Abstract
Purpose
New formulations of the glycopeptide drug dalbavancin containing 2-hydroxpropyl-β-cyclodextrin (2HPβCD) with or without divalent metal ions in phosphate buffer (pH 7.0) were tested to evaluate whether these excipients influence the aqueous solution stability of dalbavancin.
Method
Recovery of dalbavancin from phosphate buffered solutions at pH 7.0 with different concentrations of 2HPβCD and a divalent metal ion (Ca2+, Mg2+, or Zn2+) was evaluated by RP-HPLC and HP-SEC after four weeks of storage at 5°C and 55°C. A long-term study of formulations with 2HPβCD and Mg2+ was carried out over six months at 5°C, 25°C, and 40°C using RP-HPLC.
Results
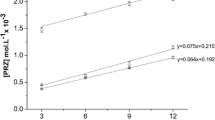
Dalbavancin solutions with either 5.5 mM or 55 mM 2HPβCD were significantly more stable with Mg2+ than with the other divalent metal ions, both at 55°C for four weeks and at 40°C for six months. Dalbavancin was found to be more stable in aqueous solutions at a concentration of 1 mg/mL than at 20 mg/mL with 2HPβCD and Mg2+ at 40°C for six months.
Conclusion
The results suggest that 2HPβCD forms an inclusion complex with dalbavancin that slows the formation of the major degradant, mannosyl aglycone (MAG). The effect of 2HPβCD is increased in the presence of Mg2+ and phosphate at pH 7.0, and the complex is more stable at a dalbavancin concentration of 1 mg/mL than at 20 mg/mL. These observations point towards the possibility of formulating a dalbavancin injection solution with a long shelf life at room temperature and physiological pH.








Similar content being viewed by others
Data Availability
All data generated or analysed during this study are included in this published article.
References
European Medicine Agency. Cyclodextrins Used as Excipients 2017 [Available from: https://www.ema.europa.eu/en/documents/scientific-guideline/questions-answers-cyclodextrins-used-excipients-medicinal-products-human-use_en.pdf. Accessed 29 Mar 2023.
Loftsson T, Brewster ME. Pharmaceutical applications of cyclodextrins: basic science and product development. J Pharm Pharmacol. 2010;62:1607–21.
Stogniew M, Colombo L, Ciabatti R. Dalbavancin compositions for treatment of bacterial infections. US-8143212-B2 (Patent). 2012.
Usach I, Martinez R, Festini T, Peris J-E. Subcutaneous Injection of Drugs: Literature Review of Factors Influencing Pain Sensation at the Injection Site. Adv Ther. 2019;36:2986–96.
Manning MC, Chou DK, Murphy BM, Payne RW, Katayama DS. Stability of protein pharmaceuticals: an update. Pharm Res. 2010;27:544–75.
Avanti C, Amorij J-P, Setyaningsih D, Hawe A, Jiskoot W, VIsser J, et al. A new strategy to stabilize oxytocin in aqueous solutions: I. The effects of divalent metal ions and citrate buffer. AAPS J. 2011;13(2):284–90.
Jakaria SM, Budil DE, Murtagh J. Glycopeptide antibiotic drug stability in aqueous solution. AAPS Open. 2022;8(1):20.
DALVANCE, Dalbavancin injection, powder, lyophilized for solution [package insert]: Durata Therapeutics Inc; 2014 [Available from: https://dailymed.nlm.nih.gov/dailymed/drugInfo.cfm?setid=4b4674d8-4d1e-4728-8465-d42ada33fa5c. Accessed 29 Mar 20223.
Grimm W. Extension of the International Conference on Harmonization Tripartite Guideline for Stability Testing of New Drug Substances and Products to countries of climatic zones III and IV. Drug Dev Ind Pharm. 1998;24:313–25.
