Abstract
Purpose
To find out which symptoms most frequently and severely affect breast cancer patients during radiotherapy and how patients manage the symptoms and unmet needs.
Methods
A cross-sectional survey was conducted with 111 patients who receive radiotherapy for breast cancer from January to April 2015 at Samsung Medical Center in Seoul, South Korea. Participants were asked about symptoms and discomfort due to radiotherapy, management methods for radiation dermatitis, unmet needs for radiation dermatitis care, and clinical and socio-demographic information.
Results
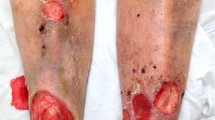
Of total, 108 out of 111 patients (97.3%) reported symptoms related to radiation dermatitis. Hyperpigmentation was the most commonly reported uncomfortable symptom followed by erythema. On average, patients reported 8.6 radiotherapy-induced skin problems (range, 0–11). Of total, 59 (53.2%) patients stated that they wanted care for radiation dermatitis, and 80.0, 59.4, and 51% of patients searched for information, used products, and visited the hospital to manage radiotherapy-related skin problems. Patients who experienced dryness, burning feelings, irritation, roughness, and hyperpigmentation were 11.73, 7.02, 5.10, 4.27, and 2.80 times more likely to have management needs than patients without those symptoms, respectively, adjusting age, current cycle of radiation therapy, chemotherapy, and type of surgery.
Conclusions
Most of the breast cancer patients experience multiple symptoms associated with radiation dermatitis. Hyperpigmentation was the most common and uncomfortable symptom followed by erythema. Majority of patients wanted management for radiation dermatitis and patients who experienced dryness, burning feelings, irritation, roughness, and hyperpigmentation had higher needs for radiation dermatitis management.

Similar content being viewed by others
References
Waghmare, C. M. (2013). Radiation burn–from mechanism to management. Burns, 39(2), 212–219.
Ha, B., Suh, H. S., Lee, J., Lee, K. J., Lee, R., & Moon, B. I. (2013). Long-term results of forward intensity-modulated radiation therapy for patients with early-stage breast cancer. Radiation Oncology Journal, 31(4), 191–198.
Hymes, S. R., Strom, E. A., & Fife, C. (2006). Radiation dermatitis: Clinical presentation, pathophysiology, and treatment 2006. Journal of the American Academy of Dermatology, 54(1), 28–46.
Chan, R. J., Webster, J., Chung, B., Marquart, L., Ahmed, M., & Garantziotis, S. (2014). Prevention and treatment of acute radiation-induced skin reactions: A systematic review and meta-analysis of randomized controlled trials. BMC Cancer, 14, 53.
Neben-Wittich, M. A., Atherton, P. J., Schwartz, D. J., Sloan, J. A., Griffin, P. C., Deming, R. L., Anders, J. C., Loprinzi, C. L., Burger, K. N., Martenson, J. A., & Miller, R. C. (2011). Comparison of provider-assessed and patient-reported outcome measures of acute skin toxicity during a Phase III trial of mometasone cream versus placebo during breast radiotherapy: The North Central Cancer Treatment Group (N06C4). International Journal of Radiation Oncology, Biology, Physics, 81(2), 397–402.
Basch, E., Iasonos, A., McDonough, T., Barz, A., Culkin, A., Kris, M. G., Scher, H. I., & Schrag, D. (2006). Patient versus clinician symptom reporting using the National Cancer Institute Common Terminology Criteria for Adverse Events: results of a questionnaire-based study. The Lancet Oncology, 7(11), 903–909.
Chen, A. P., Setser, A., Anadkat, M. J., Cotliar, J., Olsen, E. A., Garden, B. C., & Lacouture, M. E. (2012). Grading dermatologic adverse events of cancer treatments: the Common Terminology Criteria for Adverse Events Version 4.0. Journal of the American Academy of Dermatology, 67(5), 1025–1039.
Harsolia, A., Kestin, L., Grills, I., Wallace, M., Jolly, S., Jones, C., Lala, M., Martinez, A., Schell, S., & Vicini, F. A. (2007). Intensity-modulated radiotherapy results in significant decrease in clinical toxicities compared with conventional wedge-based breast radiotherapy. International Journal of Radiation Oncology, Biology, Physics, 68(5), 1375–1380.
Kang, H. C., Ahn, S. D., Choi, D. H., Kang, M. K., Chung, W. K., & Wu, H. G. (2014). The safety and efficacy of EGF-based cream for the prevention of radiotherapy-induced skin injury: results from a multicenter observational study. Radiation Oncology Journal, 32(3), 156–162.
