Abstract
Background
Immunotherapy provides new hope to individuals with small cell lung cancer (SCLC). Predicting biomarkers for clinical effects is crucial for SCLC patients receiving programed death-ligand 1 (PD-L1) inhibitor treatment.
Aim
The aim of this study was to clarify the value of serum lipids as predictors of immune related adverse events (irAEs) and the anti-tumour effects in SCLC patients who received PD-L1 inhibitors as first-line treatment.
Method
This study included patients with SCLC who received at least one cycle of PD-L1inhibitors at Shanghai Pulmonary Hospital from August 2020 to December 2023. We collected the clinical data of the SCLC patients, including basic information and serum lipid levels, before immunotherapy.
Results
The irAEs rate was 16.1% of 124 enrolled patients. In multivariate analysis, the triglyceride (TG)/high-density lipoprotein cholesterol (HDL-C) ratio was an independent predictor of irAEs (p = 0.045). Tumour response analysis indicated that the objective response rate (ORR) was 43.4% and the disease control rate (DCR) was 79.5%. Seventy-seven patients experienced any progression-free survival (PFS) event. The median PFS was longer in the HDL-C-high group (10.03 months) than in the HDL-C-low group (6.67 months) (p = 0.043). In Cox regression analysis, the serum HDL-C level was an independent predictor of PFS (p = 0.002). For patients of the high TG/HDL-C ratio, the ORR significantly differed between patients who suffered from any irAEs and those who did not (p = 0.0139).
Conclusion
This study found that serum lipid levels might predict the responses to anti-PD-L1 as first-line treatment for SCLC.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Impact statements
-
Real world data suggest that serum lipids could predict irAEs and antitumour effects in SCLC patients receiving PD-L1 inhibitors as first line treatment.
-
Clinicians and pharmacists can predict subgroups of SCLC patients who will most benefit from immunotherapy and who will suffer irAEs according to their serum lipid levels.
Introduction
Small cell lung cancer (SCLC) is an aggressive disease that is characterized by rapid growth, early metastasis, and recurrence after treatment [1]. Patients with SCLC are typically divided into those with limited-stage (LS) disease and those with extensive-stage (ES) disease by using the Veterans Administration Lung Study Group (VALG) staging system [2]. Most patients with ES-SCLC at presentation have a poor prognosis, and they have a low survival rate [3].
Immune checkpoint inhibitors (ICIs) have improved survival outcomes for many advanced cancer patients [4,5,6,7]. Recently, remarkable developments have been made in immunotherapy-based approaches for SCLC, such as programed death-ligand 1 (PD-L1) inhibitors in combination with chemotherapy for first-line treatment of ES-SCLC [8]. Immunotherapy provides new hope for SCLC patients, but a small subset of patients benefit from it, and it also has an unprecedented toxicity profile. The toxicities, broadly referred to as immune-related adverse events (irAEs), can affect any organ system [9]. These irAEs are generally mild and can be managed, but some irAEs may be severe and even life-threatening [10, 11]. Along with the increasing application of immunotherapy in clinical practice, irAEs should not be overlooked. Therefore, predicting biomarkers for irAEs and the anti-tumour effects of the drugs is crucial for SCLC patients receiving ICI treatment.
An unbalanced lipid profile is often associated with cardiovascular diseases. Moreover, it might promote tumorigenesis, metastasis, recurrence, and even drug resistance, impacting the overall survival of patients with cancer [12,13,14,15]. Recent studies have shown the prognostic impact of serum lipid levels on the outcome of patients undergoing immunotherapy [16,17,18]. Cholesterol and other components of the lipid profile could become new biomarkers to predict the efficacy of ICIs and may also become new biomarkers for irAEs. However, there has not been a definitive study on the relationship between serum lipids and the responses of SCLC patients to PD-L1 inhibitors.
Aim
Clinical and molecular markers that can predict responses to anti-PD-L1 therapy will help clinicians and pharmacists predict the occurrence of irAEs and the anti-tumour effects in cancer patients. We conducted a single centre observational study that aimed to determine whether these serum lipids could predict irAEs and the anti-tumour effects in SCLC patients who received PD-L1 inhibitors as first-line treatment.
Ethics approval
This study was approved by the Ethics Committee of Shanghai Pulmonary Hospital (Ethical approval number: K24-325).
