Abstract
Apocynaceae, one of the largest plant families with over 5.100 widely distributed species, serves as a significant model for evolutionary and biochemical studies. Within this family, compounds such as alkaloids, flavonoids, and terpenoids with potential therapeutic activities have been isolated. Among its subfamilies, Asclepiadoideae is recognized as a rich source of C21 pregnane derivatives. This paper presents a comprehensive review of pregnane glycosides (PG) obtained from Asclepiadoideae, employing an interdisciplinary approach that combines chemophenetics with neural network data analysis to identify promising species and bioactive compounds. A dataset comprising 660 unique PGs and 705 botanical occurrences was compiled for this purpose. The ethnopharmacological aspects, as well as the extraction, isolation, and biological activity of PGs, were analyzed and discussed. Chemophenetic analysis using Self-Organizing Maps (SOM) and phylogeny reconstruction revealed similarities between the Marsdenia-Ceropegiae tribes and highlighted the occurrence of seco and diseco pregnane types predominantly within the Asclepiadeae tribe. Both scaffolds exhibited intriguing structural features, and their derivatives have demonstrated anti-inflammatory and cytotoxic activities. These findings provide valuable insights for future research on these compounds and their medicinal plant sources.
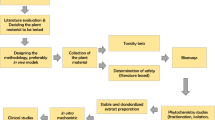
Graphical abstract

















Similar content being viewed by others
References
Abdallah HM, Osman A-MM, Almehdar H, Abdel-Sattar E (2013) Acylated pregnane glycosides from Caralluma quadrangula. Phytochemistry 88:54–60. https://doi.org/10.1016/j.phytochem.2012.12.005
Abdel-Sattar E, Ali DE (2022) Russelioside B: a pregnane glycoside with pharmacological potential. Rev Bras 32:188–200. https://doi.org/10.1007/s43450-022-00245-x
Abdel-Sattar E, Harraz FM, Al-ansari SMA et al (2008) Acylated pregnane glycosides from Caralluma tuberculata and their antiparasitic activity. Phytochemistry 69:2180–2186. https://doi.org/10.1016/j.phytochem.2008.05.017
Abdel-Sattar E, Shehab NG, Ichino C et al (2009) Antitrypanosomal activity of some pregnane glycosides isolated from Caralluma species. Phytomedicine 16:659–664. https://doi.org/10.1016/j.phymed.2009.02.009
Abdel-Sattar EA, Abdallah HM, Khedr A et al (2013) Antihyperglycemic activity of Caralluma tuberculata in streptozotocin-induced diabetic rats. Food Chem Toxicol 59:111–117. https://doi.org/10.1016/j.fct.2013.05.060
Abdel-Sattar E, EL-Maraghy SA, El-Dine RS, Rizk SM (2017) Antihyperglycemic activity of Caralluma quadrangula in streptozotocin-induced diabetic rats. Bull Faculty Pharm Cairo Univ 55:269–272. https://doi.org/10.1016/j.bfopcu.2017.07.002
Adnan M, Jan S, Mussarat S et al (2014) A review on ethnobotany, phytochemistry and pharmacology of plant genus C. aralluma R. Br J Pharm Pharmacol 66:1351–1368. https://doi.org/10.1111/jphp.12265
Albaser N, Ghanem N, Shehab M et al (2014) Investigation of pharmacological activity of Caralluma penicillata: anti-inflammatory properties and gastritis protection against indomethacin in adult guinea pigs. Int Sch Res Notices 2014:1–9. https://doi.org/10.1155/2014/738493
Al-Fatimi M (2019) Ethnobotanical survey of medicinal plants in central Abyan governorate. Yemen J Ethnopharmacol 241:111973. https://doi.org/10.1016/j.jep.2019.111973
Al-Musayeib NM, Mothana RA, Al-Massarani S et al (2012) Study of the in vitro antiplasmodial, antileishmanial and antitrypanosomal activities of medicinal plants from Saudi Arabia. Molecules 17:11379–11390. https://doi.org/10.3390/molecules171011379
Alonso-Castro AJ, Arana-Argáez V, Yáñez-Barrientos E et al (2021) Pharmacological activities of Asclepias curassavica L. (Apocynaceae) aerial parts. J Ethnopharmacol 281:114554. https://doi.org/10.1016/j.jep.2021.114554
An TY, Run-qiu H, Zhao Y et al (2001) Alkaloids from Cynanchum komarovii with inhibitory activity against the tobacco mosaic virus. Phytochemistry 58:1267–1269. https://doi.org/10.1016/s0031-9422(01)00382-x
Ansari B, Behl T, Pirzada AS, Khan H (2022) Caralluma edulis (Apocynaceae): a comprehensive review on its traditional uses, phytochemical profile and pharmacological effects. Curr Top Med Chem 22:1501–1514. https://doi.org/10.2174/1568026622666220527092825
Araya JJ, Binns F, Kindscher K, Timmermann BN (2012) Verticillosides A-M: polyoxygenated pregnane glycosides from Asclepias verticillata L. Phytochemistry 78:179–189. https://doi.org/10.1016/j.phytochem.2012.02.019
Atanasov AG, Zotchev SB, Dirsch VM, Supuran CT (2021) Natural products in drug discovery: advances and opportunities. Nat Rev Drug Discov 20:200–216. https://doi.org/10.1038/s41573-020-00114-z
Aumeeruddy MZ, Mahomoodally MF (2020) Traditional herbal therapies for hypertension: a systematic review of global ethnobotanical field studies. S Afr J Bot 135:451–464. https://doi.org/10.1016/j.sajb.2020.09.008
Babu K (2014) Evaluation of in vitro antibacterial activity of Caralluma umbellata haw used in traditional medicine by indian tribes. Annu Res Rev Biol 4:840–855. https://doi.org/10.9734/ARRB/2014/6401
