Abstract
Purpose
Caco-2 monolayers are the most common model of the intestinal epithelium and are critical to the development of oral drug delivery strategies and gastrointestinal disease treatments. However, current monolayer systems are cost- and/or time-intensive, hampering progress. This study evaluates two separate methods to reduce resource input: FB Essence as a fetal bovine serum (FBS) alternative and a new, 3-day Caco-2 system deemed “thrifty, rapid intestinal monolayers” (TRIM).
Methods
Caco-2 cells were cultured with FB Essence and compared to cells in 10% FBS for proliferation and monolayer formation. TRIM were compared to commonly-used 21-day and Corning® HTS monolayer systems, as well as mouse intestines, for permeability behavior, epithelial gene expression, and tight junction arrangement.
Results
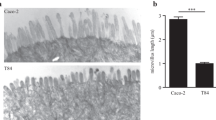
No amount of FB Essence maintained Caco-2 cells beyond 10 passages. In contrast, TRIM compared favorably in permeability and gene expression to intestinal tissues. Furthermore, TRIM cost $109 and required 1.3 h of time per 24-well plate, compared to $164 and 3.7 h for 21-day monolayers, and $340 plus 1.0 h for the HTS system.
Conclusions
TRIM offer a new approach to generating Caco-2 monolayers that resemble the intestinal epithelium. They are anticipated to accelerate the pace of in vitro intestinal experiments while easing financial burden.






Similar content being viewed by others
Abbreviations
- BIEDE:
-
Biocoat® Intestinal Epithelium Differentiation Environment
- CLDN1:
-
Claudin 1
- DMEM:
-
Dulbecco’s Modified Eagles Medium
- FBE:
-
FBEssence
- FBS:
-
Fetal bovine serum
- FITC-DX4:
-
4 kDa molecular weight dextran, tagged with fluorescein isothiocyanate
- HTS:
-
Corning® Biocoat® HTS monolayer system
- P##:
-
Passage number ## of Caco-2 cells
- PPZ:
-
1-phenylpiperazine
- qPCR:
-
Quantitative polymerase chain reaction
- Rhod123:
-
Rhodamine 123
- SLS:
-
Sodium lauryl sulfate
- TEER:
-
Transepithelial electrical resistance
- TRIM:
-
Thrifty, rapid intestinal monolayers
- ZO-1:
-
Zonula occludens 1
References
Pinto M, Robine-Leon S, Appay M-D, Kedinger M, Triadou N, Dussaulx E, et al. Enterocyte-like differentiation and polarization of the human colon carcinoma cell line Caco-2 in culture. Biol Cell. 1983;47(323):323–30.
Sambuy Y, De Angelis I, Ranaldi G, Scarino ML. Stammati a., Zucco F. the Caco-2 cell line as a model of the intestinal barrier: influence of cell and culture-related factors on Caco-2 cell functional characteristics. Cell Biol Toxicol. 2005;21(1):1–26.
Sun H, Chow EC, Liu S, Du Y, Pang KS. The Caco-2 cell monolayer: usefulness and limitations. Expert Opin Drug Metab Toxicol. 2008;4(4):395–411.
Salama NN, Eddington ND, Fasano A. Tight junction modulation and its relationship to drug delivery. Adv Drug Deliv Rev. 2006;58(1):15–28.
Morishita M, Peppas N. a. Is the oral route possible for peptide and protein drug delivery? Drug Discov Today. 2006;11(19–20):905–10.
Maher S, Mrsny RJ, Brayden DJ. Intestinal permeation enhancers for oral peptide delivery. Adv Drug Deliv Rev. 2016;106:277–319. Available from: https://doi.org/10.1016/j.addr.2016.06.005.
Ball RL, Knapp CM, Whitehead KA. Lipidoid nanoparticles for siRNA delivery to the intestinal epithelium: in vitro investigations in a Caco-2 model. PLoS one. 2015;10(7):e0133154 Available from: https://doi.org/10.1371/journal.pone.0133154.
Hua S, Marks E, Schneider JJ, Keely S. Advances in oral nano-delivery systems for colon targeted drug delivery in inflammatory bowel disease: selective targeting to diseased versus healthy tissue. Nanomedicine nanotechnology, biol med. 2015;11(5):1117–32. Available from:. https://doi.org/10.1016/j.nano.2015.02.018.
Zhang M, Viennois E, Prasad M, Zhang Y, Wang L, Zhang Z, et al. Edible ginger-derived nanoparticles: a novel therapeutic approach for the prevention and treatment of inflammatory bowel disease and colitis-associated cancer. Biomaterials. 2016;101:321–40 Available from: https://doi.org/10.1016/j.biomaterials.2016.06.018.
Schumann M, Kamel S, Pahlitzsch ML, Lebenheim L, May C, Krauss M, et al. Defective tight junctions in refractory celiac disease. Ann N Y Acad Sci. 2012;1258(1):43–51.
Fisichella M, Bérenguer F, Steinmetz G, Auffan M, Rose J, Prat O. Intestinal toxicity evaluation of TiO2 degraded surface-treated nanoparticles: a combined physico-chemical and toxicogenomics approach in Caco-2 cells. Part Fibre Toxicol. 2012;9(1):39 Available from: https://doi.org/10.1186/1743-8977-9-18.
Yu H, Huang Q. Investigation of the cytotoxicity of food-grade nanoemulsions in Caco-2 cell monolayers and HepG2 cells. Food Chem. 2013;141(1):29–33.
Konsoula R, Barile F. a. Correlation of in vitro cytotoxicity with paracellular permeability in mortal rat intestinal cells. J Pharmacol Toxicol Methods. 2007;55(2):176–83.
Press B, Di Grandi D. Permeability for intestinal absorption: Caco-2 assay and related issues. Curr Drug Metab. 2008;9(9):893–900.
