Abstract
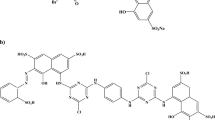
Removal of Reactive Red 120 (RR120) dye in aqueous solution was investigated using immobilized TiO2 in the presence of UV light. TiO2 was deposited onto microscope slides by heat attachment method. SEM analyses confirmed that all microscope slides were coated with TiO2 particles completely after single, two, three and four layers coating. The removal efficiency increased with repetitive coating up to three layers. The highest removal efficiency of 94% was obtained at pH 3 within 300 min of reaction time. Complete removal was observed with 5 and 10 mg dm−3 dye concentrations whereas removal efficiency decreased from 96 to 52% when initial RR120 concentration increased 15 mg dm−3 to 30 mg dm−3. The results of nonlinear optimization showed that the kinetics of removal of RR120 in the range of concentration studied described by L–H kinetic expression. The experiments conducted with radical scavengers indicated that hydroxyl radicals and photogenerated hole and electrons were main reactive species in photocatalytic removal of RR120. The photonic efficiency of immobilized system was lower than that of suspended system. Minor drop (6%) in the removal of RR120 with three layers coated slide after four consecutive cycles of 300 min indicated the reusability and stability of coated slides. A possible degradation mechanism for degradation of RR120 was proposed according to detected intermediate compounds. Mineralization of RR120 was monitored by generation of NO3− and SO42− ions in solution during photocatalytic reaction.






Similar content being viewed by others
Data availability
The datasets generated or analysed during the current study are available from the corresponding author on reasonable request.
References
Oller I, Malato S, Sanchez-Perez JA (2011) Combination of advanced oxidation processes and biological treatments for wastewater decontamination—a review. Sci Total Environ 409:4141–4166. https://doi.org/10.1016/j.scitotenv.2010.08.061
Bilinska L, Gmurek M, Ledakowicz S (2016) Comparison between industrial and simulated textile wastewater treatment by AOPs—biodegradability, toxicity and cost assessment. Chem Eng J 306:550–559. https://doi.org/10.1016/j.cej.2016.07.100
Selveraj V, Karthika TS, Mansiya C, Alagar M (2021) An over review on recently developed techniques, mechanisms and intermediate involved in the advanced azo dye degradation for industrial applications. J Mol Struct 1224:129195. https://doi.org/10.1016/j.molstruc.2020.129195
Al-Mamuna MR, Kaderb S, Islamb MS, Khan MZH (2019) Photocatalytic activity improvement and application of UV-TiO2 photocatalysis in textile wastewater treatment: a review. J Environ Chem Eng 7:103248. https://doi.org/10.1016/j.jece.2019.103248
Ghoreishian SM, Badii K, Norouzi M, Rashidi A, Montazer M, Sadeghi M, Vafaee M (2014) Decolorization and mineralization of an azo reactive dye using loaded nano-photocatalysts on spacer fabric: kinetic study and operational factors. J Taiwan Inst Chem Eng 45:2436–2446. https://doi.org/10.1016/j.jtice.2014.04.015
Zhou K, Hu X-Y, Chen B-Y, Hsueh C-C, Zhang Q, Wang J, Lin Y-J, Chan C-T (2016) Synthesized TiO2/ZSM-5 composites used for the photocatalytic degradation of azo dye: intermediates, reaction pathway, mechanism and bio-toxicity. Appl Surf Sci 383:300–309. https://doi.org/10.1016/j.apsusc.2016.04.155
Verma AK, Dash RR, Bhunia PA (2012) Review on chemical coagulation/flocculation technologies for removal of colour from textile wastewaters. J Environ Manag 93:154–168. https://doi.org/10.1016/j.jenvman.2011.09.012
Ajmal A, Majeed I, Malik RN, Idriss H, Nadeem MA (2014) Principles and mechanisms of photocatalytic dye degradation on TiO2 based photocatalysts: a comparative overview. RCS Adv 4:37003–37026. https://doi.org/10.1039/C4RA06658H