Bai H, Li W, Asada Y et al (2009) Twelve pregnane glycosides from Cynanchum atratum. Steroids 74:198–207. https://doi.org/10.1016/j.steroids.2008.10.007
Bais HP, Loyola-Vargas VM, Flores HE, Vivanco JM (2001) Root-specific metabolism: the biology and biochemistry of underground organs. Vitro Cell Dev Biol Plant 37:730–741. https://doi.org/10.1007/s11627-001-0122-y
Bhadane BS, Patil MP, Maheshwari VL, Patil RH (2018) Ethnopharmacology, phytochemistry, and biotechnological advances of family Apocynaceae: a review. Phytother Res 32:1181–1210. https://doi.org/10.1002/ptr.6066
Cavalcanti ABS, Costa Barros RP, de Oliveira Costa VC et al (2019) Computer-aided chemotaxonomy and bioprospecting study of diterpenes of the Lamiaceae family. Molecules. https://doi.org/10.3390/molecules24213908
Cech NB, Medema MH, Clardy J (2021) Benefiting from big data in natural products: importance of preserving foundational skills and prioritizing data quality. Nat Prod Rep 38:1947–1953. https://doi.org/10.1039/d1np00061f
Chen J, Ye Y, Sun C, Pan Y (2008) Rapid identification of oleanane-type saponins in the roots of Stephanotis mucronata by liquid chromatography/electrospray tandem mass spectrometry. Anal Chim Acta 613:74–82. https://doi.org/10.1016/j.aca.2008.02.052
Chen FY, Yi-Ping Y, Hong-Xiang S et al (2009) Stemucronatoside L, a pregnane glycoside from the roots of Stephanotis mucronata, inhibits Th1/Th2 immune responses in vitro. Chem Biodivers 6:916–923. https://doi.org/10.1002/cbdv.200800159
Chen F, Ni Y, Ye Y et al (2010) Comparison of immunosuppressive activity of Stephanoside E and its aglycone from Stephanotis mucronata in vitro. Int Immunopharmacol 10:1153–1160. https://doi.org/10.1016/j.intimp.2010.06.021
Chen W, Zhang Z, Ban Y et al (2019) Cynanchum bungei Decne and its two related species for “Baishouwu”: a review on traditional uses, phytochemistry, and pharmacological activities. J Ethnopharmacol 243:112110. https://doi.org/10.1016/j.jep.2019.112110
Chen J, Cheng C, Chen J et al (2020) Cynanchum paniculatum and its major active constituents for inflammatory-related diseases: a review of traditional use, multiple pathway modulations, and clinical applications. Evid Based Complement Altern Med 2020:1–16. https://doi.org/10.1155/2020/7259686
Chen W-Y, Yang T, Yang J et al (2021) Wild plants used by the Lhoba people in Douyu Village, characterized by high mountains and valleys, in southeastern Tibet, China. J Ethnobiol Ethnomed 17:46. https://doi.org/10.1186/s13002-021-00472-x
Chen X, Luo Z, Liu X et al (2022) Marsdenia tenacissima (Roxb) Moon injection exerts a potential anti-tumor effect in prostate cancer through inhibiting ErbB2-GSK3β-HIF1α signaling axis. J Ethnopharmacol 295:115381. https://doi.org/10.1016/j.jep.2022.115381
Chinedu E, Ehiabhi OS (2023) Ethnomedicinal, phytochemical, and pharmacological review of asclepiadaceae. J Prevent Diagnos Treat Strategies Med 2:3–18
Correia MV, Scotti MT, Ferreira MJP et al (2008) Self-organizing maps as a new tool for classification of plants at lower hierarchical levels. Nat Prod Commun. https://doi.org/10.1177/1934578X0800301029
De Bruyn A, Martin DP, Lefeuvre P (2014) Phylogenetic reconstruction methods: an overview. Methods Mol Biol 1115:257–277. https://doi.org/10.1007/978-1-62703-767-9_13
Deleo M, Detommasi N, Sanogo R et al (2005) New pregnane glycosides from Caralluma dalzielii. Steroids 70:573–585. https://doi.org/10.1016/j.steroids.2005.03.013
Dong J, Peng X, Li L et al (2018) C21 steroidal glycosides with cytotoxic activities from Cynanchum otophyllum. Bioorg Med Chem Lett 28:1520–1524. https://doi.org/10.1016/j.bmcl.2018.03.078
Dong J, Yue GG-L, Lee JK-M et al (2020) Potential neurotrophic activity and cytotoxicity of selected C21 steroidal glycosides from Cynanchum otophyllum. Med Chem Res 29:549–555. https://doi.org/10.1007/s00044-020-02506-7
Dzobo K (2022) The Role of Natural Products as Sources of Therapeutic Agents for Innovative Drug Discovery. Comprehensive Pharmacology. Elsevier, pp 408–422
El Sayed AM, AbdElSattar E, Khalil MN (2020) New calogenin pregnane glycoside derivative from Huernia Saudi-Arabica and its Lipase and α-glucosidase inhibitory activities. Biomed Pharmacother 127:110143. https://doi.org/10.1016/j.biopha.2020.110143
El-Aziz MMA, Ashour AS, Melad ASG (2019) A review on saponins from medicinal plants: chemistry, isolation, and determination. J Nanomed Res 8:6–12. https://doi.org/10.15406/jnmr.2019.08.00199
El-Fitiany RA, Khasawneh MA (2022) Leptadenia pyrotechnica (Forsk.) Decne: from edibility to drug discovery (a comprehensive review). Food Rev Int. https://doi.org/10.1080/87559129.2022.2126853
El-Shiekh RA, Hassan M, Hashem RA, Abdel-Sattar E (2021) Bioguided isolation of antibiofilm and antibacterial pregnane glycosides from Caralluma quadrangula: disarming multidrug-resistant pathogens. Antibiotics 10:811. https://doi.org/10.3390/antibiotics10070811
Fan Y, Jin J, Jian J et al (2022) Three new pregnanes isolated from the Cynanchum auriculatum. Chem Biodivers. https://doi.org/10.1002/cbdv.202200243
