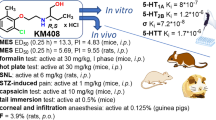
Phencyclidine (PCP, I) and many of its analogs have showed many pharmacological effects through several neurotransmitter systems. In addition to binding to the N-methyl-d-aspartate (NMDA) subtype of the glutamate receptor, it also interferes with other brain functions. In this study, new derivatives (V – VII) were synthesized by changing aromatic moiety (phenyl, thienyl, and benzothiophene) and modifying cyclohexyl and piperidine rings (4-methylcyclohexyl and 3-piperidinol, respectively) of PCP. The acute and chronic pain activities of these new drugs were studied by using the tail immersion and formalin tests on mice and compared to control, PCP, and ketamine treated groups. The obtained results indicated that all new synthesized drugs showed better activity in decreasing acute thermal and chemical pains in tail immersion and formalin tests compared to PCP and ketamine. Also, compound VII (benzothiophene analog) produced more pronounced analgesic effects on acute thermal pain in tail immersion test, as well as compound V (thiophene analog)on acute chemical and chronic pains in formalin test as compared to other drugs.




Similar content being viewed by others
References
A. Ferle-Vidović, M. Kaštelan, D. Petrović, et al., Eur. J. Med. Chem., 28 (3), 243 – 250 (1993).
A. Mozayani, Forensic Sci. Rev., 15(1), 61 – 74 (2002).
J. P. Vincent, J. N. Bidard, M. Lazdunski, et al., Fed. Proc., 42(9), 2570 – 2573 (1983).
E. X. Albuquerque, L. G. Aguayo, J. E. Warnick, et al., Fed. Proc., 42(9), 2584 – 2589 (1983).
K. M. Johnson, L. D. Snell, and R. S. Morter, Pharmacol. Biochem. Behav., 28, 128 (1987).
R. Sircar and S. R. Zukin, Further evidence of phencyclidine / sigma opioid receptor commonality, in: Phencyclidine: An Update, D. H. Clouet (Ed.), NIDA Research Monograph 64, National Institute on Drug Abuse, Rockville, MD (1986), pp. 14 – 23.
J. Diaz, How Drugs Influence Behavior: A Neurobehavioral Approach, Prentice Hall, Englewood Cliffs (1996).
A. Ahmadi, M. Khalili, S. Abbassi, et al., Arzneimittelforsch., 59(4), 202 – 206 (2009).
J. M. Kamenka, J. Hamon, and J. Vignon, US Patent no. 6342511 (2002).
A. Ahmadi, M. Khalili, S. Marami, et al., Mini Rev. Med. Chem., 14(1),64 – 71 (2014).
A. Ahmadi, M. Kermani, N. Naderi, et al., Curr. Med. Chem., 19(5), 763 – 769 (2012).
O. A. al-Deeb, Arzneimittelforschung, 46(5), 505 – 508 (1996).
I. Chaudieu, J. Vinon, M. Chicheportiche, et al., Eur. J. Med. Chem., 22, 359 – 362 (1987).
I. Chaudieu, J. Vignon, M. Chicheportiche, et al., Pharmacol. Biochem. Behav., 32(3), 699 – 705 (1989).
J. Vignon, R. Chicheportiche, M. Chicheportiche, et al., Brain Res., 280, 194 – 197 (1983).
P. Y. Pawar, B. Y. Mane, and S. D. Magar, Curr. Pharm. Res., 2(4), 649 – 654 (2012).
S. Patterson, D. C. Jones, E. J. Shanks, et al., Chem. Med. Chem., 4, 1341 – 1353 (2009).
D. Dubuisson and S. G. Dennis, Pain, 4(2), 161 – 174 (1977).
K. Ramabadran, M. Bansinath, H. Turndorf, and M. M. Puig, J. Pharmacol. Methods, 21 (1), 21 – 31(1989).
P. Bruylants, Bull. Soc. Chim. Belg., 35, 139 – 154 (1926).
V. H. Maddox, E. F. Godefroi, and R. F. Parcell, J. Med. Chem., 8(2), 230 – 235 (1965).
J. March, Advanced Organic Chemistry: Reactions, Mechanisms and Structure, 2nd ed., McGraw-Hill, St. Louis, MO (1977), p. 816
F. A. Carey and R. J. Sundberg, Advanced Organic Chemistry, Part B: Reactions and Synthesis, Plenum Press, New York (1977), pp. 172 – 175.
J. Yoshimura, Y. Ohgo, and T. Sato, J. Am. Chem. Soc., 86, 3858 – 3862 (1964).
Y. Itzhak, A. Kalir, B. A. Weissman, et al., J. Med. Chem., 24, 496 – 499 (1981).
K. M. Carlson and G. C. Wagner, Life Sci., 77, 372 – 385 (2005).
A. Ishii, H. Seno, K. Watanabe-Suzuki, et al., Anal. Chem., 72(2), 404 – 405(2000).
A. Jirgensons, V. Kauss, I. Kalvinsh, et al., Eur. J. Med. Chem., 35, 555 – 565 (2000).
M. Michaud, H. Warren, M. J. Drian, et al., Eur. J. Med. Chem., 29, 869 – 876 (1994).
E. Florence, H. Jacques, H. Hélène, et al., Eur. J. Med. Chem., 35, 323 – 331 (2000).
V. Jacques, P. Valérie, C. Catherine, et al., Eur. J. Pharmacol., 148(3), 427 – 436 (1988).
S. H. Xiao, P. R. Lionel, V. M. Mariena, et al., J. Med. Chem., 36(9), 1188 – 1193(1993).
Acknowledgements
This work was a research project at Karaj Branch, Islamic Azad University, Iran and the authors would like to express their gratitude to them. They thank Fariba Ansari for her assistance with the pharmacological tests. They appreciate Mojtaba Chaichi, EFL educator at Safir English Language Academy, for proofreading the initial draft of this article.
Author information
Authors and Affiliations
Corresponding author
Additional information
Conflict of Interests
Authors declare no conflict of interests.
Rights and permissions
About this article
Cite this article
Ahmadi, A., Khalili, M., Fallah, M. et al. Synthesis and Analgesic Properties of New Modified Analogs of Phencyclidine with Specific Binding on PCP Receptor or Dopamine Inhibition Reuptake Activities. Pharm Chem J 49, 613–619 (2015). https://doi.org/10.1007/s11094-015-1339-0
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-015-1339-0