Abstract
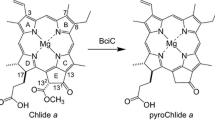
Chlorophyll-a (Chl-a) was extracted from cyanobacterial cells and modified to methyl pyropheophorbide-a. The 3-vinyl-chlorin was transformed to zinc complex of the corresponding 3-acetyl-porphyrin. The zinc porphyrin was oxidized to give cis-7,8- and 17,18-dihydroxy-chlorins as well cis-7,8-cis-17,18-tetrahydroxybacteriochlorin. After zinc-demetallation, the isolated cis-7,8- and 17,18-diols were reduced at the 3-acetyl group and triply dehydrated under acidic conditions to afford two regioisomeric 3-vinyl-porphyrins, methyl divinyl-pyroprotopheophorbide-a possessing the 8-vinyl group and 17-propionate residue (one of the divinyl-protoChl-a derivatives) and methyl pyropheophorbide-c 1 possessing the 8-ethyl group and 17-acrylate residue (one of the Chl-c 1 derivatives), respectively. The resulting 7,8,17,18-tetrol was reduced and then acidically treated, giving five-fold dehydrated free base porphyrin, methyl pyropheophorbide-c 2 possessing the 3,8-divinyl groups and 17-acrylate residue (one of the Chl-c 2 derivatives). The visible absorption and fluorescence emission spectra of the three semi-synthetic 3-vinyl-porphyrins in dichloromethane were compared with those of the corresponding 8-ethyl-porphyrin bearing the 17-propionate residue, methyl pyroprotopheophorbide-a (one of the protoChl-a derivatives). The Soret and Qy absorption maxima were shifted to longer wavelengths with an increase of π-conjugation in a molecule: protoChl-a (8-CH2CH3/17-CH2CH2COOCH3) < divinyl-protoChl-a (8-CH=CH2/17-CH2CH2COOCH3) < Chl-c 1 (8-CH2CH3/17-CH=CHCOOCH3) < Chl-c 2 derivatives (8-CH=CH2/17-CH=CHCOOCH3). The 171,172-dehydrogenation broadened the absorption bands. The emission maxima were bathochromically shifted in the same order. The reaction mechanism of the present dehydration indicates that the biosynthetic pathway of Chls-c would include the hydroxylation of the 17-propionate reside at the 171-position and successive dehydration to the 17-acrylate residue.





Similar content being viewed by others
Abbreviations
- APCI:
-
Atmospheric pressure chemical ionization
- CD:
-
Circular dichroism
- Chl:
-
Chlorophyll
- DV:
-
Divinyl
- FCC:
-
Flash column chromatography
- HR:
-
High resolution
- MS:
-
Mass spectra
- ODS:
-
Octadecylated silica gel
- PChl-a H :
-
Protochlorophyllide-a
- pPhe:
-
Pyropheophorbide
- pPPhe:
-
Pyroprotopheophorbide
- pTSA:
-
p-Toluenesulfonic acid
- RP:
-
Reversed-phase
References
Álvarez S, Zapata M, Garrido JL, Vaz B (2012) Characterization of [8-ethyl]-chlorophyll c 3 from Emiliania huxleyi. Chem Commun 48:5500–5502
Álvarez S, Rodríguez F, Riobó P, Garrido JL, Vaz B (2013) Chlorophyll c CS-170 isolated from Ostreococcus sp. is [7-methoxycarbonyl-8-vinyl]protochlorophyllide a. Org Lett 15:4430–4433
Blankenship RE (2014) Molecular mechanisms of photosynthesis, 2nd edn. Wiley Blackwell, West Sussex
Chang CK, Sotiriou C (1987) A novel method of functionalizing the ethyl chain of octaethylporphyrin. J Org Chem 52:926–929
Chen M (2014) Chlorophyll modifications and their spectral extension in oxygenic photosynthesis. Annu Rev Biochem 83:317–340
Clezy PS, Fookes CJR (1978) Chemistry of pyrrolic compounds. XLI. The synthesis of the phaeophorbides c 1 and c 2 methyl esters. Aust J Chem 31:2491–2504
