Abstract
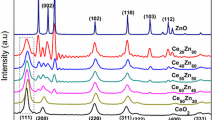
The present study investigates the effect of different catalyst preparation methods (namely, co-precipitation, COP; sol gel, SG; urea gelation, UG; and solution combustion, SC) on the characteristics and performance of a non-noble metal based supported heterogeneous catalyst (CuO/CeO2) for the treatment of synthetic wastewater. To test their efficacy, two wastewater solutions were selected, i.e. inorganic: sulfide-laden wastewater, S2− concentration = 250 mg/L and organic: phenolic wastewater (phenol and o-cresol with total phenolic concentration = 1500 mg/L). The fresh catalyst samples were characterized for particle size distribution using sieves (25–125 µm), transmission electron microscopy, X-ray diffraction, X-ray photoelectron spectroscopy, BET surface area and ICP-AES. The recovered catalysts were also subjected to different analyses to understand the changes in the catalyst during reaction. Maximum oxidation of sulfide and phenolic compounds occurred with H2O2 in the presence of the catalyst prepared by SC method. This method also resulted in the most stable catalyst showing low amount of active metal leaching (Cu = 2% and Ce = 0.2%) as compared to COP (25.8, 7.0%), SG (38.3, 31.3%) and UG (39.6, 26.5%) methods, for which Cu and Ce leaching were ~ 26–40 and 7–31%, respectively.








Similar content being viewed by others
References
Ali E, Rahman M, Sarkar SM, Bee S, Hamid A (2014) J Nanomater 209:1–23
Levec J, Pintar A (2007) Catal Today 124:172–184
Mata-Alvarez J, Macé S, Llabrés P (2000) Bioresour Technol 74:3–16
Pirkanniemi K, Sillanpää M (2002) Chemosphere 48:1047–1060
Hočevar S, Krašovec UO, Orel B, Aricó AS, Kim H (2000) Appl Catal B Environ 28:113–125
Liu W, Flytzani-Stephanopoulos M (1996) Chem Eng J 64:283–294
Garg A, Mishra A (2013) J Hazard Toxic Radioact 17:89–96
Yadav BR, Garg A (2012) Ind Eng Chem Res 51:15778–15785
Bera P, Aruna ST, Patil KC, Hegde MS (1999) J Catal 186:36–44
Liu Y, Fu Q, Stephanopoulos MF (2004) Catal Today 93–95:241–246
Hariz IB, Monser L (2014) Int Water Technol J 4:264–267
Kulkarni SJ (2013) Int J Sci Res 3:1–5
Heidarinasab A, Hashemi SR (2011) Int Conf Chem Ecol Environ Sci (ICCEES)
Oprea F, Fendu EM, Nicolae M, Pantea O, Dunka M (2010) Rev Chim 61:608–666
Hawari A, Ramadan H, Abu-Reesh I, Ouederni M (2015) J Environ Manage 151:105–112
Schieder D, Schneider R, Bischof F (2000) Water Sci Technol 41:181–187
Kavitha V, Palanivelu K (2004) Chemosphere 55:1235–1243
Kavitha V, Palanivelu K (2005) Water Res 39:3062–3072
Kang YW, Hwang KY (2000) Water Res 34:2786–2790
Patterson AL (1939) Phys Rev 56:978–982
Packer AP, Larivière D, Li C, Chen M, Fawcett A, Nielsen K, Mattson K, Chatt A, Scriver C, Erhardt LS (2007) Anal Chim Acta 588:166–172
APHA (2012) Standard methods for the examination of water and wastewater, 22nd edn. American Public Health Association, Washington
Alves AK, Bergmann CP, Berutti FA (2013) Novel synthesis and characterization of nanostructured materials. Springer, Berlin
Avgouropoulos G, Ioannides T, Matralis H (2005) Appl Catal B Environ 56:87–93
Krylova V, Andruleviçius M (2009) Int J Photoenergy 2009
Perez-Benito JF (2001) Monatshefte Chem 132:1477–1492
Acknowledgements
The authors acknowledge the support of the Sophisticated Analytical Instrument Facility (SAIF), Indian Institute of Technology Bombay (IIT Bombay), Central Facility, Department of Chemistry, IIT Bombay and Metallurgical Engineering & Materials Science Department, IIT Bombay, for helping us in the catalyst characterization.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Gupta, D., Garg, A. Effect of the preparation method on the catalytic activity of the heterogeneous catalyst CuO/CeO2 for the oxidative degradation of sulfide and phenolic compounds. Reac Kinet Mech Cat 124, 101–121 (2018). https://doi.org/10.1007/s11144-017-1318-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-017-1318-4