Abstract
Aims
This study aims to identify some of the key soil-related factors that underpin the distribution of plant communities and provide a more comprehensive understanding of an old, climatically buffered infertile landscape including kwongkan habitats. We investigate the pedological, topographical and hydrological correlates of abundance and floristic community distribution within the Alison Baird Reserve, a floristically diverse, old and climatically weathered landscape in south-west Western Australia.
Methods
Fifty-four soil samples were collected across 0.165 km2 and analysed for a range of physical and chemical properties. Samples were taken from the topsoil and subsurface, and a combination of statistical and geostatistical methods were used to examine soil–plant community distribution relationships.
Results
Elevation, sub-surface moisture, electrical conductivity and clay content were the most important factors in determining vegetation composition and distribution; with sub-surface properties varying more between floristic communities and correlating more strongly than topsoil factors. Associations between soil nutrients (Phosphorus, Nitrogen, Carbon) and community distributions were not as significant as expected, likely due to the depleted nature of the soil system.
Conclusion
The significance of soil parameters including particle size, moisture, elevation, water repellence and depth to water table suggests that water availability and retention within the soil is a key correlate of the distribution of floristic communities at the site and may act as a pathway to explain large scale variation in plant communities across the south-west of Western Australia. This understanding will help conservation and management of areas with such high biodiversity including relocation of endangered flora.
Similar content being viewed by others
Introduction
Soils provide the foundation upon which most terrestrial life depends. The interactions between soil, topography, hydrology, and climate provide the foundation of flora and fauna diversity (Lambers et al. 2009). The specific factors that determine the distribution and abundance of vegetation are variable and largely site specific (Didham et al. 2007; Shackelford et al. 2014). The success of conservation and management approaches, especially in an urban context, rely on a thorough understanding of the interrelationships between these biophysical characteristics (Coates et al. 2014; Monks et al. 2019).
South-western Australia contains one of the global biodiversity hotspots (Lambers et al. 2013; Marchese 2015; Myers 1990), the Southwest Australian Floristic Region (SWAFR; Hopper and Gioia 2004). The Swan Coastal Plain is part of the SWAFR, a roughly 30 km wide strip of land stretching several hundred kilometres north and south of Perth, Western Australia, with the Banksia woodlands forming a characteristic ecological community (Ritchie et al. 2021). Perth and the Swan Coastal Plain have a Mediterranean climate with cool wet winters and hot, dry summers (Köppen climate classification Csa,). Geologically, the region is characterised by a series of dune systems with wetlands dotting the landscape as surficial expressions of the underlying aquifer (Lane and Evans 2019).
Alison Baird Reserve, part of the Greater Brixton Street Wetlands, is the most floristically biodiverse area on the Swan Coastal Plain, with an extensive number of endemic species represented (Tauss et al. 2019; Tauss and Weston 2010). The site comprises a combination of wetland and sand dune landscapes and is seasonally affected by water availability; waterlogged in winter and dry during the summer, with occasionally perched water tables (Speck and Baird 1984). More than 400 native species have been identified at the site with more than 90% of the flora endemic to Western Australia (Speck and Baird 1984; Tauss et al. 2019).
The location of the site within the greater Perth metropolitan region implies exposure to a range of associated urban pressures. Stormwater pollution, exotic and invasive flora and fauna species, as well as introduced pathogens all compound to challenge the existing ecosystem (Alston and Richardson 2006; Gill and Williams 1996; Struglia and Winter 2002; Tauss and Weston 2010). Rapid urban sprawl and associated land clearing threatens the survivability of endemic flora and fauna and highlights the importance of protecting species-richness (Ritchie et al. 2021).
The general nature of soils at the Alison Baird Reserve has been described by Leopold and Zhong (2019); soils are characteristic of old climatically-buffered infertile landscapes (OCBIL; Hopper 2009; Hopper et al. 2016). Due to their uncommon occurrence, the soils of these landscapes are not well understood as the majority of research to date has focused on young often-disturbed, fertile landscapes (YODFEL) in the northern hemisphere or in habitats surroundings OCBILs (Hopper 2009; Hopper et al. 2016). Conservation and preservation approaches often used with YODFELs have been applied to OCBILs with limited success, given the vast differences between the soil and vegetation characteristics of these two landscapes (Gosper et al. 2021a; Standish et al. 2007; Standish and Hobbs 2010; Silveira et al. 2021). In recent years OCBIL theory has been further developed through examination of multiple sites across the southern hemisphere (Gosper et al. 2021b; Silveira et al. 2021). Many OCBILs are small and fragmented, with Hopper (2009) identifying that traditional conservation methods used to preserve YODFELs are often detrimental to OCBILs. Soils in OCBIL systems are extremely nutrient impoverished due to long-term weathering and leaching (Gilkes et al. 2006; Hopper and Gioia 2004). In particular, phosphorus availability has been suggested to drive the distribution of vegetation within these systems (Hayes et al. 2021; Laliberté et al. 2015; Nichols and Beardsell 1981). Although, when compared with YODFEL soils, the concentration of nutrients is generally lower, nutrient gradients, even over a small range, are hypothesised to be of importance to microbial (Gleeson et al. 2016; O’Brien et al. 2019) and vegetation distribution (Gao et al. 2020; Lambers et al. 2013, 2011). Importantly, conservation planning is largely underpinned by the availability and abundance of spatial information (Gosper et al. 2021b; Rouget et al. 2003; Pressey et al. 2007). To expand our understanding of OCBILs, a multitude of small-scale studies are required due to the uniqueness of these sites. The more we understand how these individual areas function, the higher the chances for a successful implementation of conservation strategies.
Where OCBILs are present in south-western Australia, kwongkan (local Noongar Indigenous word for sand) vegetation is a dominant vegetation community and has been poorly studied (Hopper and Gioia 2004). Kwongkan, or sandplain shrubland, is distributed across the south-west of Western Australia (Mucina et al. 2014). It has been defined as a “biological treasure house” due to the number of endemic species present, and the capacity of the species to persist in depleted soils (Hopper and Gioia 2004). Retaining existing kwongkan vegetation and understanding the factors associated with their distribution is of cultural, economic and biological significance (Hopper 2014). A lack of comprehensive analyses of kwongkan vegetation remains a barrier to developing a more complete understanding of the system (Tsakalos et al. 2018). Much of the current research on soil-vegetation dynamics in kwongkan habitats has focussed on locations north or south of the Swan Coastal Plain (Lambers et al. 2011; Tsakalos et al. 2019, 2018; Yates et al. 2010a, b), with fewer studies on the plain itself (Laliberté et al. 2013). Alison Baird Reserve comprises of a mixture of kwongkan, banksia woodland and low wetland vegetation habitats.
The dynamics between species degradation, expansion and diversity is largely system and species dependent (Didham et al. 2007; Shackelford et al. 2014). In order to achieve a more thorough understanding of kwongkan vegetation dynamics, soil–plant relationships need to be examined. Previous studies have found that phosphorus is a key limiting nutrient for plants within kwongkan habitats (Laliberté et al. 2015; Lambers et al. 2013). Others have hypothesised that water availability is more important (Prober and Wiehl 2012; Tsakalos et al. 2019, 2018).
Studies by Speck and Baird (1984) and Wege (1992) hypothesised that depth to clay layer and salinity were the most significant drivers of vegetation distribution specific to the Alison Baird Reserve. Clay is influential due to its greater ability to retain nutrients and water in comparison to coarser particles with smaller specific surface areas (White 2005). Others have suggested that topography plays a role in vegetation distribution (Moeslund et al. 2013). Variation in the drivers of vegetation distribution is commonplace, highlighting the importance of examining the system as a whole, particularly as previous attempts to relocate rare flora within the Brixton Street Wetlands area were unsuccessful due to lack of understanding of specific habitat requirements (Wyrwoll et al. 2014).