Couch PW, Games DE, Jackson AH (1976) Synthetic and biosynthetic studies of porphyrins. Part 1. Synthesis of the ‘S-411’ porphyrin obtained from meconium. J Chem Soc Perkin Trans 1:2492–2501
Croce R, van Amerongen H (2014) Natural strategies for photosynthetic light harvesting. Nat Chem Biol 10:492–501
Glazer AN, Wedemayer GJ (1995) Cryptomonad biliproteins – an evolutionary perspective. Photosynth Res 46:93–105
Green BR (2011) After the primary endosymbiosis: an update on the chromalveolate hypothesis and the origins of algae with Chl c. Photosynth Res 107:103–115
Helfrich M, Bommer B, Oster U, Klement H, Mayer K, Larkum AWD, Rüdiger W (2003) Chlorophylls of the c family: absolute configuration and inhibition of NADPH:protochlorophyllide oxidoreductase. Biochim Biophys Acta 1605:97–103
Ito H, Tanaka A (2014) Evolution of a new chlorophyll metabolic pathway driven by the dynamic changes in enzyme promiscuous activity. Plant Cell Physiol 55:593–603
Kunieda M, Tamiaki H (2009) Self-aggregation of synthetic bacteriochlorophyll-d analogues possessing a B-ring reduced chlorin π-system. J Org Chem 74:8437–8440
Kunieda M, Mizoguchi T, Tamiaki H (2004) Syntheses and optical properties of stable 8-alkylidene-bacteriochlorins mimicking the molecular structures of natural bacteriochlorophylls-b and g. Tetrahedron 60:11349–11357
Kunieda M, Mikata Y, Tamiaki H (2007) Regioselective transformation of octaethylporphyrin into a phytoporphyrin analogue. J Org Chem 72:7398–7401
Liu C, Dobhal MP, Ethirajan M, Missert JR, Pandey RK, Balasubramanian S, Sukumaran DK, Zhang M, Kadish KM, Ohkubo K, Fukuzumi S (2008) Highly selective synthesis of the ring-B reduced chlorins by ferric chloride-mediated oxidation of bacteriochlorins: effects of the fused imide vs isocyclic ring on photophysical and electrochemical properties. J Am Chem Soc 130:14311–14323
Mizoguchi T, Nagai C, Kunieda M, Kimura Y, Okamura A, Tamiaki H (2009) Stereochemical determination of the unique acrylate moiety at the 17-position in chlorophylls-c from a diatom Chaetoceros calcitrans and its effect upon electronic absorption properties. Org Biomol Chem 7:2120–2126
Mizoguchi T, Kimura Y, Tamiaki H (2010) Exclusive observation of the (132 R)-enantiomer of chlorophyll-c from a diatom Chaetoceros calcitrans. Photochem Photobiol 86:311–315
Mizoguchi T, Kimura Y, Yoshitomi T, Tamiaki H (2011) The stereochemistry of chlorophyll-c 3 from the haptophyte Emiliania huxleyi: the (132 R)-enantiomers of chlorophylls-c are exclusively selected as the photosynthetically active pigments in chromophyte algae. Biochim Biophys Acta 1807:1467–1473
Montforts FP, Glasenapp-Breiling M (2002) Naturally occurring cyclic tetrapyrroles. Fortschr Chem Org Naturst 84:1–51
Niedzwiedzki DM, Blankenship RE (2010) Singlet and triplet excited state properties of natural chlorophylls and bacteriochlorophylls. Photosynth Res 106:227–238
Pandey RK, Isaac M, MacDonald I, Medforth CJ, Senge MO, Dougherty TJ, Smith KM (1997) Pinacol–pinacolone rearrangements in vic-dihydroxychlorins and bacteriochlorins: effect of substituents at the peripheral positions. J Org Chem 62:1463–1472