Din MI, Khalid R, Hussain Z (2018) Minireview: silver-doped titanium dioxide and silver-doped zinc oxide photocatalysts. Anal Lett 51:897–907. https://doi.org/10.1080/00032719.2017.1363770
Juang R-S, Lin S-H, Hsueh P-Y (2010) Removal of binary azo dyes from water by UV-irradiated degradation in TiO2 suspensions. J Hazard Mater 182:820–826. https://doi.org/10.1016/j.jhazmat.2010.06.113
Lambropoulou DA, Konstantinou IK, Albanis TA, Fernandez-Alba AR (2011) Photocatalytic degradation of the fungicide fenhexamid in aqueous TiO2 suspensions: identification of intermediates products and reaction pathways. Chemosphere 83:367–378. https://doi.org/10.1016/j.chemosphere.2010.12.006
Gupta VK, Jain R, Mittal A, Saleh TA, Nayak A, Agarwal S, Sikarwar S (2012) Photo-catalytic degradation of toxic dye amaranth on TiO2/UV in aqueous suspensions. Mater Sci Eng C 32:12–17. https://doi.org/10.1016/j.msec.2011.08.018
Antonopoulou M, Konstantinou I (2014) Photocatalytic treatment of metribuzin herbicide over TiO2 aqueous suspensions: removal efficiency, identification of transformation products, reaction pathways and ecotoxicity evaluation. J Photochem Photobiol A 294:110–120. https://doi.org/10.1016/j.jphotochem.2014.08.008
Wetchakun K, Wetchakun N, Sakulsermsuk S (2019) An overview of solar/visible light-driven heterogeneous photocatalysis for water purification: TiO2- and ZnO-based photocatalysts used in suspension photoreactors. J Ind Eng Chem 71:19–49. https://doi.org/10.1016/j.jiec.2018.11.025
Khataee AR, Pons MN, Zahraa O (2009) Photocatalytic degradation of three azo dyes using immobilized TiO2 nanoparticles on glass plates activated by UV light irradiation: influence of dye molecular structure. J Hazard Mater 168:451–457. https://doi.org/10.1016/j.jhazmat.2009.02.052
Chong MN, Tneu ZY, Poh PE, Jin B, Aryal R (2015) Synthesis, characterization and application of TiO2–zeolite nanocomposites for the advanced treatment of industrial dye wastewater. J Taiwan Inst Chem Eng 50:288–296. https://doi.org/10.1016/j.jtice.2014.12.013
Srikanth B, Goutham R, Narayan RB, Ramprasath A, Gopinath KP (2017) Recent advancements in supporting materials for immobilised photocatalytic applications in waste water treatment. J Environ Manag 200:60–78. https://doi.org/10.1016/j.jenvman.2017.05.063
He Y, Sutton NB, Rijnaarts HHH, Langenhoff AAM (2016) Degradation of pharmaceuticals in wastewater using immobilized TiO2 photocatalysis under simulated solar irradiation. Appl Catal B 182:132–141. https://doi.org/10.1016/j.apcatb.2015.09.015
Cherif LY, Yahiaoui I, Aissani-Benissad F, Madi K, Benmehdi N, Fourcade F, Amrane A (2014) Heat attachment method for the immobilization of TiO2 on glass plates: application to photodegradation of Basic Yellow dye and optimization of operating parameters using response surface methodology. Ind Eng Chem Res 53:3813–3819. https://doi.org/10.1021/ie403970m
Khataee AR (2009) Photocatalytic removal of C.I. Basic Red 46 on immobilized TiO2 nanoparticles: artificial neural network modelling. Environ Technol 30(11):1155–1168
Sampaio MJ, Silva CG, Silva AMT, Vilar VJP, Boaventura RAR, Faria JL (2013) Photocatalytic activity of TiO2-coated glass raschig rings on the degradation of phenolic derivatives under simulated solar light irradiation. Chem Eng J 224:32–38. https://doi.org/10.1016/j.cej.2012.11.027
Jouali A, Salhi A, Aguedach A, Aarfane A, Ghazzaf H, Lhadi EK, El Krati M, Tahiri S (2019) Photo-catalytic degradation of Methylene Blue and Reactive Blue 21 dyes in dynamic mode using TiO2 particles immobilized on cellulosic fibers. J Photochem Photobiol A 338:1112013. https://doi.org/10.1016/j.jphotochem.2019.112013
