ABSTRACT
Purpose
In general, the surface functionalization of polymeric nanoparticles is carried out by covalently bounding ligands to the nanoparticle surface. This process can cause a lack or decrease of the ligand specificity to its target receptor, besides the need of purification steps. We proposed a ligand-metal-chitosan-lecithin complex as a new strategy to functionalize the surface of biodegradable nanoparticles.
Methods
One pot synthesis of scFv anti-LDL(−)-functionalized nanocapsules was carried out by self-assembly and interfacial reactions. Particle sizing techniques, lipid peroxidation and molecular recognition by enzyme linked immuno sorbent assays were carried out.
Results
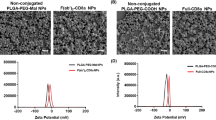
The selected formulation had unimodal size distribution with mean diameter of about 130 nm. The metals in the complex did not enhance the oxidative stress, and the scFv anti-LDL(−)-functionalized nanocapsules recognized LDL(−) and did not react with native LDL indicating the maintenance of the active site of the fragment.
Conclusions
The one pot synthesis, using the ligand-metal-chitosan-lecithin complex to functionalize the surface of the biodegradable nanocapsules, maintained the active site of the antibody fragment making the device interesting for applications in nanomedicine.








Similar content being viewed by others
Abbreviations
- DLS:
-
Dynamic light scattering
- ELISA:
-
Enzyme linked immuno sorbent assay
- LD:
-
Laser diffractometry
- LDE:
-
Laser Doppler electrophoresis
- LDL:
-
Low density lipoprotein
- LDL(−):
-
Electronegative low density lipoprotein
- LNC:
-
Lipid-core nanocapsules
- MCMN:
-
Metal complex multi-wall nanocapsules
- MDA:
-
Malondialdehyde
- MN:
-
Multi-wall nanocapsules
- nLDL:
-
Native low density lipoprotein
- NTA:
-
Nanoparticle tracking analysis
- PCL:
-
Poly(ε-caprolactone)
- PDI:
-
Polydispersity index
- Phe:
-
Phenylalanine
- scFv anti-LDL(−):
-
Anti-electronegative LDL single-chain fragment variable
- SPAN:
-
Polydispersity
- TBA:
-
Thiobarbituric acid
- TBARS:
-
Thiobarbituric acid reactive substances
- TEM:
-
Transmission electron microscopy
REFERENCES
Haley B, Frenkel E. Nanoparticles for drug delivery in cancer treatment. Urol Oncol-Semin Ori. 2008;26:57–64.
Wang M, Thanou M. Targeting nanoparticles to cancer. Pharmacol Res. 2010;62:90–9.
Segala K, Dutra RL, de Oliveira EN, Rossi LM, Matos JR, Paula MMS, et al. Characterization of poly-{trans-RuCl2(vpy)(4)-styrene-4-vinylpyridine} impregnated with silver nanoparticles in non aqueous medium. J Braz Chem Soc. 2006;17:1679–82.
Byrne JD, Betancourt T, Brannon-Peppas L. Active targeting schemes for nanoparticle systems in cancer therapeutics. Adv Drug Deliv Rev. 2008;60:1615–26.
Huang JT, Hou SY, Fang SB, Yu HW, Lee HC, Yang CZ. Development of a biochip using antibody-coated gold nanoparticles to detect specific bioparticles. J Ind Microbiol Biotechnol. 2008;35:1377–85.
Betancourt T, Byrne JD, Sunaryo N, Crowder SW, Kadapakkam M, Patel S, et al. PEGylation strategies for active targeting of PLA/PLGA nanoparticles. J Biomed Mater Res A. 2009;91A:263–76.
Guan XP, Quan DP, Liao KR, Wang T, Xiang P, Mai KC. Preparation and characterization of cationic chitosan-modified poly(D, L-lactide-co-glycolide) copolymer nanospheres as DNA carriers. J Biomater Appl. 2008;22:353–71.
Bernardi A, Zilberstein A, Jager E, Campos MM, Morrone FB, Calixto JB, et al. Effects of indomethacin-loaded nanocapsules in experimental models of inflammation in rats. Br J Pharmacol. 2009;158:1104–11.
Zalipsky S, Wang L, Ding Z, Luo B. Nanoparticle comprises nucleic acid and composition formed by combining cationic polymer conjugate covalently linked to hydrophilic polymer and unconjugated cationic polymer, where nucleic acid and cationic polymer form non-covalent complex. US Patent No. WO2011011631-A2, 2011.
Zhang L, Swift J, Butts SA, Dmochowski IJ. INOR 631-protein-templated gold nanoparticle synthesis and assembly. Abstr Pap Am Chem Soc. 2008;236:631–INOR.
Cheng XH, Chen G, Rodriguez WR. Micro- and nanotechnology for viral detection. Anal Bioanal Chem. 2009;393:487–501.
Li D, Teoh WY, Gooding JJ, Selomulya C, Amal R. Functionalization strategies for protease immobilization on magnetic nanoparticles. Adv Funct Mater. 2010;20:1767–77.
Letchford K, Liggins R, Wasan KM, Burt H. In vitro human plasma distribution of nanoparticulate paclitaxel is dependent on the physicochemical properties of poly(ethylene glycol)-block-poly(caprolactone) nanoparticles. Eur J Pharm Biopharm. 2009;71:196–206.
Jager E, Venturini CG, Poletto FS, Colome LM, Pohlmann JPU, Bernardi A, et al. Sustained release from lipid-core nanocapsules by varying the core viscosity and the particle surface area. J Biomed Nanotechnol. 2009;5:130–40.