Sanchez-Puerta MV, Bachvaroff TR, Delwiche CF (2007) Sorting wheat from chaff in multi-gene analyses of chlorophyll c-containing plastids. Mol Phylogenet Evol 44:885–897
Senge MO, Ryan AA, Letchford KA, MacGowan SA, Mielke T (2014) Chlorophylls, symmetry, chirality, and photosynthesis. Symmetry 6:781–843
Smith KM, Goff DA, Simpson DJ (1985) Meso substitution of chlorophyll derivatives: direct route for transformation of bacteriopheophorbides d into bacteriopheophorbides c. J Am Chem Soc 107:4946–4954
Tamiaki H, Kunieda M (2011) Photochemistry of chlorophylls and their synthetic analogs. Handb Porphyr Sci 11:233–290
Tamiaki H, Tanaka T (2015) Synthesis and physical properties of carbonylated chlorophyll derivatives. Tetrahedron 71:1915–1923
Tamiaki H, Miyatake T, Tanikaga R (1997a) Self-aggregation of synthetic zinc chlorins possessing “inverse” keto and hydroxyl groups. Tetrahedron Lett 38:267–270
Tamiaki H, Tomida T, Miyatake T (1997b) Synthesis of methyl bacteriopheophorbide-d with 8-propyl group. Bioorg Med Chem Lett 7:1415–1418
Tamiaki H, Yagai S, Miyatake T (1998a) Synthetic zinc tetrapyrroles complexing with pyridine as a single axial ligand. Bioorg Med Chem 6:2171–2178
Tamiaki H, Takeuchi S, Tsudzuki S, Miyatake T, Tanikaga R (1998b) Self-aggregation of synthetic zinc chlorins with a chiral 1-hydroxyethyl group as a model for in vivo epimeric bacteriochlorophyll-c and d aggregates. Tetrahedron 54:6699–6718
Tamiaki H, Watanabe T, Miyatake T (1999) Facile synthesis of 131-oxo-porphyrins possessing reactive 3-vinyl or 3-formyl group, protochlorophyll-a/d derivatives by 17,18-dehydrogenation of chlorins. J Porphyr Phthalocyanines 3:45–52
Tamiaki H, Omoda M, Saga Y, Morishita H (2003) Synthesis of homologously pure bacteriochlorophyll-e and f analogues from BChls-c/d via transformation of the 7-methyl to formyl group and self-aggregation of synthetic zinc methyl bacteriopheophorbides-c/d/e/f in non-polar organic solvent. Tetrahedron 59:4337–4350
Tamiaki H, Shibata R, Mizoguchi T (2007) The 17-propionate function of (bacterio)chlorophylls: biological implication of their long esterifying chains in photosynthetic systems. Photochem Photobiol 83:152–162
Wedemayer GJ, Wemmer DE, Glazer AN (1991) Phycobilins of cryptophycean algae. J Biol Chem 266:4731–4741
Xu M, Tamiaki H (2014) Transformation of natural chlorophyll-a into chlorophyll-c analogs possessing the 17-acrylate residue. Chem Lett 43:1864–1866
Zapata M, Rodríguez F, Fraga S, Barra L, Ruggiero MV (2011) Chlorophyll c pigment patterns in 18 species (51 strains) of the genus pseudo-nitzschia (bacillariophyceae). J Phycol 47:1274–1280
Zheng G, Dougherty TJ, Pandey RK (1999) A simple and short synthesis of divinyl chlorophyll derivatives. J Org Chem 64:3751–3754
Acknowledgments
This work was partially supported by Grants-in-Aid for Scientific Research (A) (No. 22245030) as well as on Innovative Areas “Artificial Photosynthesis (AnApple)” (No. 24107002) from the Japan Society for the Promotion of Science (JSPS).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Xu, M., Kinoshita, Y., Matsubara, S. et al. Synthesis of chlorophyll-c derivatives by modifying natural chlorophyll-a . Photosynth Res 127, 335–345 (2016). https://doi.org/10.1007/s11120-015-0190-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-015-0190-1