Method
Study design and patient population
The study population consisted of 124 patients who were diagnosed with SCLC and who received first-line chemotherapy combined with PD-L1 inhibitors at Shanghai Pulmonary Hospital from August 2020 to December 2023. Eligible patients met the following criteria: (1) age ≥ 18 years; (2) histological confirmation of SCLC; (3) confirmed record of at least one cycle of PD-L1 inhibitors and etoposide with platinum administration as first-line treatment; (4) serum lipid levels assessed before immunotherapy.
Data extraction
The demographic and clinical characteristics, including age, sex, smoking history, tumour stage, Eastern Cooperative Oncology Group performance status (ECOG PS), diabetes status, cardiovascular disease status, hypertension status, body mass index (BMI) (kg/m2), total cholesterol (TC) (mmol/L), triglycerides (TG) (mmol/L), high-density lipoprotein cholesterol (HDL-C) (mmol/L), low-density lipoprotein cholesterol (LDL-C) (mmol/L), apolipoprotein A-1 (ApoA-1) (g/L), apolipoprotein B (ApoB) (g/L), lipoprotein (a) (Lp(a)) (mg/L), small-dense LDL-C (sdLDL-C) (mmol/L), and their six derivatives, non-high-density lipoprotein cholesterol (non-HDL-C) (mmol/L), TG/HDL-C, non-HDL-C/HDL-C, HDL-C/TC, LDL-C/TC and AopB/ApoA-1, were retrospectively extracted from institutional electronic medical records.
The response to immunotherapy was evaluated according to the Response Evaluation Criteria in Solid Tumors (RECIST) Version 1.1. The objective response rate (ORR) was defined as the sum of complete response (CR) and partial response (PR). The disease control rate (DCR) was defined as the sum of CR, PR and stable disease (SD). Progression-free survival (PFS) was calculated as the time from the initiation of treatment to the occurrence of progressive disease (PD) or death. Data on the effectiveness and irAEs of PD-L1 inhibitors were collected from clinical notes, hospital records, and laboratory data.
Two authors used a standardized form to gather patient-related information, and the two result sets were checked against each other to ensure accuracy and reduce the risk of bias.
Statistical analysis
Continuous variables are expressed by the median and interquartile range (IQR; 25th-75th percentile), while categorical variables are presented as n (%). Student’s t-test and the chi-square test were used to compare continuous and categorical variables, respectively. Factors associated with clinical effects were detected by logistic regression. Survival and subgroup analyses were performed using the Kaplan–Meier method and log-rank test. The discriminative ability of serum lipids for clinical outcomes was evaluated as the areas under receiver operating characteristic (ROC) curves (AUC). Statistical processing was performed via IBM SPSS 23.0 (SPSS, Chicago, IL, USA) as well as GraphPad Prism 9.5 (GraphPad Software, La Jolla, CA, USA). When p value < 0.05, the difference is considered to be statistically significant.
Results
Baseline characteristics
A total of 124 patients were included in this study. Most were males (93.6%). The median age was 65 (IQR 59–70) years, and 53.2% of the patients were over 65. A smoking history was present in 71.0% of the patients. Most patients had ES-SCLC (82.3%). A total of 91.13% of the patients had an ECOG PS ≤ 1. The median BMI was 23.99 (IQR 21.13–25.96) kg/m2, and 39.5% of the patients had BMI ≥ 25 kg/m2.
Table 1 presents an overview of the clinical features of the enrolled patients, including their serum lipid levels (median and IQR). The median TG, TC, HDL-C, LDL-C, non-HDL-C, ApoA-1, ApoB, Lp(a) and sdLDL levels were 1.36 mmol/L, 4.22 mmol/L, 1.08 mmol/L, 2.79 mmol/L, 3.18 mmol/L, 1.39 g/L, 0.85 g/L, 182.75 mgl/L and 0.69 mmol/L, respectively. The median TG/HDL-C, non-HDL-C/HDL-C, HDL-C/TC, LDL-C/TC and ApoB/ApoA-1 ratios were 1.37, 3.09, 0.24, 0.66 and 0.63, respectively.
Serum lipid levels can be influenced by various factors, such as lifestyle habits and genetics. The 124 patients in this study had wide ranges of serum lipid levels. We dichotomized the patients around the median of each serum lipid level, yielding low and high groups for each.