Understanding the factors associated with plant diversity and distribution within highly diverse plant communities is critical for estimating the impact of climatic and anthropogenic pressures (Franks et al. 2009; Gosper et al. 2021b; Laliberté et al. 2013), in order to identify appropriate management solutions. This study aims to identify some of the key soil-related factors that underpin the distribution of plant communities in part of Alison Baird Reserve. It will provide a baseline assessment for use in monitoring change, and to aid conservation efforts. It aims to provide a more comprehensive understanding of local OCBIL habitats and associated soil–plant relationships. The following hypotheses allow for the examination of associations between soil properties, topography and plant community distribution:
-
i)
Differences in quantity and quality of soil nutrients and water availability are associated with the distribution of floristic communities and key species;
-
ii)
Soil texture, respectively clay, is positively correlated with the location of certain floristic communities;
-
iii)
Topography and associated water-related soil properties influence floral species diversity.
Materials and methods
Study site
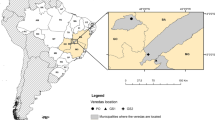
The study site was the northern part of Alison Baird Reserve (32.021685° S, 115.980202° E), located to the east of the Perth Central Business District in Western Australia (Fig. 1). Alison Baird Reserve is a part of the Greater Brixton Street Wetlands and has been owned and managed by the University of Western Australia (UWA) since 1949 for teaching and research purposes.
Top: location of the study site in its greater Australian and Perth context. Bottom: soil sampling locations, electrical resistivity tomography (ERT) and ground-penetrating radar (GPR) survey lines and locations of floristic communities (for details see Table 1). Coordinate system: GDA 1994 MGA Zone 50
The hydro-pedology of Alison Baird Reserve is variable with low-lying areas to the north-east and south-west experiencing seasonal waterlogging, forming wetlands during winter. A large dune from the Bassendean Dune System, estimated to be up to 2000 ka old (Turner et al. 2018), runs through the middle of the reserve with heights of up to 5 m (Leopold and Zhong 2019). The sand system is underlain by clay rich sediments of the stratigraphic Leederville formation (Gao et al. 2020; Lambers 2019).
Geophysical methods
Geophysical methods offer fast and reliable 2D-images of the subsurface, which can be used to strategically locate the best places for subsequent drillings or excavations. They further extend the spatially limited information of soil pits to a larger area to better understand the sedimentological context of a site. These techniques are non- or minimally-invasive, which makes them ideal survey tools for ecologically sensitive areas. Ground-penetrating radar (GPR) and electric-resistivity tomography (ERT) were used to obtain deep subsurface information to set the stratigraphic context for the soils and related plant communities (transect pathway shown in Fig. 1). A mala X3m GPR system coupled with a shielded 100 MHz antenna was used to obtain radar images across the dune area. Raw data were filtered and subsequently visualised using ReflexW software following the protocols of Neal (2004). Additionally, a Lippmann 4point light multi-electrode ERT system was used to identify the location within the subsurface where moisture and high clay content prevents GPR signal return due to attenuation. Apparent electric resistivity values from dipole–dipole arrays with 100 electrodes were inverted to specific electric resistivity values using RES2DINV software from Geotomo (Loke 2019) following the protocol of Leopold et al. (2021). GPR and ERT images were stratigraphically interpreted using borehole data provided in Gao et al. (2020) as ground-truthing information.
Floristic community identification
Floristic communities used in this study were derived from Tauss et al. (2019), who carried out a floristic survey of the entire Greater Brixton Street Wetlands (Table 1). The spatial distribution of these communities is thought to correlate with soil properties and topographic position.
Interest species
Additional to floristic communities, we explored the soil properties associated with individual ‘species of interest’ within the reserve to gain an understanding of their habitat. Due to their scarcity and importance as threatened flora, the following species were selected to test soil properties in their vicinity.
-
Andersonia gracilis DC:
This small shrub is listed as a Threatened Flora Species (Endangered) and there is very little information on its preferred habitat.
-
Beaufortia squarrosa Schauer:
This species is of interest at the site given its idiosyncratic distribution in Alison Baird Reserve, its rare occurrence in the Perth Region of the Swan Coastal Plain, and the lack of habitat data.
-
Grevillea thelemanniana Endl.:
This medium-height shrub is a Threatened Flora Species (Critically Endangered). In the wild, this shrub is confined to the Greater Brixton Street Wetlands (Tauss et al. 2019; WA Herbarium 1998-) and the adjacent suburbs of Kenwick and Wattle Grove, both proposed for industrial development (Tauss and Weston 2010). It is of particular interest because of its narrow distribution, the high threats posed to the persistence and genetic integrity of wild specimens and the very limited understanding of its natural habitat (Gao et al. 2020).
Soil sampling and analyses
Soil sampling locations were initially identified using a grid-based mapping approach to ensure multiple sampling of the diverse floristic communities. However, some sampling positions had to be adjusted according to ease of access while on site. On 23 October 2018, 54 topsoil (0–10 cm) and 531 sub-surface (50–60 cm) soil samples were collected using a stainless-steel trowel and hand auger. Additional soil samples were collected at locations where species of interest were identified (Fig. 1).
Samples were placed in plastic bags, labelled and subsequently air dried at 40˚C for one week. Samples were then sieved to fine earth fraction (< 2 mm) and analysed for relevant soil properties (Table 2). Blanks and replicates were included for Carbon, Nitrogen and Phosphorus (C, N and P) concentrations and particle size analysis. Additional samples were taken at the same location using a metal ring of known volume (293 cm3) for subsequent bulk density calculation after drying for 48 h at 105˚C.
Statistical analyses
A mixture of statistical tests (p-values), ordination, classification trees, graphs, and maps were used to interpret soil-flora associations.
All soil data were examined for normality and equal group variance in R using a combination of Shapiro–Wilk, Bartlett and Fligner tests (Reimann et al. 2008). Where possible, transformations were conducted on soil variables to create a normal distribution and/or equal variance (Reimann et al. 2008). Transformations included the Box-Cox, log, square root and power transformations. ANOVA and the non-parametric Kruskal–Wallis test were used for soil, topography and vegetation association analysis as vegetation complex was described as a categorical variable (Reimann et al. 2008). Levene Test and visual histogram analysis were used to identify homogeneity of variance and determined whether ANOVA or Kruskal–Wallis was performed.
Statistical analysis was undertaken in R (R Core Team 2022) using the base, vegan (Oksanen 2015) and car packages (Fox and Weisberg 2015). A Spearman’s Correlation Matrix was constructed to inform linear statistical relationships and guide further analysis.
Ordination plots were created for: sub-surface soil properties, topsoil properties and significant only variables (Oksanen 2015). Soil and topography variables were fitted to the ordinations to visually investigate gradients using the envfit function in the vegan package in R (Oksanen 2015).
Classification tree analysis was undertaken to further assist in evaluation of key environmental variables and their correlation with the floristic distribution at the site (De'ath and Fabricius 2000). The package rpart (Therneau and Atkinson 2019) was used in R (R Core Team 2022), based on the original work by Breiman et al. (1984).
We further investigated if depth to water table was related to vegetation distribution. Water table depths were extracted from the Department of Water online database (Western Australia DoW 2015) for each soil sample location. ANOVA tests were used for soil properties and depth to water table data. Topographical variables measured were elevation above sea level (m), slope (degree) and aspect (degree). Topographical variables were derived from a Digital Elevation model (Moeslund et al. 2013) calculated from stereo digital aerial photography as part of the CSIRO Urban Monitor program (Landgate, Western Australia; Caccetta et al. 2012), with an approximate resolution of 20 cm.
