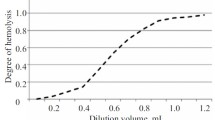
Documentation for standardization of the method for determining hemagglutinins (HAs) in human immunoglobulin preparations used in Russia is presented. The proposed general pharmacopoeial monograph “Determination of anti-A and anti-B hemagglutinins in medicinal preparations of human immunoglobulin” allows quality control of human immunoglobulin preparations for HAcontent using indirect hemagglutination under standard conditions. The positive and negative components of the domestic standard sample (SS) of HAs contain 1:32 anti-A and 1:16 anti-B, respectively; less than 1:2 anti-A and -B; and 1:64 components of the HA content limit. Introduction of the SS for contents of anti-A and anti-B HAs into pharmacopoeial practice would ensure that results obtained in various ranges are accurate and enable the quantitative contents of the corresponding HAs in human immunoglobulin preparations to be determined.
Similar content being viewed by others
References
R. Padmore, Transfusion, 55, S59-S64 (2015); DOI:https://doi.org/10.1111/trf.13090.
O. G. Kornilova, E. V. Paramonova, A. V. Nechaev, et al., Med. Immunol., 19(5), 513 – 520 (2017); DOI:https://doi.org/10.15789/1563-0625-2017-5-513-520.
S. J. Thorpe, Transfusion, 55, S80-S85 (2015).
C. L. Bellac, D. Polatti, T. Hottiger, et al., Biologicals, 42, 57 – 64 (2014).
S. J. Thorpe, B. J. Fox, C. D. Dolman, et al., Biologicals, 33, 111 – 116 (2005).
European Pharmacopoeia EDQM, 8,5 Ed.; http: //online6.edqm.eu/ep801 /# (accessed Apr. 24, 2017).
General Pharmacopoeial Monograph “Determination of anti-A and anti-B hemagglutinins in human immunoglobulin medicinal preparations” (GPM 1.8.2.0005.15); http: //pharmacopoeia.ru/[http: //pharmacopoeia.ru/ofs-1-8-2-0005-15-opredelenie-anti-a-i-anti-v-gemagglyutininov-v-lekarstvennyhpreparatah-immunoglobulinov / ] (accessed Apr. 24, 2017).
S. J. Thorpe, B. Fox, G. Sharp, et al., Vox Sang., 97, 160 – 168 (2009); http: //onlinelibrary.wiley.com/doi/https://doi.org/10.1111/j.1423-0410.2009.01194.x/full[doi:https://doi.org/10.1111/j.1423-0410.2009.01194.x].
General Pharmacopoeial Monograph “Determination of protein” (GPM.1.2.3.0012.15); http: //pharmacopoeia.ru/ofs-1-2-3-0012-15-opredelenie-belka / (accessed Apr. 24, 2017).
S. Glantz, Primer of Biostatistics, McGraw-Hill, New York (1997).
RU Pat. Appl. No. 2017119536, 2017.
Recommendations for the preparation, characterization and establishment of international and other biological reference standards, WHO Technical Report Series 932, annex 2, Geneva (2004), pp. 73 – 131.
GOST 8.315–97, GSI, Certified reference materials for the composition and properties of substances and materials. Basic principles, Izd. Standartov, Moscow (2004).
Author information
Authors and Affiliations
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 51, No. 11, pp. 57 – 60, November, 2017.
Rights and permissions
About this article
Cite this article
Kornilova, O.G., Nechaev, A.V., Borisevich, I.V. et al. Standardization of the Method for Determining Hemagglutinin Content in Human Immunoglobulin Preparations in Russia. Pharm Chem J 51, 1041–1044 (2018). https://doi.org/10.1007/s11094-018-1736-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-018-1736-2