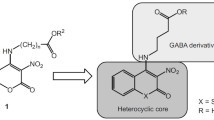
The synthesis and anticonvulsant activity of new coumarin derivatives are reported. N-(3-Nitrocoumarin-4-yl)-4-aminobutyric acid (1a) at doses of 60 and 80 mg/kg (in the MES test) and methyl N-(3,6-dinitrocoumarin-4-yl)-4-aminobutyrate (1e) at doses of 20 and 40 mg/kg (in the corazole antagonism test) possessed the greatest anticonvulsant activity of the synthesized compounds. The results indicated that the coumarin derivatives were promising for further development as potential anticonvulsant agents.






Similar content being viewed by others
References
G. N. Avakyan, Ya. B. Yudel'son, N. N. Maslova, and E. I. Gusev, Zh. Nevropatol. Psikhiatr., 9, 9 – 15 (2003).
V. Rempel, N. Volz, F. Glaser, et al., J. Med. Chem., 56(11), 4798 – 4810 (2013).
M. Curini, G. Cravotto, F. Epifano, and G. Giannone, Curr. Med. Chem., 13, 199 – 222 (2006).
A. A. Emmanuel-Giota, K. C. Fylaktakidou, D. J. Hadjipavlou-Litina, et al., J. Heterocycl. Chem., 38, 717 – 722 (2001).
Z. M. Nofal, M. I. El-Zahar, and S. S. Abd El-Karim, Molecules, 5, 99 – 113 (2000).
B. Dekic, V. Dekic, N. Radulovic, et al., Chem. Pap., 64, 354 – 359 (2010).
J. Xu, J. Ai, S. Liu, et al., Org. Biomol. Chem., 12(22), 3721 – 3734 (2014).
V. L. Savel’ev, O. S. Artamonova, and V. A. Zagorevskii, Khim. Geterotsikl. Soedin., No. 8, 1147 (1972).
V. L. Savel’ev, O. S. Artamonova, and V. S. Troitskaya, Khim. Geterotsikl. Soedin., No. 7, 885 – 890 (1973).
J. D. H. Donnay and D. Harker, Am. Mineral., 22(5), 446 – 467 (1937).
C. F. Macrae, I. J. Bruno, J. A. Chisholm, et al., J. Appl. Crystallogr., 41(2), 466 – 470 (2008).
T. A. Voronina and L. N. Nerobkova, “Methodical instructions for studies of anticonvulsant activity of pharmacological substances,” in: Handbook for Preclinical Drug Trials [in Russian], Part 1, NTsEMSP, Grif i K, Moscow, 2012, pp. 235 – 250.
W. Loscher, C. P. Fassbender, and B. Nolting, Epilepsy Res., 8(2), 79 – 94 (1991).
W. Loscher, D. Honack, C. P. Fassbender, and B. Nolting, Epilepsy Res., 8, 171 – 189 (1991).
M. Kules, M. Trkovnik, and A. Juric, Acta Pharm. Jugosl., 34, 81 (1984).
P. Giannnozzi, S. Baroni, N. Bonini, et al., J. Phys.: Condens. Matter, 21(39), 395502 (2009).
J. P. Perdew, K. Burke, and M. Ernzerhof, Phys. Rev. Lett., 77(18), 3865 – 3868 (1996).
S. Grimme, J. Comput. Chem., 27(15), 1787 – 1799 (2006).
D. Vanderbilt, Phys. Rev. B: Condens. Matter Mater. Phys., 41(11), 7892 – 7895 (1990).
A. A. Coelho, J. Appl. Crystallogr., 36(1), 86 – 95 (2003).
Bruker TOPAS 5 User Manual, Karlsruhe, Germany: Bruker AXS GmbH, 2014.
V. Favre-Nicolin and R. Cerny, J. Appl. Crystallogr., 35(6), 734 – 743 (2002).
I. J. Bruno, J. C. Cole, and M. Kessler, J. Chem. Inf. Comput. Sci., 44(6), 2133 – 2144 (2004).
W. A. Dollase, J. Appl. Crystallogr., 19(4), 267 – 272 (1986).
A. O. Dmitrienko and I. S. Bushmarinov, J. Appl. Crystallogr., 48(6), 1777 – 1784 (2015).
Acknowledgments
A. O. Dmitrienko thanks the Russian Science Foundation (Project No. 16-13-10404) for financial support for the structural studies of 1a.
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 53, No. 2, pp. 25 – 31, February, 2019.
V. L. Savel’ev is deceased.
Rights and permissions
About this article
Cite this article
Mokrov, G.V., Savel’ev, V.L., Voronina, T.A. et al. Synthesis and Anticonvulsant Activity of N-Substituted 4-Amino-3-Nitrocoumarins. Pharm Chem J 53, 118–124 (2019). https://doi.org/10.1007/s11094-019-01964-7
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-019-01964-7