Zhu XF, Tan W, Zhou F, Li Z, Duan L. The effect of phosphate buffer solutions on uniconazole complexation with hydroxypropyl-β-cyclodextrin and methyl-β-cyclodextrin. J Incl Phenom Macrocycl Chem. 2011;73:193–8.
Uekama K, Fujinaga T, Hirayama F, Otagiri M, Kurono Y, Ikeda K. Effect of cyclodextrins on the acid hydrolysis of digoxin. J Pharm Pharmacol. 1982;34:627–30.
Takashi S, Shunji H, Takashi Y, Yoichi K, Hirotsu K, Tsuneji N. Digoxin Degradation in Acidic Dissolution Medium. J Pharm Sci. 1980;69:410–3.
Gault MH, Charles JD, Sugden DL, Kepkay DC. Hydrolysis of digoxin by acid. J Pharm Pharmacol. 1977;29:27–32.
Miyake K, Arima H, Hirayama F, Yamamoto M, Horikawa T, Sumiyoshi H, et al. Improvement of Solubility and Oral Bioavailability of Rutin by Complexation with 2-Hydroxypropyl-β-cyclodextrin. Pharm Dev Tech. 2000;5(3):399–407.
Dechene EB. The relative stability of rutin and quercetin in alkaline solution. J Am Pharm Assoc. 1951;11:495–7.
Kapešová J, Petrásková L, Markošová K, Rebroš M, Kotik M, Bojarová P, et al. Bioproduction of Quercetin and Rutinose Catalyzed by Rutinosidase: Novel Concept of “Solid State Biocatalysis". Int J Mol Sci. 2019;20:1112.
Malabarba A, Goldstein BP. Origin, structure, and activity in vitro and in vivo of dalbavancin. J Antimicrob Chemother. 2005;55:ii15–20.
Beauregard DA, Williams DH, Gwynn MN, Knowles DJ. Dimerization and membrane anchors in extracellular targeting of vancomycin group antibiotics. Antimicrob Agents Chemother. 1995;39:781–5.
Hartl E, Winter G, Besheer A. Influence of Hydroxypropyl-Beta-Cyclodextrin on the Stability of Dilute and Highly Concentrated Imunoglobulin G Formulation. J Pharm Sci. 2013;102:4121–31.
Wang W, Martin-Moe S, Pan C, Musza L, Wang YJ. Stabilization of a polypeptide in non-aqueous solvents. Int J Pharm. 2008;351:1–7.
Ananthanarayanan VS, Belciug MP, Zhorov BS. Interaction of oxytocin with Ca2+: II. Proton magnetic resonance and molecular modeling studies of conformations of the hormone and its Ca2+ complex. Biopolym. 1996;40:445–64.
Liu D, Seuthe AB, Ehrler OT, Zhang X, Wyttenbach T, Hsu JF, et al. Oxytocin-receptor binding: why divalent metals are essential. J Am Chem Soc. 2005;127:2024–5.
ICH Harmonised Tripartite Guideline; Stability Testing of New Drug Substance and Products Q1A(R2); Current Step 4 version 2003 [Available from: https://database.ich.org/sites/default/files/Q1A%28R2%29%20Guideline.pdf. Accessed 29 Mar 2023.
Acknowledgements
This research work was supported by Hikma Pharmaceuticals.
Funding
The authors have received no payment for the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
The authors confirm contribution to the paper as follows;
Research work was conducted in the lab, and the manuscript was prepared by Sardar M. Jakaria. David E. Budil and James Murtagh critically reviewed and revised the draft for intellectual content. All authors reviewed, edited, and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Conflict of Interest
The authors state no conflict of interest in preparation of this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Jakaria, S.M., Budil, D.E. & Murtagh, J. Strategies To Stabilize Dalbavancin in Aqueous Solutions; Section 3: The Effects of 2 Hydroxypropyl-β-Cyclodextrin and Phosphate Buffer with and without Divalent Metal Ions. Pharm Res 40, 2027–2037 (2023). https://doi.org/10.1007/s11095-023-03525-w
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-023-03525-w