Results
Geophysical results
The GPR line shows horizontal, high-amplitude reflections at 1 to 5 m depth along the survey line, which are interpreted as reflections from the dune base (Fig. 2a). Signal attenuation below this reflection is indicative of high clay and moisture content in accordance with nearby seasonal wetland areas. The clay surface, which most likely belongs to the geological stratigraphic unit of the Leederville formation (Gao et al. 2020), rises roughly 1 m in elevation from west to east. This 1 m step functions as an accumulation trap for aeolian sands and the subsequent formation of the longitudinal dune in N-S direction. The GPR displayed an additional 1 m of sediment at the area east of the sand dune identified as a sand sheet. The accumulation resulted in a total vertical difference in elevation of more than 2 m between the west and east side of the dune which displayed a first major stratigraphic and topographic difference between the two areas adjacent to the dune.
a A 100 MHz radar image and interpretation acquired along the sand track across the dune system. Note the strong reflection between 70 and 100 ns at the base of the sand dune, which represents the local clay layer. b Electrical resistivity tomography (ERT) line east of the sand dune which documents a 1–2 m thick sand sheet over the clay-rich Leederville formation. Low resistivity values may indicate slightly saline water perched in this area. c ERT line west of the dune where slightly saline water reaches the surface perched by the clay-rich Leederville formation
The GPR results were validated by the two ERT lines that ran parallel to the dune (Fig. 2b, c). Both lines show relatively low electric resistivity values, with a maximum value of 50 Ωm. ERT line 1 displayed a 2-m thick zone with slightly higher resistivity near the surface which corresponds to the identified sandy layer in the GPR survey. This sand sheet was then underlain by a zone of low resistivity values (~ 5 Ωm) at about 4 m depth. This was followed by sediments with slightly higher resistivity values up to 20 Ωm. ERT line 2 displayed a similar distribution but lacked the 2 m thick high-resistivity zone on the surface.
Statistical tests and ordination
Floristic communities were most strongly influenced by elevation, sub-surface moisture, sub-surface clay content and EC (Table 3). Moisture content was identified as the most significant topsoil property correlated with the distribution of floristic communities.
Topsoil properties were rather homogenous across communities as indicated by overlapping of ellipses in the ordination space. However, Banksia spp. -Jacksonia floribunda (B Spp. –Jff) was significantly differentiated from other vegetation communities. Within the sub-surface, B spp.-Jff only slightly overlapped with other plant communities. Melaleuca brevifolia–Samolus junceus (Mb-Sj) and Melaleuca acutifolia–Verticordia plumosa (Ma-Vp) was the most significantly different within the ordination space; however, only one sample was recorded for each of these groups.
Moisture, followed by elevation and clay content explained most of the variation within the ordination space (Fig. 3). Viminaria juncea–Babingtonia urbana (Vj-Bu), Cytogonidium leptocarpoides–Schoenus efoliatus (Cl-Se), Banksia telmatiaea–Schoenus (Bt-Sr) and Melaleuca rhaphiophylla–Schoenus subfascicularis (Mr-Ss) displayed consistent significant soil–plant relationships, exhibited by their overlapping in the ordination plots.
Ordination plots. a Topsoil properties only. b Sub-surface soil properties. NMDS1 and NMDS2 are arbitrary axes within ordination space. The labelled ellipses represent the 95% confidence interval for each floristic community within the ordination space. Ellipses that overlap are considered similar based on the variables used as input. c Ordination plot with only significant variables for both the topsoil and sub-surface soil. Topographical variables are also included. The NMDS1 axis represented a composite elevation, depth to water table, sub-surface clay and electric conductivity (EC) gradient. The NMDS2 mostly represented a composite P—moisture gradient, resulting in the separation of the B spp.-Jf, Mb-Sj and Ma-Vp from the other floristic communities. B spp. – Jf was separated from the other communities by a higher elevation, lower moisture content and lower clay composition
Elevation accounted for 15% of the plant community distribution (Fig. 4a). There was a clear divide in elevation, with the B spp-Jf group, which is mainly located on the sand dune, being above 11.25 m elevation. All other plant communities had elevations below 11.25 m with elevation having no further impact on floristic community distribution, but topsoil pH and moisture did. Topsoil field moisture content, which was correlated with soil texture, again split the B spp-Jf group (42.6%) from the rest of the plant communities at θ = 6.5 cm3cm−3 volumetric water content. Topsoil pH and slope were next most important factors describing plant distribution (Fig. 4b).
a Classification Tree with all variables. Variable importance (rounded): Elevation 20%, topsoil moisture 14%, sub-surface moisture 14%, sub-surface clay, sub-surface EC and sub-surface sand all 10%, topsoil pH (CaCl2) 8%. b Classification tree with variables excluding elevation. When elevation was excluded, variable importance was: sub-surface moisture 15%, topsoil moisture 12%, sub-surface EC 11%, topsoil pH (CaCl2) 9%, sub-surface clay, sub-surface sand and topsoil C:N ratio all 7%
Sand and clay content in the topsoil and sub-surface soil were negatively correlated with C, N and P concentrations (Fig. 5). Higher elevations corresponded with lower sub-surface moisture levels, likely due to the depth to water table and sandy nature of the soils on the sand dune. N and C concentrations were closely correlated with texture, with a stronger correlation for the topsoil than for the sub-surface soil (Fig. 5).
Spearman's Correlation Matrix for all variables. Red circles indicate negative correlation, blue positive. The larger the circle, the more strongly correlated two variables are. Variables are listed in alphabetical order, with the prefix TS denoting topsoil and SS denoting sub-surface soil. Rep. = Repellence rating
In general, there were clear differences between most topsoil and sub-surface soil properties across all floristic communities. Topsoil total C was higher than sub-surface total C, as expected (Fig. 6). P had higher concentration in the subsurface, except for Vj-Bu, Mr-Ss, the latter representing a rather moist area, which could be responsible for the wide range of available P concentrations (2 -11.2 sub-surface mg P kg−1) in that area. The highest topsoil P values were found within the Mr-Ss community with 38.4 mg P kg−1. There was no correlation between the topsoil P concentration and the floristic community distribution. The Bpp.-Jf community exhibited a higher sub-surface soil P concentrations than the topsoil P concentration. The highest sub-surface soil P concentration was found within the Ma-Vp community with 22.8 mg P kg−1 soil. However, the sample had a relatively low P concentration in the topsoil at 2.8 mg P kg−1.
Topsoil pH (H2O) values were generally acidic between pH 5 and 6 with the exception of Mo-Lc, representing a wet low-laying area with values between pH 6 and neutral (Fig. 7a). Sub-surface pH (H2O) values were generally less acidic with several communities experiencing neutral or slightly alkaline pH (H2O) conditions. Topsoil EC was mostly low, but subsurface EC values generally differed more between plant communities than they differed within each communitys. Saline sub-surface hotspots were identified within Melaleuca osullivanii–Leptocarpus coangustatus (Mo-Lc), Mr-Ss and Mb-Sj, which were on opposite sides of the sand dune in lower-lying areas (Fig. 7b).
Boxplots across each floristic community. a Topsoil and sub-surface pH. b Topsoil and sub-surface electric conductivity (EC) The median is indicated by the black line within the interquartile range. Black dots represent values greater than 1.5 times and less than 3 times the interquartile range beyond the boxes
Interest species
C, N and silt content of the soils at the Beaufortia squarrosa site were different when compared with the surrounding site (Figs. 8, 9). The topsoil clay to sub-surface clay content ratio was similar for each interest species (Fig. 9).
Concentrations as boxplots for each interest species. a Topsoil and subsurface C. b Topsoil and sub-surface plant-available P. c Topsoil and sub-surface N. ‘Soil’ group refers to the samples from the rest of the site. AND = Anderso-nia gracilis, BFT = Beaufor-tia squarrosa, GREV = Grevillea thelemanniana. The median is indicated by the black line within the interquartile range. Black dots represent values greater than 1.5 times and less than 3 times the interquartile range beyond the boxes
Topsoil and subsurface boxplots for each interest species. a Clay. b Silt. c Depth to water table. d Repellence rating. ‘Soil’ group refers to the rest of the site. AND = Andersonia gracilis, BFT = Beaufortia squarrosa, GREV = Grevil-lea thelemanniana. Soil refers to the remaining soils across the whole site that are not associated with an interest species. The median is indicated by the black line within the interquartile range. Black dots represent values greater than 1.5 times and less than 3 times the interquartile range beyond the boxes
The soils associated with Andersonia gracilis DC and Grevillea thelemanniana all exhibited low water repellence. In comparison, the repellence rating of soils associated with Beaufortia squarrosa was significantly greater, with an average rating of MED 4, representing very severe water repellence conditions (Fig. 9d). The most significant difference in soil properties associated with interest species was the C and N concentration. Soils associated with Beaufortia squarrosa displayed significantly higher C and N concentration than any of the other species and across the site in general (Fig. 8a, c).