Félix-Silva J, Silva-Junior AA, Zucolotto SM, de Fernandes-Pedrosa MF (2017) Medicinal plants for the treatment of local tissue damage induced by snake venoms: an overview from traditional use to pharmacological evidence. Evid Based Complement Altern Med 2017:1–52. https://doi.org/10.1155/2017/5748256
Fierascu RC, Fierascu I, Ortan A et al (2020) Innovative approaches for recovery of phytoconstituents from medicinal/aromatic plants and biotechnological production. Molecules 25:309
Fillbrunn A, Dietz C, Pfeuffer J et al (2017) KNIME for reproducible cross-domain analysis of life science data. J Biotechnol 261:149–156. https://doi.org/10.1016/j.jbiotec.2017.07.028
Fishbein M, Livshultz T, Straub SCK et al (2018) Evolution on the backbone: Apocynaceae phylogenomics and new perspectives on growth forms, flowers, and fruits. Am J Bot 105:495–513. https://doi.org/10.1002/ajb2.1067
Franco JR, Cecchi G, Paone M et al (2022) The elimination of human African trypanosomiasis: achievements in relation to WHO road map targets for 2020. PLoS Negl Trop Dis 16:e0010047. https://doi.org/10.1371/journal.pntd.0010047
Ghazanfar SA (2018) Edible wild plants: a case study from oman. Global perspectives on underutilized crops. Springer, Cham, pp 207–216. https://doi.org/10.1007/978-3-319-77776-4_8
Gu X-J, Hao D-C (2016) Recent advances in phytochemistry and pharmacology of C21 steroid constituents from Cynanchum plants. Chin J Nat Med 14:0321–0334. https://doi.org/10.3724/SP.J.1009.2016.00321
Gupta VS, Kumar A, Deepak D et al (2003) Pregnanes and pregnane glycosides from Marsdenia roylei. Phytochemistry 64:1327–1333. https://doi.org/10.1016/S0031-9422(03)00468-0
Gyebi GA, Adebayo JO, Olorundare OE et al (2018) Iloneoside: a cytotoxic ditigloylated pregnane glycoside from the leaves of Gongronema latifolium Benth. Nat Prod Res 32:2882–2886. https://doi.org/10.1080/14786419.2017.1385019
Habibuddin M, Daghriri HA, Humaira T et al (2008) Antidiabetic effect of alcoholic extract of Caralluma sinaica L. on streptozotocin-induced diabetic rabbits. J Ethnopharmacol 117:215–220. https://doi.org/10.1016/j.jep.2008.01.021
Haider SI, Asif A, Rasheed HMF et al (2022) Caralluma tuberculata exhibits analgesic and anti-arthritic potential by downregulating pro-inflammatory cytokines and attenuating oxidative stress. Inflammopharmacology 30:621–638. https://doi.org/10.1007/s10787-022-00949-5
Hamed AI (2001) New steroids from Solenostemma argel leaves. Fitoterapia 72:747–755. https://doi.org/10.1016/S0367-326X(01)00308-2
Han L, Zhou X, Yang M et al (2018) Ethnobotany, phytochemistry and pharmacological effects of plants in genus Cynanchum Linn. (Asclepiadaceae). Molecules 23:1194. https://doi.org/10.3390/molecules23051194
Herrera RM, Pérez M, Martín-Herrera DA et al (1996) Antimicrobial activity of extracts from plants endemic to the canary islands. Phytother Res 10:364–366. https://doi.org/10.1002/(SICI)1099-1573(199606)10:4%3c364::AID-PTR846%3e3.0.CO;2-B
Hu Y-J, Shen X-L, Lu H-L et al (2008) Tenacigenin B derivatives reverse P-glycoprotein-mediated multidrug resistance inHepG2/Dox cells. J Nat Prod 71:1049–1051. https://doi.org/10.1021/np070458f
Huan VD, OhtaniKasai KR et al (2001) Sweet pregnane glycosides from Telosma procumbens. Chem Pharm Bull (tokyo) 49:453–460. https://doi.org/10.1248/cpb.49.453
Huang L-J, Wang B, Zhang J et al (2015) Studies on cytotoxic pregnane sapogenins from Cynanchum wilfordii. Fitoterapia 101:107–116. https://doi.org/10.1016/j.fitote.2014.10.014
Indrayanto G (2022) The importance of method validation in herbal drug research. J Pharm Biomed Anal 214:114735. https://doi.org/10.1016/j.jpba.2022.114735
Jayawardena R, Francis TV, Abhayaratna S, Ranasinghe P (2021) The use of Caralluma fimbriata as an appetite suppressant and weight loss supplement: a systematic review and meta-analysis of clinical trials. BMC Complement Med Ther 21:279. https://doi.org/10.1186/s12906-021-03450-8
Ji C-X, Li X-Y, Jia S-B et al (2012) The antidepressant effect of Cynanchum auriculatum in mice. Pharm Biol 50:1067–1072. https://doi.org/10.3109/13880209.2012.656848
Jia S, Song J, Dai R et al (2017) Structural deduction of pregnane glycosides from Dregea sinensis Hemsl by high-performance liquid chromatography and electrospray ionization-tandem mass spectrometry. Int J Mass Spectrom 415:38–43. https://doi.org/10.1016/j.ijms.2017.01.018
Jia X-N, Wang W-J, Yin B et al (2022) Deep learning promotes the screening of natural products with potential microtubule inhibition activity. ACS Omega 7:28334–28341. https://doi.org/10.1021/acsomega.2c02854
Jiang H-W, Gu S-S, Cao L et al (2019) Potential hypoglycemic effect of acetophenones from the root bark of Cynanchum wilfordii. Nat Prod Res 33:2314–2321. https://doi.org/10.1080/14786419.2018.1443100
Jin Q, Han XH, Yun C-Y et al (2018) Melanogenesis inhibitory pregnane glycosides from Cynanchum atratum. Bioorg Med Chem Lett 28:1252–1256. https://doi.org/10.1016/j.bmcl.2018.01.004
Jin-Ian Y, Wei-pei D, Jian-ping S et al (1991) Studies on the antifertility components from Marsdenia koi. J Tongji Med Univ 11:165–168. https://doi.org/10.1007/BF02888129
