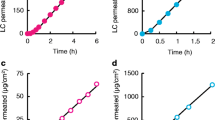
Abstract
Purpose
Although Göttingen minipigs have been widely used for the evaluation of skin absorption, the correlation of minipig skin permeability with human skin absorption remains unclear. This study was designed to investigate the prediction of human plasma concentrations after dermal application of drug products using skin permeability data obtained from minipigs.
Methods
First, in vitro skin permeabilities of seven marketed transdermal drug products were evaluated in minipigs, and compared with in vitro human skin permeability data. Next, plasma concentration-time profiles in humans after dermal applications were simulated using the in vitro minipig skin permeability data. Finally, the in vitro-in vivo correlation of minipig skin permeability was assessed.
Results
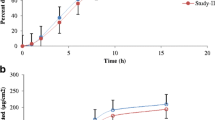
The in vitro skin permeabilities in minipigs were correlated strongly with in vitro human skin permeability data for the same drug products, indicating the utility of minipig skin as an alternative to human skin for in vitro studies. The steady-state plasma concentration or the maximum concentration of drugs was within 2-fold of the clinical data. Bioavailability was approximately 3-fold lower than in vitro permeated fraction.
Conclusions
Predictions using in vitro skin permeability data in Göttingen minipig skin can reproduce the human pharmacokinetic profile, although the prediction of in vivo skin absorption underestimates human absorption.




Similar content being viewed by others
Abbreviations
- AUC:
-
Area under the concentration-time curve
- BA:
-
Bioavailability
- HPLC:
-
High-performance liquid chromatography
- LC/MS/MS:
-
Liquid chromatography/tandem mass spectrometer
- MRT:
-
Mean residence time
REFERENCES
Adler G, Mueller B, Articus K. The transdermal formulation of rivastigmine improves caregiver burden and treatment adherence of patients with Alzheimer's disease under daily practice conditions. Int J Clin Pract. 2014;68(4):465–70.
Yamamoto S, Karashima M, Arai Y, Tohyama K, Amano N. Prediction of human pharmacokinetic profile after transdermal application using excised human skin. J Pharm Sci. 2017;106(9):2787–2794.
Qvist MH, Hoeck U, Kreilgaard B, Madsen F, Frokjaer S. Evaluation of Gottingen minipig skin for transdermal in vitro permeation studies. Eur J Pharm Sci. 2000;11(1):59–68.
Schmook FP, Meingassner JG, Billich A. Comparison of human skin or epidermis models with human and animal skin in in-vitro percutaneous absorption. Int J Pharm. 2001;215(1–2):51–6.
Priborsky J, Muhlbachova E. Evaluation of in-vitro percutaneous absorption across human skin and in animal models. J Pharm Pharmacol. 1990;42(7):468–72.
Stricker-Krongrad A, Shoemake CR, Liu J, Brocksmith D, Bouchard G. The importance of minipigs in dermal safety assessment: an overview. Cutan Ocul Toxicol. 2017:36(2):105–113.
Dick IP, Scott RC. Pig ear skin as an in-vitro model for human skin permeability. J Pharm Pharmacol. 1992;44(8):640–5.
Sato K, Sugibayashi K, Morimoto Y. Species differences in percutaneous absorption of nicorandil. J Pharm Sci. 1991;80(2):104–7.
Singh S, Zhao K, Singh J. In vitro permeability and binding of hydrocarbons in pig ear and human abdominal skin. Drug Chem Toxicol. 2002;25(1):83–92.
Barbero AM, Frasch HF. Pig and guinea pig skin as surrogates for human in vitro penetration studies: a quantitative review. Toxicol in Vitro. 2009;23(1):1–13.
Tse FL, Laplanche R. Absorption, metabolism, and disposition of [14C]SDZ ENA 713, an acetylcholinesterase inhibitor, in minipigs following oral, intravenous, and dermal administration. Pharm Res. 1998;15(10):1614–20.
Gschwind HP, Waldmeier F, Zollinger M, Schweitzer A, Grassberger M. Pimecrolimus: skin disposition after topical administration in minipigs in vivo and in human skin in vitro. Eur J Pharm Sci. 2008;33(1):9–19.
Pershing LK, Huether S, Conklin RL, Krueger GG. Cutaneous blood flow and percutaneous absorption: a quantitative analysis using a laser Doppler velocimeter and a blood flow meter. J Investig Dermatol. 1989;92(3):355–9.
