Abstract
Purpose
Long-term levodopa therapy and related fluctuating plasma concentrations are associated with between-dose periods of ‘off time’ resulting in substantial variation in symptoms and functioning throughout the day in people with Parkinson’s (PwP).
Methods
PwP across UK, France, Spain and Italy completed an online survey to explore: the impact of ‘off time’ on (1) health-related quality of life (HRQL) and (2) on functioning and ability to undertake usual activities; (3) the value of ‘off time’ relative to other factors associated with Parkinson’s through a stated preference discrete choice experiment (SPDCE).
Results
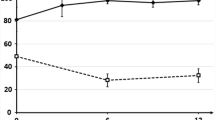
In total, 305 PwP completed the online survey. Overall mean HRQL (utility) score was significantly lower for ‘off time’ (0.37) than for ‘on time’ (0.60). All attributes within the SPDCE were significant predictors of treatment choice, although increased duration of ‘on time’ (per hour per day: odds ratio (OR) = 1.40) and predictability of ‘off time’ to within 30 min (OR = 1.42) were valued most highly.
Conclusions
‘On time’ and predictability of ‘off time’ are highly valued by PwP. Due to substantial diurnal variation of Parkinson’s symptoms, standard patient-reported outcome (PRO) assessments may not adequately capture the impact of ‘off time’ on HRQL and participation in daily activities.


Similar content being viewed by others
Notes
Sample excludes 30 pilots, includes those failing the logic and consistency checks.
References
Weintraub, D., Comella, C. L., & Stacy, H. (2010). Parkinson’s disease—Part 1: Pathophysiology, symptoms, burden, diagnosis, and assessment. The American Journal of Managed Care, 14, S40–S48.
Müller, T. (2013). Pharmacokinetic considerations for the use of levodopa in the treatment of Parkinson’s disease: Focus on levodopa/carbidopa/entacapone for treatment of levodopa-associated motor complications. Clinical Neuropharmacology, 36, 84–91.
Antonini, A., Martinez-Martin, P., Chaudhuri, R. K., Merello, M., Hauser, R., Katzenschlager, R., et al. (2011). Wearing-off scales in Parkinson’s disease: Critique and recommendations. Movement Disorders, 26(12), 2169–2175.
Stocchi, F. (2006). The levodopa wearing-off phenomenon in Parkinson’s disease: Pharmacokinetic considerations. Expert Opinion on Pharmacotherapy, 7, 1399–1407.
Hauser, R. A. (2012). IPX066: A novel carbidopa-levodopa extended-release formulation. Expert Review of Neurotherapeutics, 12, 133–140.
Huot, P., Johnston, T. H., Koprich, J. B., Fox, S. H., & Brotchie, J. M. (2013). The pharmacology of L-DOPA-induced dyskinesia in Parkinson’s disease. Pharmacological Reviews, 65, 171–222.
Gomez-Esteban, J. C., Zarranz, J. J., Lezcano, E., Tijero, B., Luna, A., Velasco, F., et al. (2007). Influence of motor symptoms upon the quality of life of patients with Parkinson’s disease. European Neurology, 57, 161–165.
Montel, S., Bonnet, A. M., & Bungener, C. (2009). Quality of life in relation to mood, coping strategies, and dyskinesia in Parkinson’s disease. Journal of Geriatric Psychiatry, 22(2), 95–102.
Rahman, S., Griffin, H. J., Quinn, N. P., & Jahanshahi, M. (2008). Quality of life in Parkinson’s disease: The relative importance of the symptoms. Movement Disorders, 23(10), 1428–1434.
Palmer, C. S., Schmier, J. K., Snyder, E., & Scott, B. (2000). Patient preferences and utilities for ‘off-time’ outcomes in the treatment of Parkinson’s disease. Quality of Life Research, 9(7), 819–827.
Herdman, M., Gudex, C., Lloyd, A., Janssen, M., Kind, P., Parkin, D., et al. (2011). Development and preliminary testing of the new five-level version of EQ-5D (EQ-5D-5L). Quality of Life Research, 20, 1727–1736.