Sub-surface P concentration varied among the soils associated with interest species and across the rest of the site (Fig. 8b). The topsoil:sub-surface soil P ratio also differed among the species. Interestingly, soils in the habitats of Andersonia gracilis and Grevillea thelemanniana had higher sub-surface P and lower topsoil P concentrations.
Discussion
Geophysical analysis
The area east of the dune exposed an up to 2 m thick sand sheet above clays from the Leederville formation. This sand sheet was missing west of the dune where only a thin layer of colluvial sandy loam overlays the sandy clays of the Leederville formation. The generalised soil texture distribution from west to east can be described as sandy-clayey topsoils over clays west of the dune, deep sandy soils over clays at the dune, and sandy soils over clays east of the dune. However, the eastern part was further differentiated by some low-lying wet areas connected to Crystal Brook in this area. This stratigraphic model was corroborated by results from the resistivity tomograms (Fig. 2b, c) where the sandy-clay rich sediments from the Leederville formation perched slightly saline water, as confirmed by higher EC subsurface values. The zone with higher resistivity exhibited in the ERT1 line represented the sand sheet also identified east of the dune by the GPR survey. This stratigraphic interpretation was further validated by various soil cores from the area described by Gao et al. (2020) and further aligns with the geological descriptions presented by Lane and Evans (2019).
Soil property distribution across site
The multivariate analysis of soil property associations with plant community distribution provided important insights into the underlying soil characteristics of current vegetation patterns. Most importantly, variation in certain soil properties showed greater variability across the site than within a specific community, suggesting that each community had a physical–chemical soil preference underlying the importance of small-scale studies in OCBIL landscapes (Hopper 2009). In reference to hypothesis (i), P did not exhibit the strongest association with floristic community distribution. P concentrations did show variation in the sub-surface among communities. However, the variation of P concentration within each community was just as large as that across the entire site, especially within the topsoil of Mr-Ss, and in the sub-surface of Vj-Bu and B spp.-Jf (Fig. 7). Sub-surface P concentration had no statistical impact on floristic distribution. Results of the cluster analysis using classification trees (Fig. 4) corroborates the latter statement as P concentration did not show up as a factor to describe the distribution of plant communities. There was also large variation within the B spp.-Jff floristic community that is located on top of the sand dune which was unexpected given the higher sand:clay ratio in the remainder of the site. It is possible that the limited P availability prevented specific species from establishing within the reserve (Laliberté et al. 2015; Lambers et al. 2018; Nichols and Beardsell 1981); however, its role in shaping the vegetation community distribution in the reserve did not appear to be the most significant within this fine scale study. Other studies point to the fact that nutrient gradients inform vegetation distribution within nutrient-poor Mediterranean landscapes (Laliberté et al. 2014; Richards et al. 1997), however, in Alison Baird Reserve soil-P is low everywhere and thus a less important factor compared to water. Although soil properties in this study were primarily associated with floristic community groups, and not the presence of a range of individual species, the result provide insight into the extent to which P is a limiting factor. However, it must be noted that we analysed only a specific part of the P-pool, and there is likely more P available to specialised plants such as proteaceous, which is not represented by this method. Different methods targeting different forms of soil P, or the analysis of organic P might deliver slightly different correlations.
Overall C, N and P concentrations were low in comparison with global averages (Hopper 2009; Laliberté et al. 2013), supporting the OCBIL hypothesis that taxa within these landscapes have evolved to survive in nutrient-poor soils (Silveira et al. 2021). The low P, C and N concentrations, high water repellence and low water-holding capacity of the B spp.-Jf community was consistent with the Bassendean sand dune systems (Bouwer 1978; Laliberté et al. 2013; Scott et al. 2012). The leaf litter originating from Banksia trees (the dominant species within the community) decomposes to form a hydrophobic humic layer (Tsakalos et al. 2018). The high sand content of the B spp.-Jf community (sub-surface mean of 98.8% and topsoil mean of 97.9%), in addition to litter produced by the banksias, likely contributed to high levels of soil water repellence (Franco et al. 2000; Ritchie et al. 2021). Here, greater C content and water repellence were strongly associated (Fig. 5), consistent with the findings of Harper and Gilkes (1994), and Cesarano et al. (2016) which highlight that leaf litter and organic matter in general increases water repellence in topsoils in south-western Australia.
Relationships between plant communities, sub-surface soil and topsoil properties with topography and hydrology
Statistical tests, classification trees and distribution maps indicate that variations among floristic communities were influenced by both topsoil and sub-surface soil parameters and their interrelationships. This highlights the importance of not limiting soil studies to topsoil when assessing ecosystems. Current studies focussing on kwongkan distributions (Gibson et al. 2004; Tsakalos et al. 2018) might further benefit from including more subsurface soil parameters. A more complete understanding of the plant community distribution in these nutrient-impoverished systems might be achieved by integrating sub-surface and topsoil samples, together with external environmental variables such as topography or depth to water table.
Soil particle size played a highly associated role with floristic community distribution within the reserve. Clay content was identified as having a high association with plant community distribution, supporting hypothesis ii. In addition, particle size influences many other important soil parameters such as porosity, water retention potential, bulk density, cation/anion exchange capacity, which all influence plant growth (McKenzie et al. 2004) . Fordyce et al. (2007) documented strong relationships between soil texture and plant community distribution in a study of jarrah (Eucalyptus marginata) forests in Western Australia. However, they also indicate that texture may be more influential on local hydrology, which then influences vegetation distribution. This is contrary to the direct link between soil texture and plant distribution, which has been identified in different YODFEL environments (Wang et al. 2015).
Resistivity tomograms and the radargram show that topography influences the preconditions by which plants access water and nutrients. The depth to the shallow, sometimes perched, water table is related to the clay layer that is found at variable depth across the reserve (Fig. 2). Topography expressed in elevation differences varied significantly among communities at the studied site and was strongly related with clay, moisture content and sub-surface N concentration. Elevation also directly related to depth to water table (Fig. 5), further compounding to show that water availability and retention were likely the most influential variables in terms of plant distribution at the site. Additionally, slope was strongly related to moisture content, indicating an indirect effect on flora through hydrological dynamics, similar to the observations by Fordyce et al. (2007). This supports the significance of elevation in all analyses conducted in this study.
In reference to hypothesis ii, the significance of the sub-surface clay content and location, together with the lower significant association with clay content in the topsoil, suggests that depth to clay influenced the distribution of vegetation within the site. Depth to ground water and clay content play important roles in limiting or allowing plants to access water (Tsakalos et al. 2019). In our case, clay behaved as a perching layer, limiting the movement of water through the soil column (Tsakalos et al. 2018; White 2005). Due to this, depth to clay layer relative to groundwater can greatly influence a plant’s ability to access water, especially during drier months. Our statistical tests show that sub-surface clay content was significantly different among communities, as well as being strongly associated with water availability, as illustrated by all ordination plots, causing the separation of the B spp-Jf, Mb-Sj and Ma-Vp from the other communities (Fig. 3). Within the classification trees, sub-surface clay content was the most significant variable when elevation was removed from the model (Fig. 4).
The absence of a relationship between plant-available soil P concentration and elevation was unexpected when these results were compared with other studies (Lambers et al. 2018; Vincent et al. 2014). One plausible explanation is the extremely P-impoverished status of the Bassendean dune system. A positive relationship was identified between plant-available P concentration and the clay content in the topsoil which is consistent with the current understanding of Bassendean sand dune systems (Wyrwoll et al. 2014). The topsoil likely has a greater overall nutrient content due to the accumulation of organic matter (White 2005), supported by the strong correlation with topsoil total C concentration (Fig. 6). Although a statistical relationship was identified, higher clay contents do not invariably infer higher relative plant-available P concentrations at the sample sites.
The significance of elevation (hypothesis iii), water repellence, depth to water table and moisture content (hypothesis i) combined with the significance of depth to clay layer (hypothesis ii) suggests that water availability and water-holding capacity of the soil is important for the distribution of vegetation across the site. Elevation and slope impact vegetation at the site through the influence on moisture content and assumed hydrological flow. This is consistent with the findings of Tsakalos et al. (2018) and Tsakalos et al. (2019) where water availability was hypothesised as a key influencing factor in vegetation distribution.