Kahksha AO, Naaz S et al (2022) Recent developments made in the assessment of the antidiabetic potential of gymnema species–from 2016 to 2020. J Ethnopharmacol 286:114908. https://doi.org/10.1016/j.jep.2021.114908
Kamble MY, Mane SS, Murugan C, Jaisankar I (2008) Diversity of Ethno-Medicinal Plants of Tropical Islands–with Special Reference to Andaman and Nicobar Islands. Biodiversity and Climate Change Adaptation in Tropical Islands. Elsevier, pp 55–103. https://doi.org/10.1016/B978-0-12-813064-3.00003-X
Kamel MS (2003) Acylated phenolic glycosides from Solenostemma argel. Phytochemistry 62:1247–1250. https://doi.org/10.1016/S0031-9422(03)00022-0
Kanakhara R, Rudrappa H, Shukla V, Acharya R (2018) Detail pharmacognostical and analytical profile of Telosma pallida (l.) kurz. (leaf): a folklore medicinal plant of Gujarat State. Anc Sci Life 37:120. https://doi.org/10.4103/asl.ASL_97_17
Katoh K (2002) MAFFT: a novel method for rapid multiple sequence alignment based on fast Fourier transform. Nucl Acids Res 30:3059–3066. https://doi.org/10.1093/nar/gkf436
Kell G, Rao A, Katsikitis M (2019) A randomised placebo controlled clinical trial on the efficacy of Caralluma fimbriata supplement for reducing anxiety and stress in healthy adults over eight weeks. J Affect Disord 246:619–626. https://doi.org/10.1016/j.jad.2018.12.062
Khan F, Sarker MdMR, Ming LC et al (2019) Comprehensive review on phytochemicals pharmacological and clinical potentials of Gymnema sylvestre. Front Pharmacol. https://doi.org/10.3389/fphar.2019.01223
Kim M, Baek J, Park J et al (2005) Wilfoside K1N isolated from Cynanchum wilfordii inhibits angiogenesis and tumor cell invasion. Int J Oncol. https://doi.org/10.3892/ijo.26.6.1533
Kommidi DR, Koorbanally NA, Moodley B (2016) Lung cancer activity of Caralluma species, an overview. Austin J Lung Cancer Res 1:1–5
Lai C-Z, Liu J-X, Pang S-W et al (2016) Steroidal glycosides from the roots of Cynanchum stauntonii and their effects on the expression of iNOS and COX-2. Phytochem Lett 16:38–46. https://doi.org/10.1016/j.phytol.2016.02.016
Lee KY, Yoon JS, Kim ES et al (2005) Anti-acetylcholinesterase and anti-amnesic activities of a pregnane glycoside, Cynatroside B, from Cynanchum atratum. Planta Med 71:7–11. https://doi.org/10.1055/s-2005-837743
Li X, Sun H, Ye Y et al (2005) Three New Immunomodulating C21-steroidal glycosides from the stems of Stephanotis mucronata. Chem Biodivers 2:1701–1711. https://doi.org/10.1002/cbdv.200590140
Li X, Zhang M, Xiang C et al (2015) Antiepileptic C 21 steroids from the roots of Cynanchum otophyllum. J Asian Nat Prod Res 17:724–732. https://doi.org/10.1080/10286020.2014.1001380
Li J-L, Gao Z-B, Zhao W-M (2016a) Identification and evaluation of antiepileptic activity of C 21 steroidal glycosides from the roots of Cynanchum wilfordii. J Nat Prod 79:89–97. https://doi.org/10.1021/acs.jnatprod.5b00766
Li X, Luo Y, Li G-P, Yang Q-X (2016b) Pregnane glycosides from the antidepressant active fraction of cultivated Cynanchum otophyllum. Fitoterapia 110:96–102. https://doi.org/10.1016/j.fitote.2016.02.014
Li S, Pei W, Yuan W et al (2022) Multi-omics joint analysis reveals the mechanism of action of the traditional Chinese medicine Marsdenia tenacissima (Roxb.) Moon in the treatment of hepatocellular carcinoma. J Ethnopharmacol 293:115285. https://doi.org/10.1016/j.jep.2022.115285
Li X, Liang H, Wu J et al (2023a) Investigation of herbal plant medicines Baishouwu on the mechanism of the digestion of body: a review. J Funct Foods. https://doi.org/10.1016/j.jff.2022.105379
Li X, Zhang JJ, Li YH, Yang QX (2023b) Cynanotophyllosides E-F, two minor pregnane glycosides from the roots of cultivated Cynanchum otophyllum. J Asian Nat Prod Res. https://doi.org/10.1080/10286020.2022.2158085
Li Y, Yang H, Li Z et al (2023c) Advances in the biosynthesis and molecular evolution of steroidal saponins in plants. Int J Mol Sci. https://doi.org/10.3390/ijms24032620
Libman A, Zhang H, Ma C et al (2008) A first new antimalarial pregnane glycoside from Gongronema napalense. Asian J Tradit Med 3:203–210
Lin J, Ruan J, Zhu H et al (2021) Tenacissoside H Induces autophagy and radiosensitivity of hepatocellular carcinoma cells by PI3K/Akt/mTOR signaling pathway. Dose-Response 19:155932582110110. https://doi.org/10.1177/15593258211011023
Lindemann P (2015) Steroidogenesis in plants–biosynthesis and conversions of progesterone and other pregnane derivatives. Steroids 103:145–152. https://doi.org/10.1016/j.steroids.2015.08.007
Lins FSV, de Souza TA, Opretzka LCF et al (2022) New pregnane glycosides from Mandevilla dardanoi and their anti-inflammatory activity. Molecules. https://doi.org/10.3390/molecules27185992
Liu Y, Li J, Yu S et al (2008a) Rapid structural determination of modified pregnane glycosides from Cynanchum forrestii by liquid chromatography–diode-array detection/electrospray ionization multi-stage tandem mass spectrometry. Anal Chim Acta 611:187–196. https://doi.org/10.1016/j.aca.2008.01.076
Liu Y, Qu J, Yu S et al (2008b) Nine novel C-21 steroidal glycosides substituted with orthoacetate from Dregea sinensis var. corrugata. Steroids 73:184–192. https://doi.org/10.1016/j.steroids.2007.09.009