Volpe DA. Variability in Caco-2 and MDCK cell-based intestinal permeability assays. J Pharm Sci. 2008;97(2):712–25.
Shen L, Weber CR, Raleigh DR, Yu D, Turner JR. Tight junction pore and leak pathways: a dynamic duo. Annu Rev Physiol. 2011;73:283–309.
Artursson P, Palm K, Luthmanb K. Caco-2 monolayers in experimental and theoretical drug transport predictions of drug transport. Adv Drug Deliv Rev. 2001;46(96):27–43.
Natoli M, Leoni BD, D’Agnano I, Zucco F, Felsani A. Good Caco-2 cell culture practices. Toxicol in Vitro. 2012;26(8):1243–6 Available from: https://doi.org/10.1016/j.tiv.2012.03.009.
Chong S, Dando SA, Morrison RA. Evaluation of Biocoat intestinal epithelium differentiation environment (3-day cultured Caco-2 cells) as an absorption screening model with improved screening productivity. Pharm Res. 1997;14:1835–7.
Lentz KA, Hayashi J, Lucisano LJ, Polli JE. Development of a more rapid, reduced serum culture system for Caco-2 monolayers and application to the biopharmaceutics classification system. Int J Pharm. 2000;200(1):41–51.
Yamashita S, Furubayashi T, Kataoka M, Sakane T, Sezaki H, Tokuda H. Optimized conditions for prediction of intestinal drug permeability using Caco-2 cells. Eur J Pharm Sci. 2000;10(3):195–204.
Yamashita S, Konishi K, Yamazaki Y, Taki Y, Sakane T, Sezaki H, et al. New and better protocols for a short-term Caco-2 cell culture system. J Pharm Sci. 2002;91(3):669–79.
Bravo SA, Nielsen CU, Amstrup J, Frokjaer S, Brodin B. In-depth evaluation of Gly-Sar transport parameters as a function of culture time in the Caco-2 cell model. Eur J Pharm Sci. 2004;21(1):77–86.
Piletz JE, Drivon J, Eisenga J, Buck W, Yen S, Mclin M, et al. Human cells grown with or without substitutes for fetal bovine serum. Cell Med. 2018;10:1–11.
Gstraunthaler G, Lindl T, Van Der Valk J. A plea to reduce or replace fetal bovine serum in cell culture media. Cytotechnology. 2013;65:791–3.
Young L, Sung J, Stacey G, Masters JR. Detection of mycoplasma in cell cultures. Nat Protoc. 2010;5(5):929–34 Available from: https://doi.org/10.1038/nprot.2010.43.
Ferruzza S, Rossi C, Scarino ML, Sambuy Y. A protocol for differentiation of human intestinal Caco-2 cells in asymmetric serum-containing medium. Toxicol in Vitro. 2012;26:1247–51.
Casteleyn C, Rekecki A, Van Der Aa A, Simoens P, Van Den Broeck W. Surface area assessment of the murine intestinal tract as a prerequisite for oral dose translation from mouse to man. Lab Anim. 2010;44(3):176–83.
Whitehead K, Karr N, Mitragotri S. Safe and effective permeation enhancers for oral drug delivery. Pharm Res. 2008;25(8):1782–8.
Lamson NG, Cusimano G, Suri K, Zhang A, Whitehead KA. The pH of Piperazine derivative solutions predicts their utility as Transepithelial permeation enhancers. Mol Pharm. 2016;13(2):578–85.
Suzuki T. Regulation of intestinal epithelial permeability by tight junctions. Cell Mol Life Sci. 2013;70(4):631–59.
Srinivasan B, Kolli AR, Esch MB, Abaci HE, Shuler ML, Hickman JJ. TEER measurement techniques for in vitro barrier model systems. J lab autom. 2015;20(2):107–26 Available from: https://doi.org/10.1177/2211068214561025
Howell S, Kenny AJ, Turner AJ. A survey of membrane peptidases in two human colonic cell lines, Caco-2 and HT-29. Biochem J. 1992;284:595–601 Available from: https://doi.org/10.1042/bj2840595
Kam KR, Walsh LA, Bock SM, Koval M, Fischer KE, Ross RF, et al. Nanostructure-mediated transport of biologics across epithelial tissue: enhancing permeability via nanotopography. Nano Lett. 2013;13(1):164–71.
Vllasaliu D, Exposito-Harris R, Heras A, Casettari L, Garnett M, Illum L, et al. Tight junction modulation by chitosan nanoparticles: comparison with chitosan solution. Int J Pharm. 2010;400(1–2):183–93.
Fein KC, Lamson NG, Whitehead KA. Structure-function analysis of Phenylpiperazine derivatives as intestinal permeation enhancers. Pharm Res. 2017;34(6):1320–9.
Yu Q, Wang Z, Li P, Yang Q. The effect of various absorption enhancers on tight junction in the human intestinal Caco-2 cell line. Drug Dev Ind Pharm. 2013;39(4):587–92.
Whitehead K, Mitragotri S. Mechanistic analysis of chemical permeation enhancers for oral drug delivery. Pharm Res. 2008;25(6):1412–9.
Brake K, Gumireddy A, Tiwari A, Chauhan H, Kumari D. In vivo studies for drug development via Oral delivery: challenges, animal models and techniques. Pharm Anal Acta. 2017;08(09) Available from: https://doi.org/10.4172/2153-2435.1000560
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Lamson, N.G., Ball, R.L., Fein, K.C. et al. Thrifty, Rapid Intestinal Monolayers (TRIM) Using Caco-2 Epithelial Cells for Oral Drug Delivery Experiments. Pharm Res 36, 172 (2019). https://doi.org/10.1007/s11095-019-2712-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11095-019-2712-6