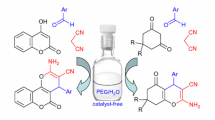
Abstract
A highly convenient and greener method for the facile synthesis of pharmaceutically diverse 2-amino-5-oxo-4-aryl-5,6,7,8-tetrahydro-4H-chromene-3-carbonitriles via a one-pot, three component condensation of aromatic aldehydes, malononitrile, and 1,3-cyclohexanedione or dimedone using 2-aminopyridine as an efficient alkaline organo catalyst is described. This new chemistry furnishes the desired heterocycles in very shorter reaction times with higher purity and has the advantages of economic viability, ease of preparation, excellent yields, and recyclability of catalyst.
Graphical Abstract





Similar content being viewed by others
References
S.L. Schreiber, Science 287, 1964 (2000)
L.F. Tietze, Chem. Rev. 96, 115 (1996)
B.M. Trost, Angew. Chem. Int. Ed. 34, 259 (1995)
G.J. Kelly, F. King, M. Kett, Green Chem. 4, 392 (2002)
J. Weitkamp, M. Hunger, U. Rymsa, Microporous Mesoporous Mater. 48, 255 (2001)
R.A. Sheldon, Pure Appl. Chem. 72, 1233 (2000)
A. Zapf, M. Beller, Top. Catal. 19, 101 (2002)
S. Bertelsen, K.A. Jorgensen, Chem. Soc. Rev. 38, 2178 (2009)
F. Giacalone, M. Gruttadauria, P. Agrigento, R. Noto, Chem. Soc. Rev. 41, 2406 (2012)
M.J. Gaunt, C.C. Johansson, A. McNally, N.T. Vo, Drug Discov. Today 12, 8 (2007)
P.A. Tempest, Curr. Opin. Drug Discov. 8, 776 (2005)
H. Fujioka, K. Murai, O. Kubo, Y. Ohba, Y. Kita, Org. Lett. 9, 1687 (2007)
N.M. Evdokimov, A.S. Kireev, A.A. Yakovenko, M.Y. Antipin, I.V. Magedov, A. Kornienko, J. Org. Chem. 72, 3443 (2007)
X.S. Wang, Q. Li, J.R. Wu, Y.L. Li, C.S. Yao, S.J. Tu, Synthesis 12, 1902 (2008)
P.A. Wender, S.T. Handy, D.L. Wright, Chem. Ind. 19, 765 (1997)
X. Li, Y. Zhao, H. Qu, Z. Mao, X. Lin, Chem. Commun. 49, 1401 (2013)
N. Erdmann, A.R. Philipps, I. Atodiresei, D. Enders, Adv. Synth. Catal. 355, 847 (2013)
F. Shi, W. Tan, R.Y. Zhu, G.J. Xing, S.J. Tu, Adv. Synth. Catal. 355, 1605 (2013)
X. Meng, C. Yu, P. Zhao, RSC Adv. 4, 8612 (2014)
X. Meng, Y. Wang, C. Yu, P. Zhao, RSC Adv. 4, 27301 (2014)
J. Skommer, D. Wlodkowic, M. Matto, M. Eray, J. Pelkonen, Leuk. Res. 30, 322 (2006)
W. Kemnitzer, S. Kasibhatla, S. Jiang, H. Zhang, J. Zhao, S. Jia, L. Xu, G.C. Grundy, R. Denis, N. Barriault, L. Vaillancourt, S. Charron, J. Dodd, G. Attardo, D. Labrecque, S. Lamothe, H. Gourdeau, B. Tseng, J. Drewe, S.X. Cai, Bioorg. Med. Chem. Lett. 15, 4745 (2005)
H. Gourdeau, L. Leblond, B. Hamelin, C. Desputeau, K. Dong, S. Kianicka, B. Tseng, Mol. Cancer Ther. 3, 1375 (2004)
G.A. Reynolds, K.H. Drexhage, Opt. Commun. 13, 222 (1975)
H. Zollinger, Color Chemistry, 3rd edn. (Verlag Helvetica Chimica Acta and Wiley, Zurikh and Weinheim, 2003)
E.R. Bissell, A.R. Mitchell, R. Smith, Eur. J. Org. Chem. 45, 2283 (1980)
G.P. Ellis, Chemistry of heterocyclic of compounds, in A. Weissberger, E.C. Taylor, (John Wiley, New York, 1977)
E.A. Hafez, M.H. Elnagdi, A.G. Elagamey, F.M.A. Taweel, Heterocycles 26, 903 (1987)
M.G. Dekamin, M. Eslami, A. Maleki, Tetrahedron 69, 1074 (2013)
D.M. Pore, K.A. Undale, B.B. Dongare, U.V. Desai, Catal. Lett. 132, 104 (2009)
A. Hasaninejad, M. Shekouhy, N. Golzar, A. Zare, M.M. Doroodmand, Appl. Catal. A 402, 11 (2011)
V.M. Joshi, R.L. Magar, P.M. Throat, S.U. Tekale, B.R. Patil, M.P. Kale, R.P. Pawar, Chin. Chem. Lett. 25, 455 (2014)
S. Khaksar, A. Rouhollahpour, S.M. Talesh, J. Fluor. Chem. 141, 11 (2012)
A. Hasaninejad, N. Golzar, M. Beyrati, A. Zare, M.M. Doroodmand, J. Mol. Catal. A Chem. 372, 137 (2013)
K. Niknam, N. Borazjani, R. Rashidian, A. Jamali, Chin. J. Catal. 34, 2245 (2013)
R.Y. Guo, Z.M. An, L.P. Mo, R.Z. Wang, H.Z. Liu, S.X. Wang, Z.H. Zhang, ACS Comb. Sci. 15, 557 (2013)
S.M. Baghbanian, N. Rezaei, H. Tashakkorian, Green Chem. 15, 3446 (2013)
M. Gupta, V.K. Gupta, New J. Chem. 39, 3578 (2015)
A.F. Shojaei, K. Tabatabaeian, F. Shirini, S.Z. Hejazi, RSC Adv. 4, 9509 (2014)
O.H. Qareaghaja, S. Mashkouria, M.R.N. Jamala, G. Kaupp, RSC Adv. 4, 48191 (2014)
A.T. Khan, M. Lal, S. Ali, M. Musawwer, Khan. Tetrahedron Lett. 52, 5327 (2011)
I. Devi, P.J. Bhuyan, Tetrahedron Lett. 45, 8625 (2004)
R. Ramesh, A. Lalitha, RSC Adv. 5, 51188 (2015)
R. Ramesh, S. Maheswari, S. Murugesan, R. Sandhiya, A. Lalitha, Res. Chem. Intermed. 41, 8233 (2015)
R. Ramesh, A. Lalitha, Res. Chem. Intermed. 41, 8009 (2015)
Acknowledgments
R. R. gratefully acknowledge the DST-Inspire Fellowship, New Delhi, India (No: DST/INSPIRE Fellowship/2012/690) for financial support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ramesh, R., Vadivel, P., Maheswari, S. et al. Click and facile access of substituted tetrahydro-4H-chromenes using 2-aminopyridine as a catalyst. Res Chem Intermed 42, 7625–7636 (2016). https://doi.org/10.1007/s11164-016-2557-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-016-2557-0