Liu K, Chen F, Zhang H (2010) Antitumor effects by wilfoside C3N treatment in ECA109 cells. Anticancer Drugs 21:625–631. https://doi.org/10.1097/CAD.0b013e328339301c
Liu S, Chen Z, Wu J et al (2013) Appetite suppressing pregnane glycosides from the roots of Cynanchum auriculatum. Phytochemistry 93:144–153. https://doi.org/10.1016/j.phytochem.2013.03.010
Liu P, Xu D-W, Li R-T et al (2021) A combined phytochemistry and network pharmacology approach to reveal potential anti-NSCLC effective substances and mechanisms in Marsdenia tenacissima (Roxb.) moon (Stem). Front Pharmacol. https://doi.org/10.3389/fphar.2021.518406
Lötsch J, Ultsch A (2014) Exploiting the structures of the U-matrix. Springer, pp 249–257
Makeen HA, Menachery SJ, Moni SS et al (2020) Documentation of bioactive principles of the exudate gel (EG) from the stem of Caralluma retrospiciens (Ehrenb) and in vitro antibacterial activity–part A. Arab J Chem 13:6672–6681. https://doi.org/10.1016/j.arabjc.2020.06.022
Mauri A (2020) alvaDesc: a tool to calculate and analyze molecular descriptors and fingerprints. Springer, pp 801–820
Mautone M, De Martino L, De Feo V (2019) Ethnobotanical research in Cava de’ Tirreni area. Southern Italy J Ethnobiol Ethnomed 15:50. https://doi.org/10.1186/s13002-019-0330-3
McGarvey BD, Liao H, Ding K, Wang X (2012) Dereplication of known pregnane glycosides and structural characterization of novel pregnanes in Marsdenia tenacissima by high-performance liquid chromatography and electrospray ionization-tandem mass spectrometry. J Mass Spectrom 47:687–693. https://doi.org/10.1002/jms.2991
Menezes RPB, Sessions Z, Muratov E et al (2021) Secondary metabolites extracted from Annonaceae and chemotaxonomy study of terpenoids. J Braz Chem Soc 32:2061–2070
Mohanty SK, Swamy MK, Sinniah UR, Anuradha M (2017) Leptadenia reticulata (Retz.) Wight & Arn (Jivanti): botanical, agronomical, phytochemical, pharmacological, and biotechnological aspects. Molecules 22:1019. https://doi.org/10.3390/molecules22061019
Mohd Nasuha NA, Choo Y-M (2016) A new flavone from Malaysia Borneo Marsdenia tinctoria. Nat Prod Res 30:1532–1536. https://doi.org/10.1080/14786419.2015.1118629
Morebise O (2015) A review on Gongronema latifolium, an extremely useful plant with great prospects. Eur J Med Plants 10:1–9. https://doi.org/10.9734/EJMP/2015/19713
Moses T, Papadopoulou KK, Osbourn A (2014) Metabolic and functional diversity of saponins, biosynthetic intermediates aDnd semi-synthetic derivatives. Crit Rev Biochem Mol Biol 49:439–462. https://doi.org/10.3109/10409238.2014.953628
Mu Q, Shen Y, Zhou Q et al (1992) Studies on the constituents of Adelostemma gracillimum. Planta Med 58:200–204. https://doi.org/10.1055/s-2006-961429
Muyumba NW, Mutombo SC, Sheridan H et al (2021) Quality control of herbal drugs and preparations: the methods of analysis, their relevance and applications. Talanta Open. https://doi.org/10.1016/j.talo.2021.100070
Nam NB, Trieu LN, Vu NT et al (2022) Micropropagation of Jasminanthes tuyetanhiae: an endemic and valuable herb in Vietnam. Plant Cell Tissue Organ Culture (PCTOC) 148:35–44. https://doi.org/10.1007/s11240-021-02158-4
Nazar N, Goyder DJ, Clarkson JJ et al (2013) The taxonomy and systematics of Apocynaceae: where we stand in 2012. Bot J Linn Soc 171:482–490. https://doi.org/10.1111/boj.12005
Nazia N, Clarkson JJ, David G et al (2019) Phylogenetic relationships in Apocynaceae based on nuclear PHYA and plastid trnL-F sequences, with a focus on tribal relationships. Caryologia 72:55–81
Odago WO, Waswa EN, Nanjala C et al (2022) Analysis of the complete plastomes of 31 species of hoya group: insights into their comparative genomics and phylogenetic relationships. Front Plant Sci. https://doi.org/10.3389/fpls.2021.814833
Ojo OA, Okesola MA, Ekakitie LI et al (2020) Gongronema latifolium Benth. leaf extract attenuates diabetes-induced neuropathy via inhibition of cognitive, oxidative stress and inflammatory response. J Sci Food Agric 100:4504–4511. https://doi.org/10.1002/jsfa.10491
Oliveira T, Silva M, Maia E et al (2023) Virtual screening algorithms in drug discovery: a review focused on machine and deep learning methods. Drugs Drug Candidates 2:311–334. https://doi.org/10.3390/ddc2020017
Ounaissia K, Pertuit D, Mitaine-Offer A-C et al (2016) New pregnane and phenolic glycosides from Solenostemma argel. Fitoterapia 114:98–104. https://doi.org/10.1016/j.fitote.2016.08.002
Pandey A, Swarnkar V, Pandey T et al (2016) Transcriptome and metabolite analysis reveal candidate genes of the cardiac glycoside biosynthetic pathway from Calotropis procera. Sci Rep. https://doi.org/10.1038/srep34464
Pandya D, Anand I (2011) A complete review on Oxystelma esculentum R. Br Pharmacogn J 3:87–90. https://doi.org/10.5530/pj.2011.19.16
Patel VK, Shirbhate E, Tiwari P et al (2022) Multi-targeted HDAC inhibitors as anticancer agents: current status and future prospective. Curr Med Chem 30:2762–2795. https://doi.org/10.2174/0929867329666220922105615
Payab M, Hasani-Ranjbar S, Shahbal N et al (2020) Effect of the herbal medicines in obesity and metabolic syndrome: a systematic review and meta-analysis of clinical trials. Phytother Res 34:526–545. https://doi.org/10.1002/ptr.6547