Clarys P, Alewaeters K, Jadoul A, Barel A, Manadas RO, Preat V. In vitro percutaneous penetration through hairless rat skin: influence of temperature, vehicle and penetration enhancers. Eur J Pharm Biopharm. 1998;46(3):279–83.
Auclair F, Besnard M, Dupont C, Wepierre J. Importance of blood flow to the local distribution of drugs after percutaneous absorption in the bipediculated dorsal flap of the hairless rat. Skin Pharmacol. 1991;4(1):1–8.
Hatanaka T, Kamon T, Morigaki S, Katayama K, Koizumi T. Ion pair skin transport of a zwitterionic drug, cephalexin. J Control Release. 2000;66(1):63–71.
Manevski N, Swart P, Balavenkatraman KK, Bertschi B, Camenisch G, Kretz O, et al. Phase II metabolism in human skin: skin explants show full coverage for glucuronidation, sulfation, N-acetylation, catechol methylation, and glutathione conjugation. Drug Metab Dispos. 2015;43(1):126–39.
Jung EC, Maibach HI. Animal models for percutaneous absorption. J Appl Toxicol. 2015;35(1):1–10.
Bollen P, Ellegaard L. The Gottingen minipig in pharmacology and toxicology. Pharmacol Toxicol. 1997;80(Suppl 2):3–4.
Simianer H, Kohn F. Genetic management of the Gottingen Minipig population. J Pharmacol Toxicol Methods. 2010;62(3):221–6.
Benowitz NL, Jacob P 3rd, Denaro C, Jenkins R. Stable isotope studies of nicotine kinetics and bioavailability. Clin Pharmacol Ther. 1991;49(3):270–7.
Hossain M, Jhee SS, Shiovitz T, McDonald C, Sedek G, Pommier F, et al. Estimation of the absolute bioavailability of rivastigmine in patients with mild to moderate dementia of the Alzheimer's type. Clin Pharmacokinet. 2002;41(3):225–34.
Willis JV, Kendall MJ, Flinn RM, Thornhill DP, Welling PG. The pharmacokinetics of diclofenac sodium following intravenous and oral administration. Eur J Clin Pharmacol. 1979;16(6):405–10.
Olkkola KT, Isohanni MH, Hamunen K, Neuvonen PJ. The effect of erythromycin and fluvoxamine on the pharmacokinetics of intravenous lidocaine. Anesth Analg. 2005;100(5):1352–6. table of contents
Leopold G, Pabst J, Ungethum W, Buhring KU. Basic pharmacokinetics of bisoprolol, a new highly beta 1-selective adrenoceptor antagonist. J Clin Pharmacol. 1986;26(8):616–21.
Drugs Interview Form, Bisono® Tape 8mg. Pharmaceuticals and Medical Devices Agency http://www.info.pmda.go.jp/.
Urae A. Comparative bioavailability study of Ba37I42 (Nicotine TTS) after a single application to different skin sites in Japanese smokers. J Clin Therap Med. 1994;10(3):53–62.
Drugs Interview Form, RIVASTACH® Path 18mg. Pharmaceuticals and Medical Devices Agency. http://www.info.pmda.go.jp/.
Yokota H. Safety and Pharmacokinetic Study of L-740. Clin Rep. 1992;26(12):4711–22.
Drugs Interview Form, EMLA® CREAM. Pharmaceuticals and Medical Devices Agency. http://www.info.pmda.go.jp/.
Drugs Interview Form, Voltaren® Tape 30 mg. Pharmaceuticals and Medical Devices Agency. http://www.info.pmda.go.jp/.
Radermacher J, Jentsch D, Scholl MA, Lustinetz T, Frolich JC. Diclofenac concentrations in synovial fluid and plasma after cutaneous application in inflammatory and degenerative joint disease. Br J Clin Pharmacol. 1991;31(5):537–41.
Takeuchi H. Prediction of blood concentration of nicotine or lidocaine after application of their dermal tapes in human from the in vitro permeation through Yucatan micropig skin. Arch Pract Pharm. 2012;72(4):251–61.
Meyer W, Neurand K. Comparison of skin pH in domesticated and laboratory mammals. Arch Dermatol Res. 1991;283(1):16–8.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yamamoto, S., Karashima, M., Sano, N. et al. Utility of Göttingen minipigs for Prediction of Human Pharmacokinetic Profiles After Dermal Drug Application. Pharm Res 34, 2415–2424 (2017). https://doi.org/10.1007/s11095-017-2247-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-017-2247-7