Associations between serum lipids and irAEs
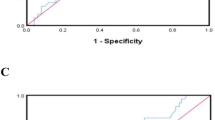
A total of twenty patients (16.1%) in this study experienced any grade of irAE. Two patients discontinued treatment due to intolerant adverse reactions. No treatment-related deaths occurred. Through multivariate logistic analysis, we found that the TG/HDL-C ratio was an independent risk factor for irAEs (hazard ratio (HR): 16.894, 95% confidence interval (CI): 1.065–268.050, p = 0.045) (Table 2). The ROC analysis demonstrated a moderate ability of the TG/HDL-C ratio to predict irAEs (AUC: 0.6938, 95% CI: 0.557–0.831, p = 0.0062) (Fig. 1).
Associations between serum lipids and anti-tumour effects of PD-L1 inhibitors
In patients with an evaluable response (122 patients of the study), tumour response analysis indicated that 53 (43.4%) patients achieved PR, and 44 (36.1%) patients achieved SD, yielding the ORR of 43.4% and the DCR of 79.5% (Table 1). Through logistic analyses, we found that the association between serum lipid levels and the ORR did not reach statistical significance (Supplementary Table 1), but we found that LDL-C and non-HDL-C were reliable predictors of the DCR (HR: 16.309, 95% CI:1.207–220.326, p = 0.036; HR:34.484, 95% CI: 1.840–646.378, p = 0.018) (Supplementary Table 2). The ROC curve analyses demonstrated a poor ability of LDL-C and non-HDL-C to predict the DCR (AUC: 0.5786, 95% CI: 0.4532–0.7039, p = 0.227; AUC: 0.5732, 95% CI: 0.4506–0.6958, p = 0.26) (Supplementary Fig. 1).
Seventy-seven patients (62.1%) experienced any PFS event, and the median PFS (mPFS) was 6.23 months. The 6-month and 1-year PFS rates were 58.4 and 14.3%, respectively. As shown in Fig. 2, LS-SCLC patients had longer mPFS than ES-SCLC patients (17.0 months vs. 6.67 months, Log Rank p = 0.0003) (Fig. 2A). Compared to the TG-high group (mPFS 6.67 months), the TG-low group had a significantly longer mPFS (9.17 months) (Log Rank p = 0.044) (Fig. 2B). The HDL-C-high group had a longer mPFS than the HDL-C-low group (10.03 months vs 6.67 months, Log Rank p = 0.043) (Fig. 2C). Notably, the TG/HDL-C-low group had longer mPFS than the TG/HDL-C-high group (9.17 months vs 6.33 months, Log Rank p = 0.023) (Fig. 2D). Through multivariate Cox regression analysis, we found that tumour stage and HDL-C level were reliable predictors of PFS (HR: 4.619, 95% CI: 1.860–11.467, p = 0.001; HR: 2.988, 95% CI: 1.500–5.950, p = 0.002) (Table 3).
Kaplan–Meier survival curves for PFS. (A) PFS for patients compared among the LS-SCLC group and ES-SCLC groups. (B) PFS for patients compared among the TG-high group and TG-low groups. (C) PFS for patients compared among the HDL-C-high group and HDL-C-low groups. (D) PFS for patients compared among the TG/HDL-C-high group and TG/HDL-C-low groups. PFS Progression-free survival, LS limited-stage, ES extensive-stage, TG triglycerides, HDL-C: high-density lipoprotein cholesterol
Associations between irAEs and anti-tumour effects of PD-L1 inhibitors
After adjusting for potential confounding factors in logistic multivariate analysis, we further investigated the correlation between irAEs and anti-tumour effects of PD-L1 inhibitors. For patients of the high TG/HDL-C ratio, the ORR significantly differed between patients who experienced any irAEs and those who did not (p = 0.0139), but no significant difference was detected between the DCR and any irAEs (Table 4).
Discussion
In the present study, we observed that the impact of the serum lipid profile on the clinical outcomes of patients with SCLC treated with ICIs may vary. Our results showed that the TG/HDL-C ratio had a moderate ability to predict irAEs. High HDL-C levels, low TG levels and low TG/HDL-C ratios at baseline were correlated with better outcomes, and HDL-C was a reliable predictor of PFS in SCLC patients received first-line chemotherapy combined PD-L1 inhibitors. In addition, for patients of the high TG/HDL-C ratio, the ORR significantly differed between patients who suffered from any irAEs and those who did not.