Jenkinson, C., Fitzpatrick, R., Peto, V., Greenhall, R., & Hyman, N. (1997). The Parkinson’s Disease Questionnaire (PDQ-39): Development and validation of Parkinson’s disease summary index score. Age and Ageing, 26, 353–357.
Braun, V., & Clarke, V. (2006). Using thematic analysis in psychology. Qualitative Research in Psychology, 3, 77–101.
Fahn, S., & Elton, R. L. (1987). Members of the UPDRS Development Committee. The United Parkinson’s Disease Rating Scale. In S. Fahn, C. D. Marsden, D. B. Caine, & M. Goldstein (Eds.), Recent developments in Parkinson’s disease, Vol 2 (pp. 153-163–293-304). Florham Park: Macmillian Healthcare Information.
McIntosh, E. (2006). Using stated preference discrete choice experiments in cost-benefit analysis: Some considerations. Pharmacoeconomics, 24(9), 855–869.
McIntosh, E., Clarke, P., Frew, E., & Louviere, J. J. (2010). Applied methods of cost-benefit analysis in health care. In A. Gray & A. Briggs (Eds.), Handbooks in health economic evaluation. Oxford: Oxford University Press.
Ryan, M., & Gerard, K. (2003). Using discrete choice experiments to value health care programmes: Current practice and future research reflections. Applied health economics and health policy, 2(1), 55–64.
Bridges, J. F., Hauber, A. B., Marshall, D., Lloyd, A., Prosser, L. A., Regier, D. A., et al. (2011). Conjoint analysis applications in health—a checklist: A report of the ISPOR Good Research Practices for Conjoint Analysis Task Force. Value in Health, 14(4), 403–413.
Petrou, S., & McIntosh, E. (2009). Using discrete choice experiments to elicit women’s preferences for alternative forms of miscarriage management. Value in Health, 12(4), 551–559.
Ossa, D. F., Briggs, A., McIntosh, E., Cowell, W., Littlewood, T., & Sculpher, M. (2007). Using direct utility elicitation and discrete choice studies to assess the impact of chemotherapy-related anaemia on the health-related quality-of-life of cancer, and the value of rHu-EPO treatment. Pharmacoeconomics, 25(3), 223–237.
Lloyd, A., McIntosh, E., Rabe, K., & Williams, A. (2007). Patient preferences for asthma therapy: A discrete choice experiment. Primary Care Respiratory Journal, 16(4), 241–248.
Kleinman, L., McIntosh, E., Ryan, M., Schmier, J., Crawley, J., Locke, G. R., & de Lissovoy, G. (2002). Willingness to pay for complete symptom relief of gastroesophageal reflux disease. Archives of Internal Medicine, 162, 1361–1366.
Ryan, M., McIntosh, E., Dean, T., & Old, P. (2000). Trade offs between location and waiting times in the provision of health care: The case of elective surgery on the Isle of Wight. Journal of Public Health Medicine, 22(2), 202–210.
Hattori, N., Fujimoto, K., Kondo, T., Murata, M., & Stacy, M. (2012). Patient perspectives on Parkinson’s disease therapy in Japan and the United States: Results of two patient surveys. Patient Related Outcomes Measures, 3, 31–38.
Burgess, L., & Street, D. J. (2005). Optimal designs for choice experiments with asymmetric attributes. Journal of Statistical Physics, 134(1), 288–301.
van Hout, B., Janssen, M. F., Feng, Y. S., Kohlmann, T., Busschbach, J., Golicki, D., et al. (2012). Interim scoring for the EQ-5D-5L: Mapping the EQ-5D-5L to EQ-5D-3L value sets. Value in Health, 15(5), 708–715.
Lancsar, E., & Louviere, J. (2008). Conducting discrete choice experiments to inform healthcare decision making. Pharmacoeconomics, 26, 661–677.