Overall, the combination of the ordination plots and statistical tests including decision trees suggest that the soil factors ‘providing access to water’ and ‘water-holding capacity’, which both relate to particle size and topography, were the most influential factors in terms of spatial distribution of vegetation grouping in this subpart of Alison Baird Reserve.
Salinity and groundwater
Soil sub-surface salinity and depth to groundwater are related at the site. EC values above 1000 µS cm−1 were found where the average groundwater depth was between 3 and 4.3 m. Soils with EC values of less than 1000 µS cm−1 are considered non-saline (Smith and Doran 1997). Six of the sub-surface samples were saline, as well as one topsoil sample. Soils with depth to groundwater greater than 4.3 m did not display sub-surface soil EC values greater than 500 µS cm−1. Rising groundwater in Western Australia is often associated with vegetation clearing (Bari and Ruprecht 2003; Croton and Bari 2001; Raper et al. 2014). During the winter months, the water table at the study site is perched (Tauss and Weston 2010), which results in the migration of salinity towards the upper layers of the soil profile. The additional issue of increased surface water flow and decreased infiltration is a risk to the site, especially considering the highlighted significance of water availability within this study. The strong relationship between EC and depth to water table suggests there is a potential risk of increased salinity posed by the clearing of land surrounding the site. Although many of the plant species in the SWAFR have developed tolerance to moderate salinity (Halse et al. 2003), the impact of increased salinity on specific endemics within the site is unknown, except for the rare Grevillea thelemanniana, which is highly sensitive to salinity (Gao et al. 2020). Further investigation is required to develop appropriate management solutions.
Potential anthropogenic influence and the role of fire
The anthropogenic impact from limited weed spreading, and human interference in modern times cannot be ignored. Given these landscapes are millions of years old, use of the land by first inhabitants are also a factor worth consideration (Silveira et al. 2021). Anthropological studies examining Noongar interactions with OCBIL and YODFEL landscapes have found that strongest human activity is predominantly associated with YODFEL landscapes in the region (Hopper et al. 2021; Lullfitz et al. 2021). Current population densities across YODFEL landscapes have more than double that of OCBIL landscapes, especially within the SWAFR (Gosper et al. 2021b). Although human interference (or historical lack thereof) is not considered to be the sole driver of diversity and species’ abundance (Gosper et al. 2021b), it has very likely somewhat contributed to the overall (limited) location of endemic species (Silveira et al. 2021).
The lack of correlations between topsoil parameters and sub-surface variables (Fig. 6) may be explained, in part, by weeding activities within the reserve. Weed control has been undertaken at the site in recent years (Hans Lambers pers. comm. 2018), as a management priority. Disturbance from such activities may have resulted in perturbation of topsoil and subsequent displacement from its original locations which could explain erroneous associations. However, the influence of weeds and weeding activity is unlikely to be greater than any signal to noise ratio found within the data collected for this study. At a larger scale, the location of the rare and endemic species at a confined location is likely influenced by the lack of competition from invasive species (Hopper 2009). The physical fragmentation of the study site from other nearby bushland habitats may have limited the migration of invasive species.
Given the age and long-term presence of vegetation at the study site, erosion is unlikely a significant contributor to movement of soil at the site. Where steeper slopes and soil water repellence coexist, soil erosion may be increased (Lowe et al. 2021). Bioturbation of the soil by small marsupials (Isoodon fusciventer, Southwestern brown bandicoot), termites and other insects likely also played a role (Lobry de Bruyn and Conacher 1990; Whitford and Eldridge 2013) as well as fire events that have reduced vegetation cover and allowed movement and redeposition of topsoil. It is possible that processes during the long-term evolution of the landscape have had a greater influence on the distribution of vegetation which limits identification of current environmental drivers (Macintyre et al. 2018; Tsakalos et al. 2018) and further highlights the importance of examining OCBILs and YODFELs as separate systems.
Interest species
We found new information concerning the habitat requirements of several interest species. Previous research suggested that Beaufortia squarrosa prefers well-drained topsoils which can be wet during the winter (Burbidge 2016). This is supported by this study, where sands provided coarse porosity. After the dry summer months, infiltration might be initially restricted by water repellence of the surface soil, but this would quickly change after initial winter rain.
The low P concentration of the soil near Beaufortia squarrosa is consistent with previous studies suggesting P-sensitivity (Nichols and Beardsell 1981). The significantly higher C and N concentrations of the soil surrounding Beaufortia squarrosa may be indicative that the species can tolerate conditions that lead to slow C recycling through low microbial activity. Andersonia gracilis is typically located on seasonally damp black sandy clay soils, near swamps (DEC 2006). At this site, it is located in seasonally damp areas, although within pale yellow topsoil and light-brown to grey sub-surface sands, which have low C and organic matter content. The differences in the silt:clay ratio between Andersonia gracilis and Grevillea thelemanniana in comparison with the rest of the site should also be noted. Relocation of declared rare flora within the Brixton Street Wetlands area has been unsuccessful, possibly due to a lack of understanding of specific habitat requirements (Tauss and Weston 2010). The results from this study will aid conservationists in more appropriately informing management decisions for the declared rare Andersonia gracilis and Grevillea thelemanniana species.
Eutrophication of soil in natural landscapes is an increasing problem, particularly with higher P concentrations resulting from fire events (Lambers et al. 2022), fire suppressants, runoff from agricultural land and verges, and from road dust (Lambers et al. 2013). Many plants within OCBILs, especially within the SWAFR, exhibit P sensitivity (Lambers et al. 2011), including Beaufortia squarrosa (Nichols and Beardsell 1981). The interest species at this site indicate a preference for low-P soils, supporting Lambers et al. (2011). It is unknown whether Andersonia gracilis is P sensitive and can exhibit symptoms of P toxicity. This highlights the risk of eutrophication to the site’s endemic species and overall diversity. Grevillea thelemanniana, like many other Grevillea species, functions at higher leaf P concentrations (Hayes et al. 2021), and, like Grevillea crithmifolia (Shane and Lambers 2006), is not P sensitive (Kosala Ranathunge, pers. comm.)
Future research
Future studies should adapt the methods used by Tsakalos et al. (2018) and Tsakalos et al. (2019) to further examine environmental variables associated with the distribution of plant species across the site. Distance-based regression and analysis can uncover further relationships in a more robust style than was possible for this study. However, species composition data for the site would be required.
A comprehensive understanding of microbial community dynamics can inform the resilience of an ecosystem (Zhao et al. 2019). Although not possible in this study, it could form part of future studies to investigate vegetation diversity drivers and predict responses to climate change. Soil samples have been collected and stored for further analysis.
Investigating hydrological parameters and their influence on water dynamics within the site would be of importance given the displayed influence of particle size, moisture and repellence on the distribution of plants. Groundwater influence at the site is relatively unknown (although hypothesised in Tauss et al. 2019) and can provide further understanding of the potential impacts of surrounding development on the study site (Kros et al. 2016). With rising temperatures and lower rainfall in the region (Fitzpatrick et al. 2008; Ritchie et al. 2021), understanding groundwater-surface water and soil interactions is important. It has recently been hypothesised that due to age of the landscapes, water-soil relationship would be fine-tuned and thus more susceptible to changes in water dynamics, of which a drying climate could be detrimental (Hopper et al. 2021; Horwitz et al. 2003). The role of the water table in drawing salts to the surface is also important, especially to understand the effects nearby clearing may have on the vegetation at the reserve. In addition, modelling weather-vegetation-soil-hydrology dynamics at the site could inform potential impacts of climate and anthropogenic pressures on the reserve and assist in future conservation efforts (Yates et al. 2010a, b).
It is important to note that the samples taken in this study only represent the northeastern part of the reserve which partly limites the number of samples across each community. Despite the partly small sample size, the soil–plant dynamics found in the study, and the relationships that were able to be ascertained, provide an apt insight into the factors influencing vegetation distribution at the site. Future studies should concentrate on achieving a more spatially representative sampling plan to build on the results from this study.
Conclusion
This study provided a baseline investigation of the soil-vegetation-topography relationships of a megadiverse reserve on the Swan Coastal Plain in Western Australia within a metropolitan region that is home to more than 1,800 native plant species (Zemunik 2019). Despite the small area of the reserve (35 ha), the relationship between species richness (> 400 native plant species) and soil characteristics is evident, suggesting the importance of small-scale studies (Hopper 2009).