Lipoproteins and serum lipids have been reported to be associated with the morbidity and mortality of cardiovascular diseases and cancer [19, 20]. Lipids have been linked to the fundamental mechanisms underlying SCLC development [21,22,23]. In addition, the serum lipid profile is useful for predicting the prognosis of SCLC patients [24, 25]. Moreover, lipid metabolism signalling plays a key role in the tumour microenvironment and immunotherapy [26]. Cholesterol and other lipids are considered determinants of several alterations occurring in immune cells [27,28,29,30].
In terms of ICI therapeutic efficacy, the serum ApoA-1 and TG levels are remarkable independent prognostic factors for overall survival (OS) in advanced intrahepatic cholangiocarcinoma patients treated with anti-PD-1 therapy [18]. High HDL-C levels at baseline and early elevation of HDL-C levels are correlated with better outcomes in metastatic colorectal cancer patients treated with anti-PD-1 therapy [31]. Early elevated TC, LDL-C, ApoA-1, and ApoB levels were significantly associated with a better ORR in recurrent or metastatic nasopharyngeal carcinoma patients treated with anti-PD-1 therapy [32]. In a retrospective study, the favourable impact of passive cholesterol diffusion on clinical outcome might reflect its main conformation in mature HDL particles, which potentially shapes the inflammatory response, and ultimately promotes ICI efficacy [33]. A multicentre cohort study revealed that a lipid score that defined subgroups of patients with advanced solid tumours who benefit differently from ICIs, where the poor-risk profile (TC < 200 and TG ≥ 150 mg/dl) represented an independent poor prognostic factor for both PFS and OS [34].
In terms of irAEs, through non-targeted lipidomics and semi-targeted lipid quantification analysis, 9 lipids exhibited a good AUC according to the ROC curve for prediction and could be utilized to develop a rapid and effective prediction model for identifying irAEs in advanced non-small cell lung cancer patients treated with first-line chemotherapy combined with immunotherapy [35].
Serum lipid levels could be predictive markers for the response of cancer patients to ICI therapy and their prognosis. However, there has not been a definite study on the relationship between the serum lipid profile and the response of SCLC patients to PD-1 inhibitors. Our research indicated that the TG/HDL-C ratio could be an independent predictive marker for irAEs and that HDL-C was a reliable predictor of PFS in SCLC patients who were treated with first-line chemotherapy combined with anti-PD-L1 therapy.
We acknowledge some limitations of this study. The major limitation was that the study was a single centre observational study, and there may be selection bias in the absence of long-term serum lipid levels after immunotherapy. Additionally, it was a small study. Serum lipid levels can be influenced by various factors, such as lifestyle habits and genetics. Further studies are needed to establish more reliable and independent data, because predicting biomarkers for immunotherapy is crucial for cancer patients.
Conclusion
Serum lipids predicted anti-PD-L1 therapy response in a study including a total of 124 patients with SCLC, and the TG/HDL-C ratio and HDL-C level may be independent predictors of clinical outcomes. These findings can help clinicians and pharmacists predict the occurrence of irAEs and anti-tumour effects based on serum lipid levels.
References
Gazdar AF, Bunn PA, Minna JD. Small-cell lung cancer: what we know, what we need to know and the path forward. Nat Rev Cancer. 2017;17(12):725–37. https://doi.org/10.1038/nrc.2017.87.
Anbazhagan R, Tihan T, Bornman DM, et al. Classification of small cell lung cancer and pulmonary carcinoid by gene expression profiles. Cancer Res. 1999;59(20):5119–22.
Yang S, Zhang Z, Wang Q. Emerging therapies for small cell lung cancer. J Hematol Oncol. 2019;12(1):47. https://doi.org/10.1186/s13045-019-0736-3.
Franzin R, Netti GS, Spadaccino F, et al. The use of immune check point inhibitors in oncology and the occurrence of AKI: where do we stand? Front Immunol. 2020;11:574271. https://doi.org/10.3389/fimmu.2020.574271.
Carlino MS, Larkin J, Long GV. Immune checkpoint inhibitors in melanoma. Lancet. 2021;398(10304):1002–14. https://doi.org/10.1016/S0140-6736(21)01206-X.
