Abstract
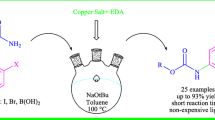
A ligand-free, powerful, and practical method for mono and polymethoxylation of unactivated aryl bromides has been developed; CuCl was used as catalyst, HCOOMe as cocatalyst, and methanolic MeONa as both nucleophile and solvent. This eco-friendly procedure is characterized by operational simplicity, inexpensive substrates (unactivated mono to polybromoarenes), full conversion, and direct recovery of pure MeOH.





Similar content being viewed by others
References
F. Ullmann, Ber. Dtsch. Chem. Ges. 37, 853 (1904)
R.G.R. Bacon, S.C. Rennison, J. Chem. Soc. C 312 (1969)
J. Lindley, Tetrahedron 40, 1433 (1984)
H.L. Aalten, G.V. Koten, D.M. Grove, T. Kuilman, O.G. Piekstra, L.A. Hulshof, R.A. Sheldon, Tetrahedron 45, 5565 (1989)
M.A. Keegstra, T.H.A. Peters, L. Brandsma, Tetrahedron 48, 3633 (1992)
P. Capdevielle, M. Maumy, Tetrahedron Lett. 34, 1007 (1993)
J. Buckingham, Dictionary of Natural Products (Cambridge University Press, Cambridge, 1994), pp. 157–181
J.E. Leet, R. Schroeder, D.R. Langley, K.L. Colson, S. Huang, S.E. Klohr, M.S. Lee, J. Golik, S.J. Hofstead, T.W. Doyle, J.A. Matson, J. Am. Chem. Soc. 115, 8432 (1993)
A.W. Czarnik, Acc. Chem. Res. 29, 112 (1996)
S. Kawata, M. Hirama, Tetrahedron Lett. 39, 8707 (1998)
W.X. Gu, X.B. Jing, X.F. Pan, A.S.C. Chan, T.K. Yang, Tetrahedron Lett. 41, 6079 (2000)
T. Temal-Laib, J. Chastanet, J.P. Zhu, J. Am. Chem. Soc. 124, 583 (2002)
Z.L. Shen, X.Z. Jiang, W.M. Mo, B.X. Hu, N. Sun, Green Chem. 7, 97 (2005)
Y.F. Ji, Z.M. Zong, X.Y. Wei, Org. Prep. Proc. Int. 35, 225 (2003)
A. Sankaranarayanan, S.B. Chandalia, Org. Process Res. Dev. 10, 487 (2006)
W.D. Zhou, E.W. Scocchera, D.L. Wright, A.C. Anderson, Med. Chem. Commun. 4, 908 (2013)
D. Barton, K. Nakanishi, O. Meth-Cohn, Comprehensive Natural Products Chemistry, vols. 1, 3, 8 (Elsevier Science, Oxford, 1999)
G.W. Klumpp, Reactivity in Organic Chemistry (Wiley, New York, 1982)
V. Snieckus, Chem. Rev. 90, 879 (1990)
R. Taylor, Electrophilic Aromatic Substitution (Wiley, New York, 1995)
A.R. Muci, S.L. Buchwald, Top. Curr. Chem. 219, 131 (2002)
W.J. Moorman, H.W. Ahlers, R.E. Chapin, G.P. Daston, P.M.D. Foster, R.J. Kavlock, J.S. Morawetz, T.M. Schnorr, S.M. Schrader, Reprod. Toxicol. 14, 293 (2000)
D. Nobel, J. Chem. Soc. Chem. Commun. 419 (1993)
P.J. Fagan, E. Hauptman, R. Shapiro, A. Casalnuovo, J. Am. Chem. Soc. 122, 5043 (2000)
M. Wolter, G. Nordmann, G.E. Job, S.L. Buchwald, Org. Lett. 4, 973 (2002)
R. Hosseinzadeh, M. Tajbakhsh, M. Mohadjerani, M. Alikarami, Synlett 16, 1101 (2005)
