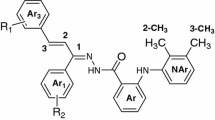
In this research, new analogs of acetaminophen (paracetamol, I) with dimethyl and ethyl substitutions on phenyl moiety and sulfonamide modified by inserting alkylpiperazine derivatives were synthesized (II – V). Then, their conjugated compounds with ibuprofen were also synthesized (VI – IX) and the analgesic and anti-inflammatory activities of new drugs (II – IX) were evaluated in formalin tests on rats. Results indicated that the best analgesic activity was observed for compounds II and VI in diminishing chronic and VI – IX in acute thermal pain tests; in addition, compound VIII showed the best activity in diminishing inflammation. It was concluded that chemical structural binding of the potent synthesized drugs (II – V) with ibuprofen produced new superior antinociceptive and anti-inflammatory drugs (VI – IX) which could alleviate both pain and inflammation.






Similar content being viewed by others
References
L. F. Prescott, Drugs, 25, 290 – 314 (1983).
O. Moling, E. Cairon, G. Rimenti, et al., Clin. Ther. 28, 755 – 760 (2006).
M. R. Yadav, D. M. Nimekar, A. Ananthakrishnan, et al., Bioorg. Med. Chem., 14, 8701 – 8706 (2006).
A. Bertolini, A. Ferrari, A. Ottani, et al., CNS Drug Rev., 12, 250 – 275 (2006).
A. N. Viswanathan, D. Feskanich, E. S. Schernhammer, and S. E. Hankinson, Cancer Res., 68, 2507 – 2513 (2008).
M. A. Altinoz and R. Korkmaz, Neoplasma, 51, 239 – 247 (2004).
B. Bryant, K. Knights, and E. Salerno, Pharmacology for Health Professionals, Elsevier, Australia (2006), p. 270.
A. L. Vaccarino, D. Paul, P. K. Mukherjee, et al., Bioorg. Med. Chem., 15, 2206 – 2215 (2007).
B. Hinz, O. Cheremina, and K. Brune, FASEB J., 22, 383 – 390 (2008).
A. S. Kalgutkar, A. B. Marnett, B. C. Crews, et al., J. Med. Chem., 43, 2860 – 2870 (2000).
M. L. Barbosa, G. M. Melo, Y. K. da Silva, et al., Eur. J. Med. Chem., 44, 3612 – 3620 (2009).
A. Ahmadi, M. Khalili, S. Ahmadian, et al., Pharm. Chem. J., 48 (2), 109 – 115 (2014).
T. J. Reilly, J. Chem. Educ., 76, 1557 (1999).
Chamberlain, Xylazine. http://www.inchem.org/documents/jecfa/jecmono/v38je03.htm.
S. S. Padi and S. K. Kulkarni, Eur. J. Pharmacol., 601, 79 – 87(2008).
H. Hamura, M. Yoshida, K. Shimizu, et al., Jpn. J. Pharmacol., 83, 286 – 292 (2000).
D. Dubuisson and S. G. Dennis, Pain, 4, 161 – 174 (1977).
M. Gupta, U. K. Mazumder, P. Gomathi, and S. V. Thamil, BMC Complement. Altern. Med., 6, 36 (2006).
R. L. Elliott and P. H. Ruehle, US Patent No. 4614798 (1986); Assignee: Vetamix, Shenandoah, Iowa.
M. F. Cheng, H. M. Yu, B. W. Ko, et al., Org. Biomol. Chem., 4, 510 – 518 (2006).
J. R. Bales, J. K. Nicholson, and P. J. Sadler, Clin. Chem., 31, 757 – 762 (1985).
Guideline 106: Control of Pain in Adults with Cancer, Sections 6.1 and 7.1.1, Scottish Intercollegiate Guidelines Network (SIGN), Scotland National Health Service (2008).
B. Kis, J. A. Snipes, and D. W. Busija, J. Pharmacol. Exp. Ther., 315, 1 – 7 (2005).
N. V. Chandrasekharan, H. Dai, and K. L. Roos, Proc. Natl. Acad. Sci. USA, 99, 13926 – 13931 (2002).
C. N. Dewall, G. Macdonald, and G. D. Webster, Psychol. Sci., 21, 931 – 937 (2010).
A. Köfalvi, Alternative interacting sites and novel receptors for cannabinoid ligands, in Cannabinoids and the Brain, Springer-Verlag, Berlin (2008), pp. 131 – 160.
A. Ottani, S. Leone, and M. Sandrini, Eur. J. Pharmacol., 531, 280 – 281 (2006).
D. A. Andersson, C. Gentry, L. Alenmyr, et al., Nat. Commun., 2, 551 (2011).
S. Alshahrani, F. Fernandez-Conti, A. Araujo, and M. DiFulvio, J. Vis. Exp., 63, e3785 (2012).
J. Muñoz, C. Navarro, V. Noriega, et al., Inflammopharmacology, 18, 65 – 71 (2010).
R. M. Rezende, D. S. França, G. B. Menezes, et al., Br. J. Pharmacol., 153, 760 – 768 (2008).
S. S. Ayoub, G. Pryce, M. P. Seed, et al., Drug Metab. Dispos., 39, 1689 – 1695 (2011).
Acknowledgments
This work was a research project at Islamic Azad University, Karaj Branch, Iran and authors would like to express their gratitude to it. They thank Fariba Ansari for her assistance with the pharmacological tests. They appreciate Mojtaba Chaichi, EFL educator at Safir English Language Academy, for proofreading the initial draft of this article.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interest
This research is not a part of our normal lecturing, employment, consultation and involvement. No institution will require any rights from this work too. In addition, no patent has been applied. No commercial right has been given to any company and/or institution, or it will not be done later either.
Rights and permissions
About this article
Cite this article
Ahmadi, A., Khalili, M., Sadeghi, S. et al. Synthesis of New Acetaminophen Analogs and Their Ibuprofen Conjugates as Novel Analgesic Drugs. Pharm Chem J 50, 369–376 (2016). https://doi.org/10.1007/s11094-016-1454-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-016-1454-6