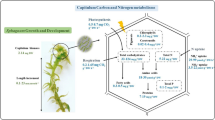
Abstract
Aims
To test the effects of characteristic ecological gradients in peatlands including oxygen-deficiency and allelopathy on Sphagnum spore persistence.
Methods
We determined the initial viability of Sphagnum spores and then stored the spores for 60 days, either dry, in ultrapure water, peatland surface water or Sphagnum water leachate (the latter two solutions rich in allelochemicals from Sphagnum). We varied oxygen concentration by injecting air at three different concentrations during the storage experiment. After retrieval from experimental storage, spore germinability index was assessed.
Results
Germinability index after air injection was lower than under oxygen-deficiency. Germinability index in the peatland surface water and Sphagnum leachate water was higher than in ultrapure water, under oxygen-deficiency. Overall, dissolved oxygen and high pH negatively affected spore germinability and, consequently, viability.
Conclusions
These results indicate that once dispersed onto bryophyte substrates or waterlogged hollows, Sphagnum spores can maintain viability better than when exposed to dry conditions or in water without allelochemicals. Extreme longevity of Sphagnum spores may be attributed to the oxygen-deficient, waterlogged and acidic Sphagnum-dominated peatland habitat with allelopathic substrates.





Similar content being viewed by others
References
Abbott GD, Swain EY, Muhammad AB, Allton K, Belyea LR, Laing CG, Cowiec GL (2013) Effect of water-table fluctuations on the degradation of Sphagnum phenols in surficial peats. Geochim Cosmochim Acta 106:177–191. https://doi.org/10.1016/j.gca.2012.12.013
Battle JM, Golladay SW (2001) Hydroperiod influence on breakdown of leaf litter in cypress-gum wetlands. Am Midl Nat 146:128–145. https://doi.org/10.1674/0003-0031(2001)146[0128:hiobol]2.0.co;2
Benvenuti S, Macchia M (1995) Effect of hypoxia on buried weed seed germination. Weed Res 35:343–351. https://doi.org/10.1111/j.1365-3180.1995.tb01629.x
Boatman DJ, Lark PM (1971) Inorganic nutrition of the protonemata of Sphagnum papillosum Lindb., S. magellanicum Brid. And S. cuspidatum Ehrh. New Phytol 70:1053–1059. https://doi.org/10.1111/j.1469-8137.1971.tb04587.x
Bu ZJ, Sundberg S, Feng L, Li HK, Zhao HY, Li HC (2017a) The methuselah of plant diaspores: Sphagnum spores can survive in nature for centuries. New Phytol 214:1398–1402. https://doi.org/10.1111/nph.14575
Bu ZJ, Li Z, Liu LJ, Sundberg S, Feng YM, Yang YH, Liu S, Song X, Zhang XL (2017b) Bryophyte spore germinability is inhibited by peatland substrates. Acta Oecol 78:34–40. https://doi.org/10.1016/j.actao.2016.11.003
Clymo RS (1963) Ion exchange in Sphagnum and its relation to bog ecology. Ann Bot 27:309–324. https://doi.org/10.1093/oxfordjournals.aob.a083847
Clymo RS (1967) Control of cation concentrations, and in particular of pH, in Sphagnum dominated communities. In: Golterman HL, Clymo RS (eds) Chemical environment in aquatic habitat. North Holland, Amsterdam, pp 273–284
Corstanje R, Reddy KR (2004) Response of biogeochemical indicators to a drawdown and subsequent reflood. J Environ Qual 33:2357–2366. https://doi.org/10.2134/jeq2004.2357
Cronberg N (1993) Reproductive biology of Sphagnum. Lindbergia 17:69–82
During HJ (2001) Diaspore banks. Bryologist 104(1):92–97. https://doi.org/10.1639/0007-2745(2001)104[0092:DB]2.0.CO;2
Fan SG, Wang YR, Baskin CC, Baskin JM, Hu XW (2016) A rapid method to determine germinability and viability of Agriophyllum squarrosum (Amaranthaceae) seeds. Seed Sci Technol 44:1–6. https://doi.org/10.15258/sst.2016.44.2.01
Feng L, Bu ZJ, Mallik A, Wang ZC, Liu SS, Wu YH (2017) Continuous waterlogging may not facilitate germinability maintenance of Sphagnum spores. Wetlands 37:1015–1022. https://doi.org/10.1007/s13157-017-0936-y