P, total C and N concentrations were not strongly associated with floristic community distribution for the study area, likely due to the nutrient-impoverished nature of the site. Plant diversity and distribution on a regional scale may be primarily driven by nutrient status (Laliberté et al. 2014); however, local-scale distribution illustrates a greater association with water-proxy factors. The combination of strong associations with elevation, particle size, depth to water table, and repellence suggests that water dynamics at the site is a key factor associated with vegetation distribution.
Groundwater salinity likely influences the distribution of species within the site. Associations with both top and sub-surface soils illustrates that it is imperative to examine factors at multiple soil depths to fully understand an ecological system.
In terms of interest species, the distribution of Beaufortia squarrosa was expected to be based on soil texture, with higher overall silt and clay, and a higher clay:silt ratio than the soils of other species of interest. C and N concentrations of soils associated with Beaufortia squarrosa were significantly higher than those of the site in general, but still low compared with global averages. Consistent with previous studies, the interest species preferred soils with lower P concentrations.
The insight into the soils and soil-vegetation-topography relationships across the site is an important step in understanding factors of vegetation distribution at Alison Baird Reserve. The results from this study can be used to further aid conservation of OCBIL landscapes on the Swan Coastal Plain and beyond, bolster preservation efforts at the site and inform management decisions to help protect vegetation from adverse effects of surrounding development. This study highlights the importance of preserving and studying even small fractured OCBIL landscapes and minimising human interference. It further highlights the importance of in-depth soil analyses including both topsoil and subsurface samples.
Abbreviations
- OCBIL:
-
Old, climatically buffered, deeply weathered, infertile landscapes
- YODFEL:
-
Young, often disturbed, fertile landscapes
- DWT:
-
Depth to water table (metres)
- SWAFR:
-
Southwest Australian Floristic Region
- ERT:
-
Electrical resistivity tomography
- GPR:
-
Ground-penetrating radar
References
Alston KP, Richardson DM (2006) The roles of habitat features, disturbance, and distance from putative source populations in structuring alien plant invasions at the urban/wildland interface on the Cape Peninsula, South Africa. Biol Conserv 1322:183–198
Bari MA, Ruprecht J (2003) Water yield response to land use change in South-west Western Australia. Department of Environment. Salinity Impact Series Report No. SLUI 31
Bouwer H (1978) Groundwater hydrology. McGraw-Hill, New York
Breiman L, Friedman JH, Olshen RA, Stone CJ (1984) Classification and regression trees. Chapman and Hall, Wadsworth, New York
Burbidge A (2016) A taxonomic revision of Beaufortia (Myrtaceae: Melaleuceae). Nuytia 27:165–202
Caccetta P, Collings S, Devereux A, Hingee K, Mcfarlane D, Traylen A, Wu X, Zhou Z (2012) Urban monitor: enabling effective monitoring and management of urban and coastal environments using digital aerial photography final report–transformation of aerial photography into digital raster information products. CSIRO. csiro:EP129592. https://doi.org/10.4225/08/58542ca49ec49
Cesarano G, Incerti G, Bonanomi G (2016) The influence of plant litter on soil water repellency: insight from 13C NMR spectroscopy. PLoS One 113:e0152565
Coates D, Byrne M, Cochrane A, Dunne C, Gibson N, Keighery G, Lambers H, Monks L, Thiele K, Yates C (2014) Conservation of the kwongan flora: threats and challenges. In: Lambers H (ed) Plant life on the sandplains in southwest Australia, a global biodiversity hotspot. UWA Publishing, pp 263–284
Croton J, Bari M (2001) Using WEC-C, a distributed, deterministic catchment model, to simulate hydrologic responses to agricultural clearing. Environ Model Softw 167:601–614
De’ath G, Fabricius KE (2000) Classification and regression trees: a powerful yet simple technique for ecological data analysis. Ecology 81:3178–3192
Department of Environment and Conservation (DEC) (2006) Slender Andersonia (Andersonia gracilis) Interim Recovery Plan 2006-2011, Interim Recovery Plan No. 228. Department of Western Australia, Perth
Didham RK, Tylianakis JM, Gemmell NJ, Rand TA, Ewers RM (2007) Interactive effects of habitat modification and species invasion on native decline. Trends Ecol Evol 229:489–496
Fitzpatrick MC, Gove AD, Sanders NJ, Dunn RR (2008) Climate change, plant migration, and range collapse in a global biodiversity hotspot: the Banksia (Proteaceae) of Western Australia. Glob Change Biol 14:1337–1352
Fordyce IR, Gilkes R, Loneragan WA, Beale S, Middleton N (2007) Vegetation zoning in relation to site and soil properties: a case study in the Darling Range, south-western Australia. J R Soc Western Australia 90:1–14
Fox J, Weisberg S (2015) An R companion to applied regression. https://socialsciences.mcmaster.ca/jfox/Books/Companion. Accessed Nov 2021
Franco CMM, Clarke PJ, Tate ME, Oades JM (2000) Hydrophobic properties and chemical characterisation of natural water repellent materials in Australian sands. J of Hydrol 231–232:47–58
Franks AJ, Yates CJ, Hobbs RJ (2009) Defining plant functional groups to guide rare plant management. Plant Ecol 204:207–216
Gao J, Wang F, Ranathunge K, Arruda AJ, Cawthray GR, Clode PL, He X, Leopold M, Roessner U, Rupasinghe T, Zhong H, Lambers H (2020) Edaphic niche characterization of four Proteaceae reveals unique calcicole physiology linked to hyper-endemism of Grevillea thelemanniana. New Phytol 228(3):869–883
Gibson N, Keighery GJ, Lyons MN, Webb A (2004) Terrestrial flora and vegetation of the Western Australian wheatbelt. Rec West Aust Mus 67:139–189
Gilkes RJ, Scholz G, Dimmock GM (2006) Lateritic deep weathering of granite. J Soil Sci 24(4):523–536
Gill AM, Williams JE (1996) Fire regimes and biodiversity: the effects of fragmentation of southeastern Australian eucalypt forests by urbanisation, agriculture and pine plantations. For Ecol Manag 851–3:261–278
Gleeson D, Mathes F, Farrell M, Leopold M (2016) Environmental drivers of soil microbial community structure and function at the Avon River Catchment Critical Zone Observatory. Sci Total Environ 15(571):1407–1418
Gosper CR, Coates DJ, Hopper SD, Byrne M, Yates C (2021a) The role of landscape history in the distribution and conservation of threatened flora in the Southwest Australian Floristic Region. Biol J Linn Soc 133:394–410
Gosper CR, Kinloch J, Coates DJ, Byrne M, Pitt G, Yates CJ (2021b) Differential exposure and susceptibility to threats based on evolutionary history: how OCBIL theory informs flora conservation. Biol J Lin Soc 133:373–393
Halse SA, Ruprecht JK, Pinder AM (2003) Salinisation and prospects for biodiversity in rivers and wetlands of south-west Western Australia. Aust J Bot 51:673–688
Harper RJ, Gilkes RJ (1994) Soil attributes related to water repellency and the utility of soil survey for predicting its occurrence. Aust J Soil Res 32:1109–1124
Hayes PE, Nge FJ, Cramer MD, Finnegan PM, Fu P, Hopper SD, Oliveira RO, Turner BL, Zemunik G, Zhong H, Lambers H (2021) Traits related to efficient acquisition and use of phosphorus promote diversification in Proteaceae in old phosphorus-impoverished landscapes. Plant Soil 462:67–88
Hopper SD (2009) OCBIL theory: towards an integrated understanding of the evolution, ecology and conservation of biodiversity on old, climatically buffered, infertile landscapes. Plant Soil 322:49–86
