Abstract
Purpose
To investigate the nature of drug-excipient interactions between indomethacin (IMC) and methacrylate copolymer Eudragit® E (EE) in the amorphous state, and evaluate the effects on formulation and stability of these amorphous systems.
Methods
Amorphous solid dispersions containing IMC and EE were spray dried with drug loadings from 20% to 90%. PXRD was used to confirm the amorphous nature of the dispersions, and DSC was used to measure glass transition temperatures (Tg). 13C and 15N solid-state NMR was utilized to investigate changes in local structure and protonation state, while 1H T1 and T1ρ relaxation measurements were used to probe miscibility and phase behavior of the dispersions.
Results
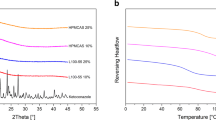
Tg values for IMC-EE solid dispersions showed significant positive deviations from predicted values in the drug loading range of 40–90%, indicating a relatively strong drug-excipient interaction. 15N solid-state NMR exhibited a change in protonation state of the EE basic amine, with two distinct populations for the EE amine at −360.7 ppm (unprotonated) and −344.4 ppm (protonated). Additionally, 1H relaxation measurements showed phase separation at high drug load, indicating an amorphous ionic complex and free IMC-rich phase. PXRD data showed all ASDs up to 90% drug load remained physically stable after 2 years.
Conclusions
15N solid-state NMR experiments show a change in protonation state of EE, indicating that an ionic complex indeed forms between IMC and EE in amorphous solid dispersions. Phase behavior was determined to exhibit nanoscale phase separation at high drug load between the amorphous ionic complex and excess free IMC.








Similar content being viewed by others
Abbreviations
- ASD:
-
Amorphous solid dispersion
- CP:
-
Cross polarization
- DSC:
-
Differential scanning calorimetry
- EE:
-
Eudragit® E
- FTIR:
-
Fourier transform infrared spectroscopy
- HPMCAS:
-
Hydroxypropyl methylcellulose acetate succinate
- HPMCP:
-
Hydroxypropyl methylcellulose phthalate
- IMC:
-
Indomethacin
- MAS:
-
Magic-angle spinning
- NMR:
-
Nuclear magnetic resonance spectroscopy
- PSSA:
-
Polystyrene sulfonic acid
- PVP:
-
Poly(vinylpyrrolidone)
- PVPVA:
-
Poly(vinylpyrrolidone-co-vinyl acetate)
- PXRD:
-
Powder X-ray diffraction
- RF:
-
Radiofrequency
- TOSS:
-
Total sideband suppression
- UV:
-
Ultraviolet spectroscopy
- XPS:
-
X-ray photoelectron spectroscopy
References
Hollenbeck RG, Mitrevej KT, Fan AC. Estimation of the extent of drug-excipient interactions involving croscarmellose sodium. J Pharm Sci. 1983;72(3):325–7.
Ahlneck C, Zografi G. The molecular basis of moisture effects on the physical and chemical stability of drugs in the solid state. Int J Pharm. 1990;62(2–3):87–95.
Durig T, Fassihi AR. Identification of stabilizing and destabilizing effects of excipient-drug interactions in solid dosage form design. Int J Pharm. 1993;97(1–3):161–70.
Claudius JS, Neau SH. The solution stability of vancomycin in the presence and absence of sodium carboxymethyl starch. Int J Pharm. 1998;168(1):41–8.
Lu Q, Zografi G. Phase behavior of binary and ternary amorphous mixtures containing indomethacin, citric acid, and PVP. Pharm Res. 1998;15(8):1202–6.
Byrn SR, Xu W, Newman AW. Chemical reactivity in solid-state pharmaceuticals: formulation implications. Adv Drug Deliv Rev. 2001;48(1):115–36.
McDaid FM, Barker SA, Fitzpatrick S, Petts CR, Craig DQM. Further investigations into the use of high sensitivity differential scanning calorimetry as a means of predicting drug-excipient interactions. Int J Pharm. 2003;252(1–2):235–40.