Plaza A, Perrone A, Balestrieri C et al (2005) New antiproliferative 14,15-secopregnane glycosides from Solenostemma argel. Tetrahedron 61:7470–7480. https://doi.org/10.1016/j.tet.2005.05.048
Prihoda D, Maritz JM, Klempir O et al (2021) The application potential of machine learning and genomics for understanding natural product diversity, chemistry, and therapeutic translatability. Nat Prod Rep 38:1100–1108. https://doi.org/10.1039/D0NP00055H
Qian X, Li B, Li P et al (2017) C21 steroidal glycosides from Cynanchum auriculatum and their neuroprotective effects against H2O2-induced damage in PC12 cells. Phytochemistry 140:1–15. https://doi.org/10.1016/j.phytochem.2017.04.014
Qin J-J, Chen X, Lin Z-M et al (2018) C 21 -steroidal glycosides and sesquiterpenes from the roots of Cynanchum bungei and their inhibitory activities against the proliferation of B and T lymphocytes. Fitoterapia 124:193–199. https://doi.org/10.1016/j.fitote.2017.11.014
Raees MA, Hussain H, Al-Rawahi A et al (2016) Anti-proliferative and computational studies of two new pregnane glycosides from Desmidorchis flava. Bioorg Chem 67:95–104. https://doi.org/10.1016/j.bioorg.2016.05.008
Ramesh M, Nageshwar Rao Y, Kumar MR et al (1999) Antinociceptive and anti-inflammatory activity of carumbelloside-I isolated from Caralluma umbellata. J Ethnopharmacol 68:349–352. https://doi.org/10.1016/S0378-8741(99)00122-1
Ramos YJ, Gouvêa-Silva JG, de Brito MD et al (2023) Chemophenetic and chemodiversity approaches: new insights on modern study of plant secondary metabolite diversity at different spatiotemporal and organizational scales. Rev Bras 33:49–72
Rapini A (2012) Taxonomy ‘under construction’: advances inthe systematics of Apocynaceae, with emphasis on the Brazilian Asclepiadoideae. Rodriguésia 63:75–88. https://doi.org/10.1590/S2175-78602012000100007
Ronquist F, Huelsenbeck JP (2003) MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics 19:1572–1574. https://doi.org/10.1093/bioinformatics/btg180
Saiki P, Kawano Y, Ogi T et al (2020) Purified gymnemic acids from Gymnema inodorum tea inhibit 3T3-L1 cell differentiation into adipocytes. Nutrients 12:2851. https://doi.org/10.3390/nu12092851
Saldívar-González FI, Aldas-Bulos VD, Medina-Franco JL, Plisson F (2022) Natural product drug discovery in the artificial intelligence era. Chem Sci 13:1526–1546. https://doi.org/10.1039/D1SC04471K
Sánchez-Camargo AP, Montero L, Mendiola JA, Herrero M, Ibáñez E (2020) Novel extraction techniques for bioactive compounds from herbs and spices. Herbs Spices Med Plants Process Health Benefits Saf. https://doi.org/10.1002/9781119036685.ch5
Scotti MT, Emerenciano V, Ferreira MJP et al (2012) Self-organizing maps of molecular descriptors for sesquiterpene lactones and their application to the chemotaxonomy of the Asteraceae family. Molecules 17:4684–4702. https://doi.org/10.3390/molecules17044684
Seeka C, Prabpai S, Kongsaeree P et al (2017) Anti-inflammatory 12,20-epoxypregnane and 11,12- seco -pregnane glycosides from the stems of Hoya kerrii. J Nat Prod 80:1714–1724. https://doi.org/10.1021/acs.jnatprod.6b00730
Sethi A, Paswan S, Srivastava S et al (2008) A new pregnane glycoside from Marsdenia roylei as potential antioxidant and antidyslipidemic agents. J Asian Nat Prod Res 10:1023–1028. https://doi.org/10.1080/10286020802280190
Sharma P, Sarma J, Sarma CM (2019) Ethnobotany and micropropagation of Asclepias curassavica L. (Apocynaceae)–a medicinally potent naturalized plant in India. Pleione 13:227. https://doi.org/10.26679/Pleione.13.2.2019.227-235
Sheng F, Mengting C, Yuan T et al (2016) Protective effects of otophylloside n on pentylenetetrazol-induced neuronal injury in vitro and in vivo. Front Pharmacol 7:224. https://doi.org/10.3389/fphar.2016.00224
Si Y, Sha XS, Shi LL et al (2022) Review on pregnane glycosides and their biological activities. Phytochem Lett 47:1–17. https://doi.org/10.1016/j.phytol.2021.10.007
Silva JPR, Acevedo CAH, de Souza TA et al (2021) Natural products as potential agents against SARS-CoV and SARSCoV-2. Curr Med Chem 28:5498–5526. https://doi.org/10.2174/0929867328666210125113938
Sireesha M, Venkata Nadh R, Suresh Babu K, Sreenivasulu M (2018) Phytochemical library of Caralluma genus. Int J Res Pharmaceut Sci 9:1201–1213
Sireesha M, Ratnakaram VN, Suresh Babu K, Sreenivasulu M (2019) Pharmacological review of Caralluma r.br: a potential herbal genus. Asian J Pharm 12:1146–1158
Slapšytė G, Dedonytė V, Adomėnienė A et al (2019) Genotoxic properties of Betonica officinalis, Gratiola officinalis, Vincetoxicum luteum and Vincetoxicum hirundinaria extracts. Food Chem Toxicol 134:110815. https://doi.org/10.1016/j.fct.2019.110815
Smith C, Krygsman A (2014) Hoodia gordonii: to eat, or not to eat. J Ethnopharmacol 155:987–991. https://doi.org/10.1016/j.jep.2014.06.033
Sobiecki JF (2002) A preliminary inventory of plants used for psychoactive purposes in southern African healing traditions. Trans R Soc South Afr 57:1–24. https://doi.org/10.1080/00359190209520523
Stafford GI, Pedersen ME, van Staden J, Jäger AK (2008) Review on plants with CNS-effects used in traditional South African medicine against mental diseases. J Ethnopharmacol 119:513–537. https://doi.org/10.1016/j.jep.2008.08.010