Frahm JP (2008) Diversity, dispersal and biogeography of bryophytes (mosses). Biodivers Conserv 17:277–284. https://doi.org/10.1007/s10531-007-9251-x
Funes G, Basconcelo S, Díaz S, Cabido M (1999) Seed size and shape are good predictors of seed persistence in soil in temperate mountain grasslands of Argentina. Seed Sci Res 9:341–345. https://doi.org/10.1017/S0960258599000355
Glime JM (2017) Bryophyte ecology. Michigan Technological University, Houghton
Griffin C (2017) Temperature signals in seed germination. Science 356(6345):1347. https://doi.org/10.1126/science.356.6345.1347-d
Hájek T, Ballance S, Limpens J, Zijlstra M, Verhoeven JTA (2011) Cell-wall polysaccharides play an important role in decay resistance of Sphagnum and actively depressed decomposition in vitro. Biogeochemistry 103:45–57. https://doi.org/10.1007/s10533-010-9444-3
Hättenschwiler S, Vitousek PM (2000) The role of polyphenols in terrestrial ecosystem nutrient cycling. Trends Ecol Evol 15:238. https://doi.org/10.1016/S0169-5347(00)01861-9
Jauhiainen S (1998) Seed and spore banks of two boreal mires. Ann Bot Fenn 35:197–201
Johnston CA, Brown TN (2013) Water chemistry distinguishes wetland plant communities of the Great Lakes coast. Aquat Bot 104:111–120. https://doi.org/10.1016/j.aquabot.2012.08.005
Kato-Noguchi H, Seki T, Shigemori H (2010) Allelopathy and allelopathic substance in the moss Rhynchostegium pallidifolium. J Plant Physiol 167:468–471. https://doi.org/10.1016/j.jplph.2009.10.018
Meuter-Gerhards A, Riegert S, Wiermann R (1999) Studies on sporopollenin biosynthesis in Cucurbita maxima (DUCH.)—II. The involvement of aliphatic metabolism. J Plant Physiol 154:431–436. https://doi.org/10.1016/S0176-1617(99)80279-X
Michel P, Burritt DJ, Lee WG (2011) Bryophytes display allelopathic interactions with tree species in native forest ecosystems. Oikos 120:1272–1280. https://doi.org/10.1111/j.1600-0706.2010.19148.x
Mishler BD (1988) Reproductive ecology of bryophytes. In: Plant reproductive biology patterns and strategies, pp 285–306
Mishler BD, Newton AE (1988) Influences of mature plants and desiccation on germination of spores and gametophyticfragments of Tortula. J Bryol 15:327–342. https://doi.org/10.1179/jbr.1988.15.2.327
Mogensen GS (1981) The biological significance of morphological characters in bryophytes: the spore. Bryologist 84:187–207. https://doi.org/10.2307/3242821
Mogensen GS (1983) The spore. In: Schuster RM (eds) New Manual of Bryology. Hattori Botanical Laboratory, Nichinan, Japan, pp 325–342
Newton AE, Mishler BD (1994) The evolutionary significance of asexual reproduction in mosses. J Hattori Bot Lab 76:127–145
Nosratti I, Amiri S, Bagheri A, Chauhan BS (2017) Environmental factors affecting seed germination and seedling emergence of foxtail Sophora (Sophora alopecuroides). Weed Sci 66(1):1–7. https://doi.org/10.1017/wsc.2017.35
Ohlrogge JB, Kernan TP (1982) Oxygen-dependent aging of seeds. Plant Physiol 70:791–794. https://doi.org/10.1104/pp.70.3.791
Oliver MJ (2008) Biochemical and molecular mechanisms of desiccation tolerance in bryophytes. Cambridge University Press
Ooi MKJ, Auld TD, Denham AJ (2009) Climate change and bet-hedging: interactions between increased soil temperatures and seed bank persistence. Glob Chang Biol 15:2375–2386. https://doi.org/10.1111/j.1365-2486.2009.01887.x
Pons TL (1991) Dormancy, germination and mortality of seeds in a chalk-grassland flora. J Ecol 79(3):765–780. https://doi.org/10.2307/2260666
Proctor MCF, Oliver MJ, Wood AJ, Alpert P, Stark LR, Cleavitt NL, Mishler BD (2007) Desiccation-tolerance in bryophytes: a review. Bryologist 110:595–621. https://doi.org/10.1639/0007-2745(2007)110[595:DIBAR]2.0.CO;2