Hopper SD, Gioia P (2004) The southwest Australian floristic region: evolution and conservation of a global hot spot of biodiversity. Annu Rev Ecol Evol Syst 35:623–650
Hopper S, Silveira F, Fiedler P (2016) Biodiversity hotspots and Ocbil theory. Plant Soil 403:167–216
Hopper SD, Lambers H, Silveira FA, Fiedler PL (2021) OCBIL theory examined: reassessing evolution, ecology and conservation in the world’s ancient, climatically buffered and infertile landscapes. Biol J Linn Soc 133:266–296
Hopper S (2014) Sandplain and Kwongkan: historical spellings, meanings, synonyms, geography and definition. In: Lambers H (Ed) Plant life on the sandplains in southwest Australia, a global biodiversity hotspot. UWA Publishing, pp 23–33
Horwitz P, Judd S, Sommer B (2003) Fire and organic substrates: soil structure, water quality and biodiversity in far southwest Western Australia. In: Abbott I, Burrows N (eds) Fire in ecosystems of south-west Western Australia: Impacts and management. Backhuys Publishers, The Netherlands, pp 381–393
Hunt N, Gilkes R (1992) Farm monitoring handbook. UWA Publishing, Perth
Kros J, Mol-Dijkstra JP, Wamelink GWW, Reinds GJ, van Hinsberg A, de Vries W (2016) Modelling impacts of acid deposition and groundwater level on habitat quality and plant species diversity. Ecol Process 5:22. https://doi.org/10.1186/s13717-016-0066-0
Laliberté E, Turner BL, Zemunik G, Wyrwoll KH, Pearse SJ, Lambers H (2013) Nutrient limitation along the Jurien Bay dune chronosequence: response to Uren & Parsons. J Ecol 1015:1088–1092
Laliberté E, Zemunik G, Turner BL (2014) Environmental filtering explains variation in plant diversity along resource gradients. Science 345:1602–1605
Laliberté E, Lambers H, Burgess TI, Wright SJ (2015) Phosphorus limitation, soil-borne pathogens and the coexistence of plant species in hyperdiverse forests and shrublands. New Phytol 2062:507–521
Lambers H (2019) A jewel in the crown of a global biodiversity hotspot. Kwongan Foundation and Western Australian Naturalists' Club Inc ISBN 0980641748, 9780980641745, 440p
Lambers H, Mougel C, Jaillard B, Hinsinger P (2009) Plant-microbe-soil interactions in the rhizosphere: an evolutionary perspective. Plant Soil 3211–2:83–115
Lambers H, Brundrett MC, Raven JA, Hopper SD (2011) Plant mineral nutrition in ancient landscapes: high plant species diversity on infertile soils is linked to functional diversity for nutritional strategies. Plant Soil 3481–2:7
Lambers H, Ahmedi I, Berkowitz O, Dunne C, Finnegan PM, Hardy GESJ, Jost R, Laliberté E, Pearse SJ, Teste FP (2013) Phosphorus nutrition of phosphorus-sensitive Australian native plants: threats to plant communities in a global biodiversity hotspot. Conserv Physiol 1(1):cot010
Lambers H, Albornoz F, Kotula L, Laliberté E, Ranathunge K, Teste FP, Zemunik G (2018) How belowground interactions contribute to the coexistence of mycorrhizal and non- mycorrhizal species in severely phosphorus-impoverished hyperdiverse ecosystems. Plant Soil 424:11–33
Lambers H, de Britto Costa P, Cawthray GR, Denton MD, Finnegan PM, Hayes PE, Oliveira RS, Power SC, Ranathunge K, Shen Q, Wang X (2022) Strategies to acquire and use phosphorus in phosphorus-impoverished and fire-prone environments. Plant Soil 476:133–160. https://doi.org/10.1007/s11104-022-05464-8
Lane P, Evans KA (2019) Geology of the proposed Yule Brook Regional park. In: Lambers H (ed) A jewel in the crown of a global biodiversity hotspot. Kwongan Foundation and the Western Australian Naturalists’ Club Inc, Perth, pp 23–31
Leopold M, Zhong H (2019) The soils of the Alison Baird Reserve. In: Lambers H (ed) A jewel in the crown of a global biodiversity hotspot. Kwongan Foundation and the Western Australian Naturalists’ Club Inc, Perth, pp 49–57
Leopold M, Gupanis-Broadway C, Baker A, Hankin S, Treble P (2021) Time lapse electric resistivity tomography to portray infiltration and hydrologic flow paths from surface to cave. J Hydrol 593:125810
Lobry de Bruyn L, Conacher AJ (1990) The role of termites and ants in soil modification: a review. Aust J Soil R 28:58–93
Loke MH (2019) RES2DINVx64 ver. 4.09, Rapid 2-D Resistivity & IP inversion using the least-squares method. Available at http://www.geotomosoft.com. 7/01/2013
Lowe MA, McGrath G, Leopold M (2021) The impact of soil water repellency and slope upon runoff and erosion. Soil Tillage Res 205:104756
Lullfitz A, Pettersen C, Reynolds R, Eades A, Dean A, Knapp L, Woods E, Woods T, Eades E, Yorkshire-Selby S, Woods S, Dortch J, Guilfoyle D, Hopper S (2021) The Noongar of south-western Australia: a case study of long-term biodiversity conservation in a matrix of old and young landscapes. Biol J Linn Soc 133:432–448
Macintyre P, Van Niekerk A, Dobrowolski M, Tsakalos J, Mucina L (2018) Impact of ecological redundancy on the performance of machine learning classifiers in vegetation mapping. Ecol Evol 8:6728–6737
Marchese C (2015) Biodiversity hotspots: a shortcut for a more complicated concept. Glob Ecol Conserv 3:297–309. https://doi.org/10.1016/j.gecco.2014.12.008
McKenzie N, Coughlan K, Cresswell H (2002) Soil physical measurement and interpretation for land evaluation. CSIRO Publishing
McKenzie N, Jacquier D, Isbell R, Brown K (2004) Australian soils and landscapes: an illustrated compendium. CSIRO Publishing, Collingwood
Moeslund JE, Arge L, Bøcher PK, Dalgaard T, Svenning JC (2013) Topography as a driver of local terrestrial vascular plant diversity patterns. Nord J Bot 312:129–144
Monks L, Barrett S, Beecham B, Byrne M, Chant A, Coates D, Cochrane JA, Crawford A, Dillon R, Yates C (2019) Recovery of threatened plant species and their habitats in the biodiversity hotspot of the Southwest Australian Floristic Region. Plant Div 41:59–74
Mucina L, Laliberté E, Thiele K, Dodson J, Harvey J (2014) The biogeography of Kwongan: origins, diversity, endemism and vegetation patterns. In: Lambers H (ed) Plant life on the sandplains in Southwest Australia a global biodiversity hotspot. UWA Publishing, Australia, pp 35–79
Myers N (1990) The biodiversity challenge: Expanded hot-spots analysis. Environmentalist 10:243–256. https://doi.org/10.1007/BF02239720
Neal A (2004) Ground-penetrating radar and its use in sedimentology: principles, problems and progress. Earth-Sci Rev 66(3):261–330
Nichols D, Beardsell D (1981) The response of phosphorus-sensitive plants to slow- release fertilizers in soil-less potting mixtures. Sci Hort 154:301–309
O’Brien FJ, Almaraz M, Foster MA, Hill AF, Huber DP, King EK, Langford H, Lowe M-A, Mickan BS, Miller VS, Moore OW, Mathes F, Gleeson D, Leopold M (2019) Soil salinity and pH drive soil bacterial community composition and diversity along a lateritic slope in the Avon River Critical Zone Observatory, Western Australia. Front Microbiol 10:1486. https://doi.org/10.3389/fmicb.2019.01486
Oksanen J (2015) Vegan: an introduction to ordination, 8:19. http://cran.r-project.org/web/packages/vegan/vignettes/introvegan.pdf. Accessed June 2022
Pressey RL, Cabeza M, Watts ME, Cowling RM, Wilson KA (2007) Conservation planning in a changing world. Trends Ecol Evol 22:583–592
Prober SM, Wiehl G (2012) Relationships among soil fertility, native plant diversity and exotic plant abundance inform restoration of forb-rich eucalypt woodlands. Divers Distrib 188:795–807
R Core Team (2022) R: a language and environment for statistical computing. R Foundation for Statistical Computing, Vienna. https://www.R-project.org. Accessed Nov 2022
Raper GP, Speed RJ, Simons JA Killen AL, Blake AI, Ryder AT, Smith RH, Stainer GS, Bourke L (2014) Groundwater trend analysis for south-west Western Australia 2007–12, Resource management technical report 388. Department of Agriculture and Food, Western Australia, Perth