Faithfull, S., Lemanska, A., & Chen, T. (2015). Patient-reported outcome measures in radiotherapy: Clinical advances and research opportunities in measurement for survivorship. Clinical Oncology, 27(11), 679–685.
FDA. (2006). Draft guidance for industry on patient-reported outcome measures: Use in medicinal product development to support labeling claims. Fed Register, 71, 5862–5863.
Kluetz, P. G., Chingos, D. T., Basch, E. M., & Mitchell, S. A. (2016). Patient-reported outcomes in cancer clinical trials: Measuring symptomatic adverse events with the national cancer institute’s patient-reported outcomes version of the common terminology criteria for adverse events (PRO-CTCAE). American Society of Clinical Oncology Educational Book, 35, 67–73.
Vacek, P. M., Winstead-Fry, P., Secker-Walker, R. H., Hooper, G. J., & Plante, D. A. (2003). Factors influencing quality of life in breast cancer survivors. Quality of Life Research, 12(5), 527–537.
Harris, R., Probst, H., Beardmore, C., James, S., Dumbleton, C., Bolderston, A., Faithfull, S., Wells, M., & Southgate, E. (2012). Radiotherapy skin care: A survey of practice in the UK. Radiography, 18(1), 21–27.
O’Donovan, A., Coleman, M., Harris, R., & Herst, P. (2015). Prophylaxis and management of acute radiation-induced skin toxicity: a survey of practice across Europe and the USA. European Journal of Cancer Care, 24(3), 425–435.
Bolderston, A., Lloyd, N. S., Wong, R. K., Holden, L., Robb-Blenderman, L., & Supportive Care Guidelines Group of Cancer Care Ontario Program in Evidence-Based, C. (2006). The prevention and management of acute skin reactions related to radiation therapy: a systematic review and practice guideline. Support Care Cancer, 14(8), 802–817.
Atherton, P. J., Burger, K. N., Loprinzi, C. L., Neben Wittich, M. A., Miller, R. C., Jatoi, A., & Sloan, J. A. (2012). Using the Skindex-16 and common terminology criteria for adverse events to assess rash symptoms: Results of a pooled-analysis (N0993). Supportive Care in Cancer, 20(8), 1729–1735.
Huang, C. J., Hou, M. F., Luo, K. H., Wei, S. Y., Huang, M. Y., Su, S. J., Kuo, H. Y., Yuan, S. S., Chen, G. S., Chu-Sung Hu, S., & Chuang, H. Y. (2015). RTOG, CTCAE and WHO criteria for acute radiation dermatitis correlate with cutaneous blood flow measurements. Breast, 24(3), 230–236.
Bottomley, A., Jones, D., & Claassens, L. (2009). Patient-reported outcomes: Assessment and current perspectives of the guidelines of the Food and Drug Administration and the reflection paper of the European Medicines Agency. European Journal of Cancer, 45(3), 347–353.
Kumar, S., Juresic, E., Barton, M., & Shafiq, J. (2010). Management of skin toxicity during radiation therapy: A review of the evidence. Journal of Medical Imaging and Radiation Oncology, 54(3), 264–279.
Rutqvist, L. E., Rose, C., & Cavallin-ståhl, E. (2003). A systematic overview of radiation therapy effects in breast cancer. Acta Oncologica, 42(5–6), 532–545.
Szumacher, E., Wighton, A., Franssen, E., Chow, E., Tsao, M., Ackerman, I., Andersson, L., Kim, J., Wojcicka, A., Ung, Y., Sixel, K., & Hayter, C. (2001). Phase II study assessing the effectiveness of Biafine cream as a prophylactic agent for radiation-induced acute skin toxicity to the breast in women undergoing radiotherapy with concomitant CMF chemotherapy. International Journal of Radiation Oncology, Biology, Physics, 51(1), 81–86.
McGale, P., Taylor, C., Correa, C., Cutter, D., Duane, F., Ewertz, M., Gray, R., Mannu, G., Peto, R., Whelan, T., Wang, Y., Wang, Z., & Darby, S. (2014). Effect of radiotherapy after mastectomy and axillary surgery on 10-year recurrence and 20-year breast cancer mortality: meta-analysis of individual patient data for 8135 women in 22 randomised trials. Lancet, 383(9935), 2127–2135.
Darby, S., McGale, P., Correa, C., Taylor, C., Arriagada, R., Clarke, M., Cutter, D., Davies, C., Ewertz, M., Godwin, J., Gray, R., Pierce, L., Whelan, T., Wang, Y., & Peto, R. (2011). Effect of radiotherapy after breast-conserving surgery on 10-year recurrence and 15-year breast cancer death: meta-analysis of individual patient data for 10,801 women in 17 randomised trials. Lancet, 378(9804), 1707–1716.