Sutcharitchan C, Wang L, Zhang H et al (2018) Pharmacognostical study of Cynanchum stauntonii and Cynanchum glaucescens, botanical sources of TCM Baiqian. Chin Herb Med 10:379–387. https://doi.org/10.1016/j.chmed.2018.08.001
Tai Y, Cao X, Li X, Pan Y (2006) Identification of C-21 steroidal glycosides from the roots of Cynanchum chekiangense by high-performance liquid chromatography/tandem mass spectrometry. Anal Chim Acta 572:230–236. https://doi.org/10.1016/j.aca.2006.05.044
Tambe DA, Chaudhari TB, Chaudhari SR (2010) Phyto-pharmacology of Caralluma adscendens Roxb: a review. Pharmacogn J 2:33–38. https://doi.org/10.1016/S0975-3575(10)80069-0
Tarkowská D (2019) Plants are capable of synthesizing animal steroid hormones. Molecules 24:2585. https://doi.org/10.3390/molecules24142585
Tatsuno S, Yokosuka A, Hatsuma F et al (2019) Pregnane glycosides from the bark of Marsdenia cundurango and their cytotoxic activity. J Nat Med 73:93–103. https://doi.org/10.1007/s11418-018-1248-0
The Japonese Pharmacopeia XVII (2016) THE MINISTRY OF HEALTH LAW. Tokyo
Thuy NTK, Phuong PT, Mai NT et al (2020) Pregnane glycosides from Dregea volubilis and their α-glucosidase inhibitory activity. Phytochem Lett 37:90–94. https://doi.org/10.1016/j.phytol.2020.04.013
Thuy NTK, Phuong PT, Hien NTT et al (2021) Pregnane glycosides from the leaves of Dregea volubilis and their α-glucosidase and α-amylase inhibitory activities. Nat Prod Res 35:3931–3938. https://doi.org/10.1080/14786419.2020.1749615
To KKW, Wu X, Yin C et al (2017) Reversal of multidrug resistance by Marsdenia tenacissima and its main active ingredients polyoxypregnanes. J Ethnopharmacol 203:110–119. https://doi.org/10.1016/j.jep.2017.03.051
Trang DT, Yen DTH, Cuong NT et al (2021) Pregnane glycosides from Gymnema inodorum and their α-glucosidase inhibitory activity. Nat Prod Res 35:2157–2163. https://doi.org/10.1080/14786419.2019.1663517
Ugwah-Oguejiofor CJ, Abubakar K, Ugwah MO, Njan AA (2013) Evaluation of the antinociceptive and anti-inflammatory effect of Caralluma dalzielii. J Ethnopharmacol 150:967–972. https://doi.org/10.1016/j.jep.2013.09.049
Ullah R, Alqahtani AS, Noman OMA et al (2020) A review on ethno-medicinal plants used in traditional medicine in the Kingdom of Saudi Arabia. Saudi J Biol Sci 27:2706–2718. https://doi.org/10.1016/j.sjbs.2020.06.020
Upadhyay S, Jeena GS, Shikha SRK (2018) Recent advances in steroidal saponins biosynthesis and in vitro production. Planta 248:519–544. https://doi.org/10.1007/s00425-018-2911-0
Vermaak I, Mulaudzi N, Fouche G (2023) Hoodia gordonii. The South African herbal pharmacopoeia. Elsevier, pp 271–291
Vesanto J, Alhoniemi E (2000) Clustering of the self-organizing map. IEEE Trans Neural Netw 11:586–600. https://doi.org/10.1109/72.846731
Viljoen A, Chen W, Mulaudzi N et al (2022) Hoodia gordonii. Phytochemical profiling of commercially important South African plants. Elsevier, pp 129–135
Wainwright CL, Teixeira MM, Adelson DL et al (2022) Future directions for the discovery of natural product-derived immunomodulating drugs: an IUPHAR positional review. Pharmacol Res 177:106076. https://doi.org/10.1016/j.phrs.2022.106076
Wang H-G, Ye Y-P (2005) Haemolytic activities and adjuvant effect of stemucronatoside A on the immune responses to ovalbumin in mice. Zhongguo Tianran Yaowu 3:382–386
Wang Y-B, Su S-S, Chen S-F et al (2017) C 21 steroidal glycosides with cytotoxic activity from Cynanchum taihangense. Phytochem Lett 20:218–223. https://doi.org/10.1016/j.phytol.2017.05.002
Wang K, Liu W, Xu Q et al (2021) Tenacissoside G synergistically potentiates inhibitory effects of 5-fluorouracil to human colorectal cancer. Phytomedicine 86:153553. https://doi.org/10.1016/j.phymed.2021.153553
Wang Y, Zhang C-F, Ochieng Odago W et al (2023) Evolution of 101 Apocynaceae plastomes and phylogenetic implications. Mol Phylogenet Evol 180:107688. https://doi.org/10.1016/j.ympev.2022.107688
Wu X, Yin C, Ma J et al (2021) Polyoxypregnanes as safe, potent, and specific ABCB1-inhibitory pro-drugs to overcome multidrug resistance in cancer chemotherapy in vitro and in vivo. Acta Pharm Sin B 11:1885–1902. https://doi.org/10.1016/j.apsb.2020.12.021
Wynberg R (2023) Biopiracy: crying wolf or a lever for equity and conservation? Res Policy. https://doi.org/10.1016/j.respol.2022.104674
Xiang L, Mi Z, Cheng X et al (2015) Antiepileptic C21 steroids from the roots of Cynanchum otophyllum. J Asian Nat Prod Res 17:724–732. https://doi.org/10.1080/10286020.2014.1001380
Xie F, Dong H, Zhang H (2021) regulatory functions of protein tyrosine phosphatase receptor type O in immune cells. Front Immunol. https://doi.org/10.3389/fimmu.2021.783370
Yan Y, Zhang J, Liu K et al (2014) Seco-pregnane steroidal glycosides from the roots of Cynanchum atratum and their anti-TMV activity. Fitoterapia 97:50–63. https://doi.org/10.1016/j.fitote.2014.03.027
Yan Y, Tang P, Zhang X et al (2022) Anti-TMV effects of seco-pregnane C21 steroidal glycosides isolated from the roots of Cynanchum paniculatum. Fitoterapia 161:105225. https://doi.org/10.1016/j.fitote.2022.105225