Rodriguez P, Meille V, Pallier S, Sawah MAA (2009) Deposition and characterisation of TiO2 coatings on various supports for structured (photo)catalytic reactors. Appl Catal A 360:154–162. https://doi.org/10.1016/j.apcata.2009.03.013
Sathishkumar P, Mangalaraja RV, Anandan S, Ashokkumar M (2013) CoFe2O4/TiO2 nanocatalysts for the photocatalytic degradation of Reactive Red 120 in aqueous solutions in the presence and absence of electron acceptors. Chem Eng J 220:302–310. https://doi.org/10.1016/j.cej.2012.11.027
Velmurugan R, Krishnakumar B, Swaminathan M (2014) Synthesis of Pd Co-doped nano-TiO2-SO42– and its synergetic effect on the solar photodegradation of Reactive Red 120 dye. Mater Sci Semicond Process 25:163–172. https://doi.org/10.1016/j.mssp.2013.10.024
Suwannaruang T, Rivera KKP, Neramittagapong A, Wantala K (2015) Effects of hydrothermal temperature and time on uncalcined TiO2 synthesis for Reactive Red 120 photocatalytic degradation. Surf Coat Technol 271:192–200. https://doi.org/10.1016/j.surfcoat.2014.12.041
Turhan-Özdemir GD, Kartal OE (2018) Determination of electrical energy cost of decolorization of Reactive Red 120 via heterogeneous photocatalysis with TiO2. J Pharm Chem Biol Sci 6(3):122–128
Zhang Y, Xiao Y, Zhang J, Chang VWC, Lim T-T (2017) Degradation of cyclophosphamide and 5-fluorouracil in water using UV and UV/H2O2: kinetics investigation, pathways and energetic analysis. J Environ Chem Eng 5:1133–1139. https://doi.org/10.1016/j.jece.2017.01.013
Dijkstra MFJ, Michorius A, Buwalda H, Panneman HJ, Winkelman JGM, Beenackers AACM (2001) Comparison of the efficiency of immobilized and suspended systems in photocatalytic degradation. Catal Today 66:487–494. https://doi.org/10.1016/S0920-5861(01)00257-7
Bouarioua A, Zerdaoui M (2017) Photocatalytic activities of TiO2 layers immobilized on glass substrates by dip-coating technique toward the decolorization of Methyl Orange as a model organic pollutant. J Environ Chem Eng 5:1565–1574. https://doi.org/10.1016/j.jece.2017.02.025
Hegedus P, Szabó-Bárdos E, Horvátha O, Szabó P, Horváth K (2017) Investigation of a TiO2 photocatalyst immobilized with poly(vinyl alcohol). Catal Today 284:179–186. https://doi.org/10.1016/j.cattod.2016.11.050
Adamek E, Baran W, Sobczak A (2015) Effect of FeCl3 on the photocatalytic processes initiated by UVa and vis light in the presence of TiO2-P25. Appl Catal B 172–173:139–144. https://doi.org/10.1016/j.apcatb.2015.02.025
Ahmad R, Ahmad Z, Khan AU, Mastoi NR, Aslam M, Kim J (2006) Photocatalytic systems as an advanced environmental remediation: recent developments, limitations and new avenues for applications. J Environ Chem Eng 4:4143–4164. https://doi.org/10.1016/j.jece.2016.09.009
Jamil TS, Sharaf El-Deen SEA (2016) Removal of persistent tartrazine dye by photodegradation on TiO2 nanoparticles enhanced by immobilized calcinated sewage sludge under visible light. Sep Sci Technol 51(10):1744–1756. https://doi.org/10.1080/01496395.2016.1170036
Zangeneh H, Zinatizadeh AAL, Habibi M, Akia M, Isa MH (2015) Photocatalytic oxidation of organic dyes and pollutants in wastewater using different modified titanium dioxides: a comparative review. J Ind Eng Chem 26:1–36. https://doi.org/10.1016/j.jiec.2014.10.043
Gaya UI, Abdullah AH (2008) Heterogeneous photocatalytic degradation of organic contaminants over titanium dioxide: a review of fundamentals, progress and problems. J Photochem Photobiol C 9:1–12. https://doi.org/10.1016/j.jphotochemrev.2007.12.003
Zeghioud H, Khellaf N, Djelal H, Amrane A, Bouhelassa M (2016) Photocatalytic reactors dedicated to the degradation of hazardous organic pollutants: kinetics, mechanistic aspects, and design—a review. Chem Eng Commun 203:1415–1431. https://doi.org/10.1080/00986445.2016.1202243