Boukouris AE, Theochari M, Stefanou D, et al. Latest evidence on immune checkpoint inhibitors in metastatic colorectal cancer: a 2022 update. Crit Rev Oncol Hematol. 2022;173:103663. https://doi.org/10.1016/j.critrevonc.2022.103663.
Tang S, Qin C, Hu H, et al. Immune checkpoint inhibitors in non-small cell lung cancer: progress, challenges, and prospects. Cells. 2022. https://doi.org/10.3390/cells11030320.
Petty WJ, Paz-Ares L. Emerging strategies for the treatment of small cell lung cancer: a review. JAMA Oncol. 2023;9(3):419–29. https://doi.org/10.1001/jamaoncol.2022.5631.
Palmieri DJ, Carlino MS. Immune checkpoint inhibitor toxicity. Curr Oncol Rep. 2018;20(9):72. https://doi.org/10.1007/s11912-018-0718-6.
Basek A, Jakubiak GK, Cieślar G, et al. Life-threatening endocrinological immune-related adverse events of immune checkpoint inhibitor therapy. Cancers. 2023;15(24):5786. https://doi.org/10.3390/cancers15245786.
Liu LL, Skribek M, Harmenberg U, et al. Systemic inflammatory syndromes as life-threatening side effects of immune checkpoint inhibitors: case report and systematic review of the literature. J ImmunoTher Cancer. 2023;11(3):e005841. https://doi.org/10.1136/jitc-2022-005841.
Cao Y. Adipocyte and lipid metabolism in cancer drug resistance. J Clin Invest. 2019;129(8):3006–17. https://doi.org/10.1172/JCI127201.
Kopecka J, Trouillas P, Gasparovic AC, et al. Phospholipids and cholesterol: inducers of cancer multidrug resistance and therapeutic targets. Drug Resist Updat. 2020;49:100670. https://doi.org/10.1016/j.drup.2019.100670.
Martin-Perez M, Urdiroz-Urricelqui U, Bigas C, et al. The role of lipids in cancer progression and metastasis. Cell Metab. 2022;34(11):1675–99. https://doi.org/10.1016/j.cmet.2022.09.023.
Liu L, Zhang S, Yang HY, et al. Lipid alterations play a role in the integration of PD-1/PD-L1 inhibitors and anlotinib for the treatment of advanced non-small-cell lung cancer. Lipids Health Dis. 2024;23(1):16. https://doi.org/10.1186/s12944-023-01960-7.
Goto W, Kashiwagi S, Kamei Y, et al. Relationship between serum lipid levels and the immune microenvironment in breast cancer patients: a retrospective study. BMC Cancer. 2022;22(1):167. https://doi.org/10.1186/s12885-022-09234-8.
Liu L, Mo M, Chen X, et al. Targeting inhibition of prognosis related lipid metabolism genes including CYP19A1 enhances immunotherapeutic response in colon cancer. J Exp Clin Cancer Res. 2023;42(1):85. https://doi.org/10.1186/s13046-023-02647-8.
Yang Z, Zhang D, Sima X, et al. Levels of pretreatment serum lipids predict responses to PD-1 inhibitor treatment in advanced intrahepatic cholangiocarcinoma. Int Immunopharmacol. 2023;115:109687. https://doi.org/10.1016/j.intimp.2023.109687.
Katzke VA, Sookthai D, Johnson T, et al. Blood lipids and lipoproteins in relation to incidence and mortality risks for CVD and cancer in the prospective EPIC-Heidelberg cohort. BMC Med. 2017;15(1):218. https://doi.org/10.1186/s12916-017-0976-4.
Bian X, Liu R, Meng Y, et al. Lipid metabolism and cancer. J Exp Med. 2021. https://doi.org/10.1084/jem.20201606.
Cristea S, Coles GL, Hornburg D, et al. The MEK5-ERK5 kinase axis controls lipid metabolism in small-cell lung cancer. Cancer Res. 2020;80(6):1293–303. https://doi.org/10.1158/0008-5472.CAN-19-1027.
Merino SM, Fernandez LP, Moreno-Rubio J, et al. Clinical features and lipid metabolism genes as potential biomarkers in advanced lung cancer. BMC Cancer. 2023;23(1):36. https://doi.org/10.1186/s12885-023-10509-x.
