, The chemical and biological properties of methoxyfenozide, a new insecticidal ecdysteroid agonist. Pest Manag. Sci. 57, 115–119 (2001)
Y. Sawada, T. Yanai, H. Nakagawa, Y. Tsukamoto, Y. Tamagawa, S. Yokoi, M. Yanagi, T. Toya, H. Sugizaki, Y. Kato, H. Shirakura, Synthesis and insecticidal activity of benzoheterocyclic analogues of N′-benzoyl-N-(tert-butyl) benzohydrazide: Part 3. Modification of N-tert-butylhydrazine moiety. Pest Manag. Sci. 59, 49–57 (2003)
S.S. Pandit, V.U. Pandit, B.P. Bandgar, Rapid and efficient synthesis of sulfonamides from sulfonic acid and amines using cyanuric chloride-DMF adduct. J. Sulfur Chem. 29, 619–622 (2008)
O.O. Ajani, O.B. Familoni, F. Wu, J.O. Echeme, Z. Sujiang, Room temperature synthesis and antibacterial activity of new sulfonamides containing N,N-diethyl-substituted amido moieties. Int. J. Med. Chem. (2012)
A. Greenfield, C. Grosanu, Convenient synthesis of primary sulfonamides. Tetrahedron Lett. 49, 6300–6303 (2008)
Y.S. Rane, R.R. Varma, L.S. Patil, S.V. Athlekar, A.S. Chowdhary, A.S. Bobade, Synthesis and antimicrobial activity of 5-substituted-2-(1-H-benzimidazole) sulfonamides. Asian J. Res. Chem. 3, 335–338 (2010)
J.R. Soare, T.C. Dinis, A.P. Cunha, L. Almeida, anti-oxidant activities of some extracts of Thymus zygis. Free Radical Res. 26, 469–478 (1997)
Acknowledgments
The authors are grateful to UGC SAP (DSA-I), New Delhi, for financial assistance.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bagul, S.D., Rajput, J.D., Tadavi, S.K. et al. Design, synthesis and biological activities of novel 5-isopropyl-2-methylphenolhydrazide-based sulfonamide derivatives. Res Chem Intermed 43, 2241–2252 (2017). https://doi.org/10.1007/s11164-016-2759-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-016-2759-5