Rayment GE, Lyons DJ (2011) Soil chemical methods. CSIRO Publishing, Australasia
Reimann C, Filzmoser P, Garrett RG, Dutter R (2008) Statistical data analysis explained. Applied environmental statistics with R. Wiley & Sons Ltd, England
Richards MB, Cowling RM, Stock WD (1997) Soil nutrient dynamics and community boundaries in the fynbos vegetation of South Africa. Plant Ecol 130:143–153
Ritchie AL, Svejcar LN, Ayre BM, Bolleter J, Brace A, Craig MD, Davis B, Davis RA, Van Etten EJ, Fontaine JB, Fowler WM (2021) A threatened ecological community: research advances and priorities for Banksia woodlands. Aust J Bot 69:111
Rouget M, Cowling RM, Pressey RL, Richardson DM (2003) Identifying spatial components of ecological and evolutionary processes for regional conservation planning in the Cape Floristic Region, South Africa. Divers Distrib 9:191–210
Scott B, Lantzke R, Nicholson D, Findlater P (2012) Comparison studies of unsaturated flow below annual and perennial plants. Water Sci Technol 6512:2162–2168
Shackelford N, Renton M, Perring MP, Brooks K, Hobbs RJ (2014) Biodiversity change in heathland and its relationships with shifting local fire regimes and native species expansion. J Plant Ecol 81:17–29
Shane MW, Lambers H (2006) Systemic suppression of cluster-root formation and net P-uptake rates in Grevillea crithmifolia at elevated P supply: a proteacean with resistance for developing symptoms of ‘P toxicity.’ J Exp Bot 57:413–423
Silveira FA, Fiedler PL, Hopper SD (2021) OCBIL theory: a new science for old ecosystems. Biol J Linn Soc 133:373–393
Smith JL, Doran JW (1997) Measurement and use of pH and electrical conductivity for soil quality analysis. In: Doran JW, Jones AJ (eds) Methods for Assessing Soil Quality. American Society of Agronomy, Crop Science Society of America, and Soil Science Society of America, Madison, pp 169–185
Speck NH, Baird AM (1984) Vegetation of Yule Brook Reserve near Perth, Western Australia. J R Soc Western Australia 66:147–162
Standish RJ, Hobbs RJ (2010) Restoration of OCBILs in South-Western Australia: response to Hopper. Plant Soil 330:15–18
Standish RJ, Cramer VA, Wild SL, Hobbs RJ (2007) Seed dispersal and recruitment limitation are barriers to native recolonization of old-fields in Western Australia. J Appl Ecol 44:435–445
Struglia R, Winter PL (2002) The role of population projections in environmental management. Environ Manag 301:13–23
Tauss C, Weston A (2010) The flora, vegetation and wetlands of the Maddington- Kenwick Strategic Employment Area. A survey of rural lands in the vicinity of the Greater Brixton Street Wetlands. Unpublished report to the City of Gosnells, Western Australia
Tauss C, Keighery GJ, Keighery BJ, Cloran PM, Genovese SD (2019) A new look at the flora and the vegetation patterns of the Greater Brixton Street Wetlands and Yule Brook. A jewel in the crown of a global biodiversity hotspot. Kwongan Foundation and the Western Australian Naturalists’ Club Inc, Perth, Australia, pp 69–207
Therneau T, Atkinson B (2019) Package ‘rpart’. https://cran.r-project.org/web/packages/rpart/rpart.pdf. Accessed Nov 2021
Tsakalos JL, Renton M, Dobrowolski MP, Feoli E, Macintyre PD, Veneklaas EJ, Mucina L (2018) Community patterns and environmental drivers in hyper-diverse kwongan scrub vegetation of Western Australia. Appl Veg Sci 214:694–722
Tsakalos JL, Renton M, Dobrowolski MP, Veneklaas EJ, Macintyre PD, Broomfield SJ, Mucina L (2019) Composition and ecological drivers of the kwongan scrub and woodlands in the northern Swan Coastal Plain, Western Australia. Austral Ecol 44:906–916
Turner BL, Hayes PE, Laliberté E (2018) A climosequence of chronosequences in southwestern Australia. Eur J Soil Sci 69:69–85
Vincent AG, Sundqvist MK, Wardle DA, Giesler R (2014) Bioavailable soil phosphorus decreases with increasing elevation in a Subarctic Tundra landscape. PLoS One 93:e92942
WA DoW - Western Australia Department of Water (2015) Perth Groundwater Map. Available via: http://www.water.wa.gov.au/maps-and-data/maps/perth-groundwater-atlas. Accessed Nov 2019
WA Herbarium (1998-) FloraBase—the Western Australian Flora. Department of Environment and Conservation. Available via http://florabase.dec.wa.gov.au. Accessed Nov 2018
Wang M, Dong Z, Luo W, Lu J, Li J (2015) Spatial variability of vegetation characteristics, soil properties and their relationships in and around China’s Badain Jaran Desert. Environ Earth Sci 74:6847–6858
Wege J (1992) Baseline studies for future management at Yule Brook Reserve, Kenwick. Unpublished thesis
White RE (2005) Principles and practice of soil science: the soil as a natural resource, 4th edn. John Wiley & Sons
Whitford WG, Eldridge DJ (2013) Effects of ants and termites on soil and geomorphological processes. In: Shroder JF (ed) Treatise on geomorphology. Academic Press, San Diego, pp 281–292
Wyrwoll K-H, Turner BL, Findlater P (2014) On the origins, geomorphology and soils of the sandplains of south-western Australia. In: Lambers H (ed) Plant life on the sandplains in Southwest Australia a global biodiversity hotspot. UWA Publishing, Australia, pp 3–22
Yates CJ, Elith J, Latimer AM, Le Maitre D, Midgley GF, Schurr FM, West AG (2010a) Projecting climate change impacts on species distributions in megadiverse South African Cape and Southwest Australian Floristic Regions: opportunities and challenges. Austral Ecol 354:374–391
Yates CJ, McNeill A, Elith J, Midgley GF (2010b) Assessing the impacts of climate change and land transformation on Banksia in the South West Australian Floristic Region. Divers Distrib 161:187–201
Zemunik G (2019) Diversity of the Yule Brook region in context. In: Lambers H (ed) A jewel in the crown of a global biodiversity hotspot. Kwongan Foundation and the Western Australian Naturalists’ Club Inc, Perth, pp 59–68
Zhao C, Long J, Liao H, Zheng C, Li J, Liu L, Zhang M (2019) Dynamics of soil microbial communities following vegetation succession in a karst mountain ecosystem, Southwest China. Sci Rep 91:2160
Acknowledgements
We thoroughly thank the editor and two anonymous reviewers for their comments and suggestions which helped to further shape and improve this study. We are very thankful to Professor Hans Lambers for introducing us to the site and for many insightful discussions and an early revision of the manuscript. We further thank Cate Tauss for her help in identifying plant communities and for her contributions in discussing results. We are also thankful to Darryl Roberts for assisting with soil analysis.
Funding
Open Access funding enabled and organized by CAUL and its Member Institutions.
Author information
Authors and Affiliations
Contributions
Caitlin Smith contributed to field work and led the laboratory and statistical analyses. She provided the first draft of the manuscript and integrated subsequent remarks from all authors. Bryan Buroff contributed to the results discussion, and the manuscript writing and editing. Gustavo Boitt helped with laboratory analyses, guided some of the statistical analyses and contributed to the discussion of results and the writing of the manuscript. Matthias Leopold oversaw the concept, helped during field and laboratory analyses and contributed to the discussion of the results and the writing of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Additional information
Responsible Editor: Hans Lambers.
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Smith, C.E., Boitt, G., Boruff, B. et al. Soil properties and floristic community distribution within a deeply weathered landscape within a biodiversity hotspot of Western Australia. Plant Soil 487, 109–132 (2023). https://doi.org/10.1007/s11104-023-05918-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-023-05918-7