Lee, S. Y., Kwon, H. C., Kim, J. S., & Lee, H.-K. (2010). An analysis of the incidence and related factors for radiation dermatitis in breast cancer patients who received radiation therapy. The Journal of the Korean Society for Therapeutic Radiology and Oncology, 28(1), 16.
Harper, J. L., Franklin, L. E., Jenrette, J. M., & Aguero, E. G. (2004). Skin toxicity during breast irradiation: pathophysiology and management. Southern Medical Journal, 97(10), 989–993.
Vogelzang, N. J., Breitbart, W., Cella, D., Curt, G. A., Groopman, J. E., Horning, S. J., Itri, L. M., Johnson, D. H., Scherr, S. L., & Portenoy, R. K. (1997). Patient, caregiver, and oncologist perceptions of cancer-related fatigue: results of a tripart assessment survey. The Fatigue Coalition. Seminars in Hematology, 34(3 Suppl 2), 4–12.
Fromme, E. K., Eilers, K. M., Mori, M., Hsieh, Y. C., & Beer, T. M. (2004). How accurate is clinician reporting of chemotherapy adverse effects? A comparison with patient-reported symptoms from the Quality-of-Life Questionnaire C30. Journal of Clinical Oncology 22(17), 3485–3490.
Patrick, D. L., Ferketich, S. L., Frame, P. S., Harris, J. J., Hendricks, C. B., Levin, B., Link, M. P., Lustig, C., McLaughlin, J., Ried, L. D., Turrisi, A. T. 3rd, Unutzer, J., & Vernon, S. W. (2003). National institutes of health state-of-the-science conference statement: Symptom management in cancer: Pain, depression, and fatigue, July 15–17, 2002. Journal of the National Cancer Institute, 95(15), 1110–1117.
Noh, J. M., Choi, D. H., Park, H., Huh, S. J., Park, W., Seol, S. W., Jeong, B. K., Nam, S. J., Lee, J. E., & Kil, W. H. (2014). Comparison of acute skin reaction following morning versus late afternoon radiotherapy in patients with breast cancer who have undergone curative surgical resection. Journal of Radiation Research, 55(3), 553–558.
Montazeri, A. (2008). Health-related quality of life in breast cancer patients: a bibliographic review of the literature from 1974 to 2007. Journal of Experimental & Clinical Cancer Research: CR, 27, 32.
Ebanks, J. P., Wickett, R. R., & Boissy, R. E. (2009). Mechanisms regulating skin pigmentation: the rise and fall of complexion coloration. The International Journal of Molecular Sciences, 10(9), 4066–4087.
McQuestion, M. (2006). Evidence-based skin care management in radiation therapy. Seminars in Oncology Nursing, 22(3), 163–173.
Lee, J. H., Kay, C. S., Maeng, L. S., Oh, S. J., Lee, A. H., Lee, J. D., Han, C. W., & Cho, S. H. (2009). The clinical features and pathophysiology of acute radiation dermatitis in patients receiving tomotherapy. Annals of Dermatology, 21(4), 358–363.
Hu, S. C., Hou, M. F., Luo, K. H., Chuang, H. Y., Wei, S. Y., Chen, G. S., Chiang, W., & Huang, C. J. (2014). Changes in biophysical properties of the skin following radiotherapy for breast cancer. The Journal of Dermatology, 41(12), 1087–1094.
Salvo, N., Barnes, E., van Draanen, J., Stacey, E., Mitera, G., Breen, D., Giotis, A., Czarnota, G., Pang, J., & De Angelis, C. (2010). Prophylaxis and management of acute radiation-induced skin reactions: a systematic review of the literature. Current Oncology, 17(4), 94–112.
Richardson, J., Smith, J. E., McIntyre, M., Thomas, R., & Pilkington, K. (2005). Aloe Vera for preventing radiation-induced skin reactions: A systematic literature review. Clinical Oncology, 17(6), 478–484.
Funding
There were no external funding sources for this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
Authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the Institutional Review Board of Samsung Medical Center, and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
All study participants provided written informed consent.
Rights and permissions
About this article
Cite this article
Lee, J., Park, W., Choi, D.H. et al. Patient-reported symptoms of radiation dermatitis during breast cancer radiotherapy: a pilot study. Qual Life Res 26, 1713–1719 (2017). https://doi.org/10.1007/s11136-017-1526-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-017-1526-4