Bandi N, Wei W, Roberts CB, Kotra LP, Kompella UB. Preparation of budesonide- and indomethacin-hydroxypropyl-β-cyclodextrin (HPBCD) complexes using a single-step, organic-solvent-free supercritical fluid process. Eur J Pharm Sci. 2004;23(2):159–68.
Vueba M, Veiga F, Sousa J, Pina ME. Compatibility studies between ibuprofen or ketoprofen with cellulose ether polymer mixtures using thermal analysis. Drug Dev Ind Pharm. 2005;31(10):943–9.
Batista de Carvalho LAE, Marques MPM, Tomkinson J. Drug-excipient interactions in ketoprofen: a vibrational spectroscopy study. Biopolymers. 2006;82(4):420–4.
Balani PN, Wong SY, Ng WK, Widjaja E, Tan RBH, Chan SY. Influence of polymer content on stabilizing milled amorphous salbutamol sulphate. Int J Pharm. 2010;391(1–2):125–36.
Bharate SS, Bharate SB, Bajaj AN. Interactions and incompatibilities of pharmaceutical excipients with active pharmaceutical ingredients: a comprehensive review. J Excipients Food Chem. 2010;1(3):3–26.
Sperger DM. Solid-state NMR analysis of excipients and drug-excipient interactions in the amorphous state. PhD Thesis; 2010. 259 pp.
Narang AS, Desai D, Badawy S. Impact of excipient interactions on solid dosage form stability. Pharm Res. 2012;29(10):2660–83.
Panakanti R, Narang AS. Impact of excipient interactions on drug bioavailability from solid dosage forms. Pharm Res. 2012;29(10):2639–59.
Dordevic SM, Radulovic TS, Cekic ND, Randelovic DV, Savic MM, Krajisnik DR, et al. Experimental design in formulation of diazepam nanoemulsions: physicochemical and pharmacokinetic performances. J Pharm Sci. 2013;102(11):4159–72.
Chadha R, Bhandari S. Drug-excipient compatibility screening-role of thermoanalytical and spectroscopic techniques. J Pharm Biomed Anal. 2014;87:82–97.
Sachin TV, Deodhar MN, Prakya V. Advances in analytical techniques used in predicting drug-excipient interactions. Int J Pharm Technol. 2014;6(1):6388–417. 6330 pp
Tita B, Ledeti I, Bandur G, Tita D. Compatibility study between indomethacin and excipients in their physical mixtures. J Therm Anal Calorim. 2014;118(2):1293–304.
Chakravarty P, Kothari S, Deese A, Lubach JW. Solid-state characterization of novel propylene glycol ester solvates isolated from lipid formulations. Mol Pharm. 2015;12(7):2551–7.
Schou-Pedersen AMV, Oestergaard J, Cornett C, Hansen SH. Evaluation of microwave oven heating for prediction of drug-excipient compatibilities and accelerated stability studies. Int J Pharm. 2015;485(1–2):97–107.
Lubach JW, Padden BE, Winslow SL, Salsbury JS, Masters DB, Topp EM, et al. Solid-state NMR studies of pharmaceutical solids in polymer matrices. Anal Bioanal Chem. 2004;378(6):1504–10.
Ueda H, Aikawa S, Kashima Y, Kikuchi J, Ida Y, Tanino T, et al. Anti-plasticizing effect of amorphous indomethacin induced by specific intermolecular interactions with PVA copolymer. J Pharm Sci. 2014;103(9):2829–38.
Yuan X, Xiang T-X, Anderson BD, Munson EJ. Hydrogen bonding interactions in amorphous indomethacin and its amorphous solid dispersions with poly(vinylpyrrolidone) and poly(vinylpyrrolidone-co-vinyl acetate) studied using 13C solid-state NMR. Mol Pharm. 2015;12(12):4518–28.
Taylor LS, Zografi G. Spectroscopic characterization of interactions between PVP and indomethacin in amorphous molecular dispersions. Pharm Res. 1997;14(12):1691–8.
Watanabe T, Ohno I, Wakiyama N, Kusai A, Senna M. Stabilization of amorphous indomethacin by co-grinding in a ternary mixture. Int J Pharm. 2002;241(1):103–11.