Wang X, Gao Y, Wang H, et al. Tumor markers for lipid metabolism-related genes: based on small cell lung cancer and bronchial asthma dual analysis. Environ Toxicol. 2024. https://doi.org/10.1002/tox.24152.
Fernández LP, Merino M, Colmenarejo G, et al. Metabolic health together with a lipid genetic risk score predicts survival of small cell lung cancer patients. Cancers. 2021;13(5):1112. https://doi.org/10.3390/cancers13051112.
Wu M, He Y, Pan C. Analysis of baseline serum lipid profile for predicting clinical outcomes of patients with extensive-stage small cell lung cancer. Cancer Manag Res. 2023;15:773–83. https://doi.org/10.2147/CMAR.S418487.
Wang Y, Wang Y, Ren Y, et al. Metabolic modulation of immune check points and novel therapeutic strategies in cancer. Semin Cancer Biol. 2022;86(Pt 3):542–65. https://doi.org/10.1016/j.semcancer.2022.02.010.
Bleve A, Durante B, Sica A, et al. Lipid metabolism and cancer immunotherapy: immunosuppressive Myeloid cells at the crossroad. Int J Mol Sci. 2020;21(16):5845. https://doi.org/10.3390/ijms21165845.
Zhang H, Zhao W, Li X, et al. Cholesterol metabolism as a potential therapeutic target and a prognostic biomarker for cancer immunotherapy. Onco Targets Ther. 2021;14:3803–12. https://doi.org/10.2147/OTT.S315998.
Galli G, Corsetto PA, Proto C, et al. Circulating fatty acid profile as a biomarker for immunotherapy in advanced non-small cell lung cancer. Clin Lung Cancer. 2022;23(7):e489–99. https://doi.org/10.1016/j.cllc.2022.07.010.
King RJ, Singh PK, Mehla K. The cholesterol pathway: impact on immunity and cancer. Trends Immunol. 2022;43(1):78–92. https://doi.org/10.1016/j.it.2021.11.007.
Xiao B, Ouyang H, Gulizeba H, et al. Nomogram for predicting the prognosis of metastatic colorectal cancer patients treated with anti-PD-1 therapy based on serum lipids analysis. Cancer Immunol Immunother. 2023;72(11):3683–92. https://doi.org/10.1007/s00262-023-03519-y.
Xiao BJ, Sima XX, Chen G, et al. Predictive and prognostic role of early apolipoprotein a-i alteration in recurrent or metastatic nasopharyngeal carcinoma patients treated with anti-PD-1 therapy. Cancer Med. 2023;12(16):16918–28. https://doi.org/10.1002/cam4.6321.
Perrone F, Favari E, Maglietta G, et al. The role of blood cholesterol quality in patients with advanced cancer receiving immune checkpoint inhibitors. Cancer Immunol Immunother. 2023;72(7):2127–35. https://doi.org/10.1007/s00262-023-03398-3.
Pecci F, Cantini L, Cognigni V, et al. Prognostic impact of blood lipid profile in patients with advanced solid tumors treated with immune checkpoint inhibitors: a multicenter cohort study. Oncologist. 2024;29(3):e372–81. https://doi.org/10.1093/oncolo/oyad273.
Yu J, Xiong F, Xu Y, et al. Lipidomics reveals immune-related adverse events in nsclc patients receiving immune checkpoint inhibitor. Int Immunopharmacol. 2024;127:111412. https://doi.org/10.1016/j.intimp.2023.111412.
Acknowledgements
None.
Funding
This research was funded by National Natural Science Cultivation Project of Shanghai Pulmonary Hospital (2023-fkzr-2488) and Supporting Discipline Project of Shanghai Pulmonary Hospital (2022-Pharmacy Department).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflicts of interest
All authors declare no conflicts of interest associated with this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International License, which permits any non-commercial use, sharing, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if you modified the licensed material. You do not have permission under this licence to share adapted material derived from this article or parts of it. The images or other third party material in this article are included in the article’s Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by-nc-nd/4.0/.
About this article
Cite this article
Chen, Q., Shu, P., Yuan, X. et al. Levels of serum lipids predict responses to PD-L1 inhibitors as first-line treatment in small cell lung cancer: an observational study. Int J Clin Pharm (2024). https://doi.org/10.1007/s11096-024-01792-0
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11096-024-01792-0