Chen Y, Yang S, Wang K, Lou L (2005) Role of primary active species and TiO2 surface characteristic in UV-illuminated photodegradation of Acid Orange 7. J Photochem Photobiol A 172:47–54. https://doi.org/10.1016/j.jphotochem.2004.11.006
Kumar KV, Porkodi K, Selvaganapathi A (2007) Constrain in solving Langmuir-Hinshelwood kinetic expression for the photocatalytic degradation of Auramine O aqueous solutions by ZnO catalyst. Dyes Pigm 75(1):246–249. https://doi.org/10.1016/j.dyepig.2006.05.035
Karakaplan N, Goz E, Tosun E, Yuceer M (2019) Kinetic and artificial neural network modeling techniques to predict the drying kinetics of Mentha spicata L. J Food Process Preserv 43(10):e14142. https://doi.org/10.1111/jfpp.14142
Kisch H, Bahneman D (2015) Best practice in photocatalysis: comparing rates or apparent quantum yields? J Phys Chem Lett 6:1907–1910. https://doi.org/10.1021/acs.jpclett.5b00521
Lee CM, Palaniandy P, Dahlan I (2017) Pharmaceutical residues in aquatic environment and water remediation by TiO2 heterogeneous photocatalysis: a review. Environ Earth Sci 76:611. https://doi.org/10.1007/s12665-017-6924-y
Dostanic J, Hus M, Loncarevic D (2020) Effect of substituents in hydroxyl radical-mediated degradation of azo pyridone dyes: theoretical approaches on the reaction mechanism. J Environ Sci 98:14–21. https://doi.org/10.1016/j.jes.2020.05.022
Naik AP, Salkar AV, Majik MS, Morajkar PP (2017) Enhanced photocatalytic degradation of amaranth dye on mesoporous anatase TiO2: evidence of C-N, N=N bond cleavage and identification of new intermediates. Photochem Photobiol Sci 16:1126–1138. https://doi.org/10.1039/C7PP00090A
Galindo C, Jacques P, Kalt A (2000) Photodegradation of the aminoazobenzene acid orange 52 by three advanced oxidation processes:UV/H2O2, UV/TiO2 and VIS/TiO2 comparative mechanistic and kinetic investigation. J Photochem Photobiol A 130:35–47. https://doi.org/10.1016/S1010-6030(99)00199-9
Paul J, Kadam AA, Govindvar SP, Kumar P, Varshney L (2013) An insight into the influence of low dose irradiation pretreatment on the microbial decolouration and degradation of Reactive Red-120 dye. Chemosphere 90:1348–1358. https://doi.org/10.1016/j.chemosphere.2012.07.049
Konstantinou IK, Albanis TA (2004) TiO2-Assisted photocatalytic degradation of azo dyes in aquoeous solution: kinetic and mechanistic investigations: a review. Appl Catal B 49:1–14. https://doi.org/10.1016/j.apcatb.2003.11.010
Zhang Q, Jing YH, Shiue A, Chang C-T, Chen B-Y, Hsueh C-C (2012) Deciphering effects of chemical structure on azo dye decolorization/degradation characteristics: bacterial vs. photocatalytic method. J Taiwan Inst Chem Eng 43:760–766. https://doi.org/10.1016/j.jtice.2012.03.001
Camarillo R, Rincon J (2011) Photocatalytic discoloration of dyes: relation between effect of operating parameters and dye structure. Chem Eng Technol 34:1675–1684. https://doi.org/10.1002/ceat.201100063
Stylidi M, Kondarides DI, Verykios XE (2004) Visible light-induced photocatalytic degradation of Acid Orange 7 in aqueous TiO2 suspensions. Appl Catal B 47:189–201. https://doi.org/10.1016/j.apcatb.2003.09.014
Funding
No funding was received for conducting this study.
Author information
Authors and Affiliations
Contributions
All authors contributed to the study conception and design. Material preparation, data collection and analysis and writing—the first draft of the manuscript were performed by HK. Conceptualization, methodology, validation, writing—review and editing of the manuscript and supervision were performed by OEK. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Karakurt, H., Kartal, O.E. Removal of Reactive Red 120 using immobilized TiO2 in the presence of UV light. Reac Kinet Mech Cat 135, 2153–2173 (2022). https://doi.org/10.1007/s11144-022-02230-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-022-02230-w