Takeuchi H, Nagira S, Yamamoto H, Kawashima Y. Solid dispersion particles of amorphous indomethacin with fine porous silica particles by using spray-drying method. Int J Pharm. 2005;293(1–2):155–64.
Vyazovkin S, Dranca I. Effect of physical aging on nucleation of amorphous indomethacin. J Phys Chem B. 2007;111(25):7283–7.
Bhugra C, Shmeis R, Pikal MJ. Role of mechanical stress in crystallization and relaxation behavior of amorphous indomethacin. J Pharm Sci. 2008;97(10):4446–58.
Alonzo DE, Zhang GGZ, Zhou D, Gao Y, Taylor LS. Understanding the behavior of amorphous pharmaceutical systems during dissolution. Pharm Res. 2010;27(4):608–18.
Greco K, Bogner R. Crystallization of amorphous indomethacin during dissolution: effect of processing and annealing. Mol Pharm. 2010;7(5):1406–18.
Karmwar P, Boetker JP, Graeser KA, Strachan CJ, Rantanen J, Rades T. Investigations on the effect of different cooling rates on the stability of amorphous indomethacin. Eur J Pharm Sci. 2011;44(3):341–50.
Ayenew Z, Paudel A, Rombaut P, Van den Mooter G. Effect of compression on non-isothermal crystallization behaviour of amorphous indomethacin. Pharm Res. 2012;29(9):2489–98.
Xiang T-X, Anderson BD. Molecular dynamics simulation of amorphous indomethacin. Mol Pharm. 2013;10(1):102–14.
Lin S-Y, Lin H-L, Chi Y-T, Huang Y-T, Kao C-Y, Hsieh W-H. Thermoanalytical and fourier transform infrared spectral curve-fitting techniques used to investigate the amorphous indomethacin formation and its physical stability in Indomethacin-Soluplus solid dispersions. Int J Pharm. 2015;496(2):457–65.
Thakral NK, Mohapatra S, Stephenson GA, Suryanarayanan R. Compression-induced crystallization of amorphous indomethacin in tablets: characterization of spatial heterogeneity by two-dimensional x-ray diffractometry. Mol Pharm. 2015;12(1):253–63.
Hattori Y, Otsuka M. Analysis of the stabilization process of indomethacin crystals via π-π and CH-π interactions measured by Raman spectroscopy and X-ray diffraction. Chem Phys Lett. 2016;661:114–8.
Chokshi RJ, Shah NH, Sandhu HK, Malick AW, Zia H. Stabilization of low glass transition temperature indomethacin formulations: impact of polymer-type and its concentration. J Pharm Sci. 2008;97(6):2286–98.
Sarode AL, Sandhu H, Shah N, Malick W, Zia H. Hot melt extrusion for amorphous solid dispersions: temperature and moisture activated drug-polymer interactions for enhanced stability. Mol Pharm. 2013;10(10):3665–75.
Kojima T, Higashi K, Suzuki T, Tomono K, Moribe K, Yamamoto K. Stabilization of a supersaturated solution of mefenamic acid from a solid dispersion with EUDRAGIT EPO. Pharm Res. 2012;29(10):2777–91.
Liu H, Zhang X, Suwardie H, Wang P, Gogos CG. Miscibility studies of indomethacin and Eudragit E PO by thermal, rheological, and spectroscopic analysis. J Pharm Sci. 2012;101(6):2204–12.
Weuts I, Kempen D, Verreck G, Peeters J, Brewster M, Blaton N, et al. Salt formation in solid dispersions consisting of polyacrylic acid as a carrier and three basic model compounds resulting in very high glass transition temperatures and constant dissolution properties upon storage. Eur J Pharm Sci. 2005;25(4–5):387–93.
Song Y, Yang X, Chen X, Nie H, Byrn S, Lubach JW. Investigation of drug-excipient interactions in lapatinib amorphous solid dispersions using solid-state NMR spectroscopy. Mol Pharm. 2015;12(3):857–66.