Rajjou L, Debeaujon I (2008) Seed longevity: survival and maintenance of high germination ability of dry seeds. CR Biol 331:796. https://doi.org/10.1016/j.crvi.2008.07.021
Rosengren F, Cronberg N (2015) Selective spore germination on shoots of Homalothecium lutescens, a moss with dwarf males. Biol Lett 11:137–141. https://doi.org/10.1098/rsbl.2015.0427
Rudolph H, Kirchhoff M, Gliesmann S (1988) Sphagnum culture techniques. In: Glime JM (eds) Methods in bryology. Hattori Botanical Laboratory, Nichinan, pp 25–34
Schwember AR, Bradford KJ (2011) Oxygen interacts with priming, moisture content and temperature to affect the longevity of lettuce and onion seeds. Seed Sci Res 21:175–185. https://doi.org/10.1017/S0960258511000080
Sundberg S (2013) Spore rain in relation to regional sources and beyond. Ecography 36:364–373. https://doi.org/10.1111/j.1600-0587.2012.07664.x
Sundberg S, Rydin H (2000) Experimental evidence for a persistent spore bank in Sphagnum. New Phytol 148:105–116. https://doi.org/10.1046/j.1469-8137.2000.00746.x
Sundberg S, Rydin H (2002) Habitat requirements for establishment of Sphagnum from spores. J Ecol 90:268–278. https://doi.org/10.1046/j.1365-2745.2001.00653.x
Thompson K, Band SR, Hodgson JG (1993) Seed size and shape predict persistence in soil. Funct Ecol 7:236–241. https://doi.org/10.2307/2389893
Thompson K, Bakker JP, Bekker RM (1997) The soil seed banks of North West Europe: methodology, density and longevity. Cambridge University Press
van Breemen N (1995) How Sphagnum bogs down other plants. Trends Ecol Evol 10:270–275. https://doi.org/10.1016/0169-5347(95)90007-1
van Zanten BO (1978) Experimental studies on trans-oceanic long-range dispersal of moss spores in the southern hemisphere. J Hattori Bot Lab 44:455–482
Venable DL, Brown JS (1988) The selective interactions of dispersal, dormancy, and seed size as adaptations for reducing risk in variable environments. Am Nat 131:360–384. https://doi.org/10.1086/284795
Verhoeven JTA, Liefveld WM (1997) The ecological significance of organochemical compounds in Sphagnum. Acta Bot Neerl 46:117–130. https://doi.org/10.1111/plb.1997.46.2.117
Wallace S, Fleming A, Wellman CH, Beerling DJ (2011) Evolutionary development of the plant spore and pollen wall. AoB Plants, plr027
Wheeler BD, Proctor MCF (2000) Ecological gradients, subdivisions and terminology of north-west European mires. J Ecol 88:187–203. https://doi.org/10.1046/j.1365-2745.2000.00455.x
Wiklund K, Rydin H (2004) Ecophysiological constraints on spore establishment in bryophytes. Funct Ecol 18(6):907–913. https://doi.org/10.1111/j.0269-8463.2004.00906.x
Yu Z, Dahlgren RA (2000) Evaluation of methods for measuring polyphenols in conifer foliage. J Chem Ecol 26(9):2119–2140. https://doi.org/10.1023/A:1005568416040
Zhao LP, Wu GL, Cheng JM (2011) Seed mass and shape are related to persistence in a sandy soil in northern China. Seed Sci Res 21:47–53. https://doi.org/10.1017/S0960258510000358
Acknowledgements
This study was funded by the National Key Research and Development Project (No. 2016YFA0602301 and No. 2016YFC0500407), the National Nature Science Foundation of China (No. 41471043, No. 41571049 and No. 41871046). The authors thank Jin-Ze Ma and Chao Liu for peatland surface water collection.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Gustavo Gabriel Strike
Electronic supplementary material
ESM 1
(DOCX 164 kb)
Rights and permissions
About this article
Cite this article
Feng, L., Sundberg, S., Ooi, M.K.J. et al. Oxygen-deficiency and allelochemicals affect Sphagnum spore persistence in peatlands. Plant Soil 432, 403–413 (2018). https://doi.org/10.1007/s11104-018-3809-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-018-3809-0