Jornada DS, Fiel LA, Bueno K, Gerent JF, Petzhold CL, Beck RCR, et al. Lipid-core nanocapsules: mechanism of self-assembly, control of size and loading capacity. Soft Matter. 2012;8:6646–55.
Reynaud F, Tsapis N, Deyme M, Vasconcelos TG, Gueutin C, Guterres SS, et al. Spray-dried chitosan-metal microparticles for ciprofloxacin adsorption: kinetic and equilibrium studies. Soft Matter. 2011;7:7304–12.
Rodrigues CA, Reynaud F, Stadler E, Drago V. Preparation, characterization, and chromatography properties of chitin modified with FeCl3. J Liq Chromatogr Relat Technol. 1999;22:761–9.
Bender EA, Adorne MD, Colome LM, Abdalla DSP, Guterres SS, Pohlmann AR. Hemocompatibility of poly(epsilon-caprolactone) lipid-core nanocapsules stabilized with polysorbate 80-lecithin and uncoated or coated with chitosan. Int J Pharm. 2012;426:271–9.
Fiel LA, Adorne MD, Guterres SS, Netz PA, Pohlmann AR. Variable temperature multiple light scattering analysis to determine the enthalpic term of a reversible agglomeration in submicrometric colloidal formulations: a quick quantitative comparison of the relative physical stability. Colloid Surface A. 2013;431:93–104.
Pohlmann AR, Fonseca FN, Paese K, Detoni CB, Coradini K, Beck RCR, et al. Poly(epsilon-caprolactone) microcapsules and nanocapsules in drug delivery. Expert Opin Drug Del. 2013;10:623–38.
Schaffazick SR, Pohlmann AR, de Cordova CAS, Creczynski-Pasa TB, Guterres SS. Protective properties of melatonin-loaded nanoparticles against lipid peroxidation. Int J Pharm. 2005;289:209–13.
Faulin TDS, de Sena KCM, Telles AER, Grosso DM, Faulin EJB, Abdalla DSP. Validation of a novel ELISA for measurement of electronegative low-density lipoprotein. Clin Chem Lab Med. 2008;46:1769–75.
Kazuma SM, Cavalcante MF, Telles AER, Maranhão AQ, Abdalla DSP. Cloning and expression of an anti-LDL(−) single chain variable fragment, and its inhibitory effect on experimental atherosclerosis. mABs. 2013;5:763–75.
Ouedraogo HZ, Traore T, Zeba AN, Dramaix-Wilmet M, Hennart P, Donnen P. Effect of an improved local ingredient-based complementary food fortified or not with iron and selected multiple micronutrients on Hb concentration. Public Health Nutr. 2010;13:1923–30.
Le TT, Saveyn P, Hoa HD, Van der Meeren P. Determination of heat-induced effects on the particle size distribution of casein micelles by dynamic light scattering and nanoparticle tracking analysis. Int Dairy J. 2008;18:1090–6.
Filipe V, Hawe A, Jiskoot W. Critical evaluation of Nanoparticle Tracking Analysis (NTA) by NanoSight for the measurement of nanoparticles and protein aggregates. Pharm Res. 2010;27:796–810.
Zimmermann AC, Mecabô A, Fagundes T, Rodrigues CA. Adsorption of Cr(VI) using Fe-crosslinked chitosan complex (Ch-Fe). J Hazard Mater. 2010;179:192–6.
Tang LG, Hon DNS. Chelation of chitosan derivatives with zinc ions. II. Association complexes of Zn2+ onto O,N-carboxymethyl chitosan. J Appl Polym Sci. 2001;79:1476–85.
Byun Y, Hwang JB, Bang SH, Darby D, Cooksey K, Dawson PL, et al. Formulation and characterization of alpha-tocopherol loaded poly epsilon-caprolactone (PCL) nanoparticles. Food Sci Technol–Leb. 2011;44:24–8.
Amorim CD, Couto AG, Netz DJA, de Freitas RA, Bresolin TMB. Antioxidant idebenone-loaded nanoparticles based on chitosan and N-carboxymethylchitosan. Nanomed Nanotech Biol Med. 2010;6:745–52.
Abdalla DSP, de Sena KCM. Lipid peroxidation biomarkers in atherosclerosis. Rev Nutr. 2008;21:749–56.
Janero DR. Malondialdehyde and thiobarbituric acid-reactivity as diagnostic indexes of lipid-peroxidation and peroxidative tissue-injury. Free Radic Biol Med. 1990;9:515–40.
Prassl R, Laggner P. Molecular structure of low density lipoprotein: current status and future challenges. Eur Biophys J. 2009;38:145–58.
Faulin TDS, de Sena-Evangelista KCM, Pacheco DB, Augusto EM, Abdalla DSP. Development of immunoassays for anti-electronegative LDL autoantibodies and immune complexes. Clin Chim Acta. 2012;413:291–7.
ACKNOWLEDGMENTS AND DISCLOSURES
EAB thanks CAPES/Brazil for his fellowship. The authors thank PRONEX e PRONEM FAPERGS/CNPq, FAPESP, INCT-IF/CNPq, CNPq/Brasilia/Brazil, Universal CNPq/MCTI and Rede Nanotecnologia Farmaceutica CAPES (Brazil) for grant financial supports.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Bender, E.A., Cavalcante, M.F., Adorne, M.D. et al. New strategy to surface functionalization of polymeric nanoparticles: one-pot synthesis of scFv anti-LDL(−)-functionalized nanocapsules. Pharm Res 31, 2975–2987 (2014). https://doi.org/10.1007/s11095-014-1392-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-014-1392-5