Song Y, Zemlyanov D, Chen X, Nie H, Su Z, Fang K, et al. Acid-base interactions of polystyrene sulfonic acid in amorphous solid dispersions using a combined UV/FTIR/XPS/ssNMR study. Mol Pharm. 2016;13(2):483–92.
Song Y, Zemlyanov D, Chen X, Su Z, Nie H, Lubach JW, et al. Acid-base interactions in amorphous solid dispersions of lumefantrine prepared by spray-drying and hot-melt extrusion using X-ray photoelectron spectroscopy. Int J Pharm. 2016;514(2):456–64.
Metz G, Wu X, Smith SO. Ramped-amplitude cross polarization in magic-angle-spinning NMR. J Magn Reson Ser A. 1994;110(2):219–27.
Pines A, Gibby MG, Waugh JS. Proton-enhanced nuclear induction spectroscopy. Method for high-resolution NMR of dilute spins in solids. J Chem Phys. 1972;56(4):1776–7.
Stejskal EO, Schaefer J, Waugh JS. Magic-angle spinning and polarization transfer in proton-enhanced NMR. J Magn Reson (1969–1992). 1977;28(1):105–12.
Dixon WT, Schaefer J, Sefcik MD, Stejskal EO, McKay RA. Total suppression of sidebands in CPMAS carbon-13 NMR. J Magn Reson (1969–1992). 1982;49(2):341–5.
Song Z, Antzutkin ON, Feng X, Levitt MH. Sideband suppression in magic-angle-spinning NMR by a sequence of 5 pi pulses. Solid State Nucl Magn Reson. 1993;2(3):143–6.
Fung BM, Khitrin AK, Ermolaev K. An improved broadband decoupling sequence for liquid crystals and solids. J Magn Reson. 2000;142(1):97–101.
Barich DH, Gorman EM, Zell MT, Munson EJ. 3-Methylglutaric acid as a 13C solid-state NMR standard. Solid State Nucl Magn Reson. 2006;30(3–4):125–9.
Hayashi S, Hayamizu K. Chemical shift standards in high-resolution solid-state NMR. 2. Nitrogen-15 nuclei. Bull Chem Soc Jpn. 1991;64(2):688–90.
Hancock BC, Shamblin SL, Zografi G. Molecular mobility of amorphous pharmaceutical solids below their glass transition temperatures. Pharm Res. 1995;12(6):799–806.
Couchman PR. Composition-dependent glass-transition temperatures and copolymers. Nature. 1982;298(5876):729–30.
Couchman PR, Karasz FE. A classical thermodynamic discussion of the effect of composition on glass-transition temperatures. Macromolecules. 1978;11(1):117–9.
Chiang P-C, Cui Y, Ran Y, Lubach J, Chou K-J, Bao L, et al. In vitro and in vivo evaluation of amorphous solid dispersions generated by different bench-scale processes, using griseofulvin as a model compound. AAPS J. 2013;15(2):608–17.
Yuan X, Sperger D, Munson EJ. Investigating miscibility and molecular mobility of nifedipine-PVP amorphous solid dispersions using solid-state NMR spectroscopy. Mol Pharm. 2014;11(1):329–37.
Pham TN, Watson SA, Edwards AJ, Chavda M, Clawson JS, Strohmeier M, et al. Analysis of amorphous solid dispersions using 2D solid-state NMR and 1H T1 relaxation measurements. Mol Pharm. 2010;7(5):1667–91.
Chakravarty P, Lubach JW, Hau J, Nagapudi K. A rational approach towards development of amorphous solid dispersions: experimental and computational techniques. Int J Pharm. 2017;519(1–2):44–57.
Author information
Authors and Affiliations
Corresponding author
Additional information
Guest Editors: Tony Zhou and Tonglei Li
Rights and permissions
About this article
Cite this article
Lubach, J.W., Hau, J. Solid-State NMR Investigation of Drug-Excipient Interactions and Phase Behavior in Indomethacin-Eudragit E Amorphous Solid Dispersions. Pharm Res 35, 65 (2018). https://doi.org/10.1007/s11095-018-2364-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11095-018-2364-y