Yang Q-X, Ge Y-C, Huang X-Y, Sun Q-Y (2011) Cynanauriculoside C-E, three new antidepressant pregnane glycosides from Cynanchum auriculatum. Phytochem Lett 4:170–175. https://doi.org/10.1016/j.phytol.2011.02.009
Yang J, Wan Q-L, Mu Q-Z et al (2015) The lifespan-promoting effect of otophylloside B in Caenorhabditis elegans. Nat Prod Bioprospect 5:177–183. https://doi.org/10.1007/s13659-015-0064-4
Yang J, Huang X-B, Wan Q-L et al (2017) Otophylloside B protects against Aβ toxicity in Caenorhabditis elegans models of Alzheimer’s disease. Nat Prod Bioprospect 7:207–214. https://doi.org/10.1007/s13659-017-0122-1
Ye H, Li C, Ye W et al (2022) Medicinal angiosperms of asclepiadaceae and periplocaceae. Common Chinese materia medica. Springer, Singapore, pp 539–598. https://doi.org/10.1007/978-981-16-5892-1_10
Yoon M-Y, Choi NH, Min BS et al (2011) Potent in vivo antifungal activity against powdery mildews of pregnane glycosides from the roots of Cynanchum wilfordii. J Agric Food Chem 59:12210–12216. https://doi.org/10.1021/jf2039185
Yoshikawa K, Ogata H, Arihara S et al (1998) Antisweet natural products. XIII. structures of alternosides I-X from Gymnema alternifolium. Chem Pharm Bull (tokyo) 46:1102–1107. https://doi.org/10.1248/cpb.46.1102
Yoshimura S, Narita H, Hayashi K, Mitsuhashi H (1983) Studies on the constituents of asclepiadaceae plants. LVI. Isolation of new antitumor-active glycosides from Dregea volubilis (L.) Benth. Chem Pharm Bull (tokyo) 31:3971–3983. https://doi.org/10.1248/cpb.31.3971
Yue GGL, Chan KM, To MH et al (2014) Potent airway smooth muscle relaxant effect of cynatratoside B, a steroidal glycoside isolated from Cynanchum stauntonii. J Nat Prod 77:1074–1077. https://doi.org/10.1021/np4008969
Yulin R, Bethany GE, Joshua MH et al (2021) Bioactive small-molecule constituents of Lao plants. J Med Plants Res 15:540–559. https://doi.org/10.5897/JMPR2021.7137
Zakaria MNM, Islam MW, Radhakrishnan R et al (2001) Anti-nociceptive and anti-inflammatory properties of Caralluma arabica. J Ethnopharmacol 76:155–158. https://doi.org/10.1016/S0378-8741(01)00208-2
Zhang M, Li X, Xiang C et al (2015a) Cytotoxicity of pregnane glycosides of Cynanchum otophyllum. Steroids 104:49–60. https://doi.org/10.1016/j.steroids.2015.08.010
Zhang M, Rao L-L, Xiang C et al (2015b) C 21 steroidal glycosides from the roots of Cynanchum saccatum. Steroids 101:28–36. https://doi.org/10.1016/j.steroids.2015.05.009
Zhang R, Liu C, Li Y et al (2021) Tenacissoside H promotes neurological recovery of cerebral ischaemia/reperfusion injury in mice by modulating inflammation and oxidative stress via TrkB pathway. Clin Exp Pharmacol Physiol 48:757–769. https://doi.org/10.1111/1440-1681.13398
Zhang X, Gao M, Rao Z et al (2022a) The antitumour activity of C21 steroidal glycosides and their derivatives of Baishouwu: a review. J Ethnopharmacol. https://doi.org/10.1016/j.jep.2022.115300
Zhang Y, Yang Y, Yan C et al (2022b) A review of the ethnopharmacology, phytochemistry and pharmacology of Cynanchum atratum. J Ethnopharmacol. https://doi.org/10.1016/j.jep.2021.114748
Zhao D, Su S-S, Chen S-F et al (2018) Two new C21 steroidal glycosides isolated from Cynanchum komarovii. Chin J Nat Med 16:610–614. https://doi.org/10.1016/S1875-5364(18)30098-0
Zhou X, Xia W, Zhang Y et al (2020) Cynanchum paniculatum (Bunge) Kitag ex H. Hara: a review of its ethnopharmacology, phytochemistry and pharmacology. J Ethnopharmacol 260:112994. https://doi.org/10.1016/j.jep.2020.112994
Zhu Y, Ouyang Z, Du H et al (2022) New opportunities and challenges of natural products research: when target identification meets single-cell multiomics. Acta Pharm Sin B 12:4011–4039. https://doi.org/10.1016/j.apsb.2022.08.022
Zidorn C (2019) Plant chemophenetics−a new term for plant chemosystematics/plant chemotaxonomy in the macro-molecular era. Phytochemistry 163:147–148. https://doi.org/10.1016/j.phytochem.2019.02.013
Funding
This work was supported by Coordenação de Aperfeiçoamento do Pessoal de Nível Superior (CAPES, Finance Code 001) and Fianciadora de Estudos e Projetos (FINEP) by means of INCT-RENNOFITO.
Author information
Authors and Affiliations
Contributions
TAS, FSVL: Equally contributed designing the study, analyzing data and writing the manuscript first draft; SPC: Phylogenetic reconstruction; JSL, AFA, TAMB: Data survey and analysis; LS, MTS: Machine Learning; LSA and MSS: Critical reading of the manuscript first draft; JFT: Work supervision.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare do not have any commercial or associative interest that represents a conflict of interest in connection with the work submitted.
Informed consent
All authors have read the final manuscript and approved the submission.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
de Souza, T.A., Lins, F.S.V., da Silva Lins, J. et al. Asclepiadoideae subfamily (Apocynaceae): ethnopharmacology, biological activities and chemophenetics based on pregnane glycosides. Phytochem Rev (2024). https://doi.org/10.1007/s11101-024-09914-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11101-024-09914-2