Abstract
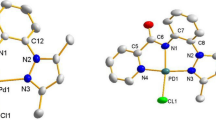
Two new palladium complexes derived from the di(2-pyridinyl)methanone N-(2-pyridinyl)hydrazone (DPMNPH) ligand are reported. The compounds were characterized by elemental analysis, spectroscopic studies, and, for the DPMNPH ligand, single-crystal X-ray diffraction analysis. The DPMNPH ligand crystallized as orthorhombic with the space group P212121. The H1 atom is intramolecularly bonded to the pyridinic N4 with N1–H1 = 0.92(3) Å, H1···N4 = 1.87(2) Å, N1···N4 = 2.615(2) Å, and N1–H1···N4 = 137(2)°. Both complexes were excellent catalysts in the Heck reaction in the presence of base.






Similar content being viewed by others
References
M.C. Feller, R. Robson, Aust. J. Chem. 21, 2919 (1968)
A.C. Casado, G. Barandika, B. Bazan, M.-K. Urtiagaa, M.-I. Arriortuaa, CrystEngComm 15, 5134–5143 (2013)
C. Godard, S.B. Dyckett, S. Parsons, R.N. Perutz, Chem. Commun. 18, 23332 (2004)
C. Mock, I. Puscasu, M.J. Rauterkus, G. Tallen, J.E.A. Wolf, B. Krebs, Inorg. Chim. Acta 319, 109 (2001)
A. C. Deveson, S. L. Hearth, C. J. Harding, A. K. Powell, J. Chem. Soc. Dalton Trans. 3173 (1996). http://pubs.rsc.org/en/content/articlelanding/1996/dt/dt9960003173#!divAbstract
S. Kar, N. Chanda, S.M. Mobin, F.A. Urbanos, M. Niemeyer, V.G. Puranik, R. Jimenez-Aparico, G.K. Lahiri, Inorg. Chem. 44, 1571 (2005)
J.F. Berry, F.A. Cotton, L.M. Daniels, C.A. Murillo, X. Wang, Inorg. Chem. 42, 2418 (2003)
Y. Zhang, X.-Z. Sun, Y.-H. Deng, S.-L. Cao, C.-Q. Wan, Inorg. Chim. Acta 421, 411–415 (2014)
J. Tsuji, Palladium Reagents and Catalysts. Chapter 3 (Wiley, New York, 2004)
F. Diederich, P.J. Stang, Metal-Catalyzed Cross-Coupling Reactions (VCH, Weinheim, 1997)
N. Shahnaz, B. Banik, P. Das, Tetrahedron Lett. 54, 2886 (2013)
M. Keles, H. Keles, D.M. Emir, Appl. Organometal. Chem. 29, 543–548 (2015)
M.N. Alam, S.M. Sarkar, M.R. Miah, Catal. Lett 98, 383–389 (2009)
M. Bakherad, A. Keivanloo, S. Samangooei, Tetrahedron Lett. 53(2012), 5773–5776 (2012)
I. Warad, M. Azam, S.I. Al-Resayes, M.S. Khan, P. Ahmad, M. Al-Nuri, S. Jodeh, A. Husein, S.F. Haddad, B. Hammouti, M. Al-Noaimi, Inorg. Chem. Commun. 43, 155–161 (2014)
M. Bagherzadeh, M. Amini, A. Ellern, L.K. Woo, Inorg. Chim. Acta 383, 46 (2012)
CrysAlisPro, Agilent Technologies, Version 1.171.35.11 (release 16-05-2011 CrysAlis171.NET) (compiled May 16 2011)
O.V. Dolomanov, L.J. Bourhis, R.J. Gildea, J.A.K. Howard, H. Puschmann, OLEX2: a complete structure solution, refinement and analysis program. J. Appl. Cryst. 42, 339 (2009)
G.M. Sheldrick, SHELXTL version 6.10, Structure Determination Software Suite (Bruker AXS Inc., Madison, 2000)
K. Nakamoto, Infrared and Raman Spectra of Inorganic and Coordination Compounds (Wiley, New York, 1986)
L.J. Baucher, J. Inorg. Nucl. Chem. 36, 531 (1974)
G.R. Garcia, N.A. Lopez, J.G.A. Rodriguez, D.M. Otero, R.F. Esparzo, M.F. Alamo, Polyhedron Polyhedron 30, 1324 (2011)
V.L. Siji, M.R. Sudarsanakumar, S. Suma, Polyhedron 29, 2035 (2010)
A.B.P. Lever, Inorganic Electronic Spectroscopy, 2nd edn. (Elsevier, New York, 1984)
M. Ulusoy, O. Birel, O. Sahin, O. Buyukgungor, B. Cetinkya, Polyhedron 38, 141 (2012)
F. Alono, I.P. Beletskaya, M. Yus, Tetrahedron 61, 11771 (2005)
G.P. Chiusoli, M. Catellani, M. Costa, E. Motti, N.D. Ca, G. Maestri, Coord. Chem. Rev. 254, 456 (2010)
Acknowledgments
The authors would like to extend their sincere appreciation to the Dean of Scientific Research at King Saud University for funding this work through research group number RG -1435-091.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Warad, I., Azam, M., Al-Resayes, S.I. et al. Palladium complexes bearing the dipyridyl ligand: synthesis, structural studies, and use in the Heck reaction. Res Chem Intermed 42, 379–389 (2016). https://doi.org/10.1007/s11164-015-2256-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-015-2256-2