H. Zhang, D.W. Ma, W.G. Cao, Synlett 18, 243 (2007)
R.A. Altman, A. Shafir, A. Choi, P.A. Lichtor, S.L. Buchwald, J. Org. Chem. 73, 284 (2008)
J.J. Niu, H. Zhou, Z.G. Li, J.W. Xu, S.J. Hu, J. Org. Chem. 73, 7814 (2008)
A.B. Naidu, G. Sekar, Tetrahedron Lett. 49, 3147 (2008)
A.B. Naidu, E.A. Jaseer, G. Sekar, J. Org. Chem. 74, 3675 (2009)
W.B. Huang, C.Y. Du, J.A. Jiang, Y.F. Ji, Res. Chem. Intermed. 39, 2849 (2013)
J.G. Huang, Y. Guo, H.W. Liu, Y.F. Ji, Res. Chem. Intermed. (2014). doi:10.1007/s11164-014-1801-8
J.A. Jiang, J.L. Du, D.H. Liao, Z.G. Wang, Y.F. Ji, Tetrahedron Lett. 55, 1406 (2014)
J.A. Jiang, C. Chen, J.G. Huang, H.W. Liu, S. Cao, Y.F. Ji, Green Chem. 16, 1248 (2014)
J.A. Jiang, C. Chen, Y. Guo, D.H. Liao, X.D. Pan, Y.F. Ji, Green Chem. 16, 2807 (2014)
J.F. Carpentier, Y. Castanet, J. Brocard, A. Mortreux, F. Petit, Tetrahedron Lett. 32, 4705 (1991)
O. Jogunola, T. Salmi, M. Kangas, J.P. Mikkola, Chem. Eng. J. 203, 469 (2012)
M. Hakansson, S. Jagner, Inorg. Chem. 29, 5241 (1990)
T. Cohen, J. Wood, A.G. Dietz, Tetrahedron Lett. 40, 3555 (1974)
J. Sicé, J. Am. Chem. Soc. 75, 3697 (1953)
M.N. Mohamad Ibrahim, R.B. Sriprasanthi, S. Shamsudeen, F. Adam, S.A. Bhawani, BioResources 7, 4377 (2012)
Y.F. Ji, J.A. Jiang, H.W. Liu, D.H. Liao, X.Y. Wei, Syn. Comm. 43, 1517 (2013)
L. Pouységu, M. Marguerit, J. Gagnepain, G. Lyvinec, A.J. Eatherton, S. Quideau, Org. Lett. 10, 5211 (2008)
P.N. Chatterjee, A.K. Maity, S.S. Mohapatra, S. Roy, Tetrahedron Lett. 69, 2816 (2013)
N.M. Thomson, K.D. Seibert, S. Tummala, S. Bordawekar, W.F. Kiesman, E.A. Irdam, B. Phenix, D. Kumke, Org. Process Res. Dev. (2014). doi:10.1021/op500187u
M.L. Bender, Chem. Rev. 60, 53 (1960)
R.A. McClelland, G. Patel, J. Am. Chem. Soc. 103, 6912 (1981)
S.V. Ley, A.W. Thomas, Angew. Chem. Int. Ed. 42, 5400 (2003)
F. Monnier, M. Taillefer, Angew. Chem. Int. Ed. 48, 6954 (2009)
Acknowledgments
The authors are grateful to the National Natural Science Foundation of China (project nos 21176074 and 21476074), the Research Fund for the Doctoral Program of Higher Education of China (project no. 20130074110009), and the Shandong Rongyuan Pharmaceutical Co. Ltd. for financial support.
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Guo, Y., Ji, SZ., Chen, C. et al. A ligand-free, powerful, and practical method for methoxylation of unactivated aryl bromides by use of the CuCl/HCOOMe/MeONa/MeOH system. Res Chem Intermed 41, 8651–8664 (2015). https://doi.org/10.1007/s11164-014-1917-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-014-1917-x