Johnson, F. R., Hauber, B., Ozdemir, S., Siegel, C. A., Hass, S., & Sands, B. E. (2010). Are gastroenterologists less tolerant of treatment risks than patients? Benefit-risk preferences in Crohn’s disease management. Journal of Managed Care Pharmacy, 16(8), 616–628.
Walters, S. J., & Brazier, J. E. (2005). Comparison of the minimally important difference for two health state utility measures: EQ-5D and SF-6D. Quality of Life Research, 14(6), 1523–1532.
Pickard, A. S., Neary, M. P., & Cella, D. (2007). Estimation of minimally important differences in EQ-5D utility and VAS scores in cancer. Health Qual Life Outcomes, 5, 70.
Chapuis, S., Ouchchane, L., Metz, O., Gerbaud, L., & Durif, F. (2005). Impact of the motor complications of Parkinson’s disease on the quality of life. Movement Disorders, 20(2), 224–230.
Haahr, A., Kirkevold, M., Hall, E. O., & Østergaard, K. (2011). Living with advanced Parkinson’s disease: A constant struggle with unpredictability. Journal of Advanced Nursing, 67(2), 408–417.
Marras, C., Lang, A., Krahn, M., Tomlinson, G., & Naglie, G. (2004). Quality of life in early Parkinson’s disease: Impact of dyskinesias and motor fluctuations. Movement Disorders, 19(1), 22–28.
Gómez-Esteban, J. C., Tijero, B., Somme, J., Ciordia, R., Berganzo, K., Rouco, I., et al. (2011). Impact of psychiatric symptoms and sleep disorders on the quality of life of patients with Parkinson’s disease. Journal of Neurology, 258(3), 494–499.
Forsaa, E. B., Larsen, J. P., Wentzel-Larsen, T., Herlofson, K., & Alves, G. (2008). Predictors and course of health-related quality of life in Parkinson’s disease. Movement Disorders, 23(10), 1420–1427.
Goetz, C. G., Tilley, B. C., Shaftman, S. R., Stebbins, G. T., Fahn, S., Martinez-Martin, P., et al. (2008). Movement Disorder Society-sponsored revision of the Unified Parkinson’s Disease Rating Scale (MDS-UPDRS): Scale presentation and clinimetric testing results. Movement Disorders, 23(15), 2129–2170.
Morley, D., Dummett, S., Kelly, L., Dawson, J., Fitzpatrick, R., & Jenkinson, C. (2013). Development of the Oxford Participation & Activities Questionnaire: Semi structured interviews with potential users. Journal of the Neurological Sciences, 333, e651.
Acknowledgments
European Parkinson’s Disease Association and Parkinson’s UK reviewed qualitative stage materials/methods and assisted with participant recruitment for qualitative interviews.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
GlaxoSmithKline funded this study (HO-11-802), which was conducted by ICON plc. Helen Smith and James Cooper are employees and shareholders of GlaxoSmithKline. Emily Lloyd was an employee of GlaxoSmithKline at the time of manuscript development and is a shareholder of GlaxoSmithKline. Charlotte Kosmas is an employee of ICON plc. Cicely Kerr and Karissa Johnston were employees of ICON plc at the time of manuscript development. Andrew Lloyd is a consultant for ICON plc and also receives some financial support from EuroQol group and the journal Value in Health where he is a co-editor. Emma McIntosh is an employee of University of Glasgow and is funded in part by Parkinson’s UK (Parkinson’s UK Research Fellow). The views expressed are those of the authors and not the funding bodies or Parkinson’s UK.
Additional information
Cicely Kerr was working at ICON Patient Reported Outcomes, Oxford, UK, at the time the work was conducted.
Karissa Johnston was working at ICON Epidemiology, Vancouver, Canada, at the time the work was conducted.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kerr, C., Lloyd, E.J., Kosmas, C.E. et al. Health-related quality of life in Parkinson’s: impact of ‘off’ time and stated treatment preferences. Qual Life Res 25, 1505–1515 (2016). https://doi.org/10.1007/s11136-015-1187-0
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-015-1187-0