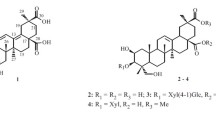
Triterpenes isolated from the dichloromethane extract of Barringtonia asiatica, namely, a mixture of betulinic acid (1) and 22-O-tigloylcamelliagenin A (2) in a 1 : 2 ratio and a mixture of 3β-olean-18-en-3-yl palmitate (7), 3β-urs-12-en-3-yl palmitate (8) and 3β-olean-12-en-3-yl palmitate (9) in a 4 : 1 : 2 ratio (isolated from the bark), as well as germanicol caffeoyl ester (3), germanicol trans-coumaroyl ester (4), germanicol cis-coumaroyl ester (5) and germanicol (6) (from the leaves), and a phenolic compound, verimol K (10) from the flowers, were assessed for cytotoxicity against a human cancer cell line, colon carcinoma (HCT 116), using the MTT assay. The mixture of 1 and 2 exhibited IC50 =8.0 μg · mL–1 against this cell line, while 6 exhibited an IC50 value of 29.6 μg · mL–1. The other compounds tested (3 – 5, 7 – 10) were inactive against the HCT 116 cell line. The mixture of 1 and 2 and compound 6 were further tested for cytotoxicity against the non-small cell lung adenocarcinoma (A549) cell line. The mixture of 1 and 2 and compound 6 exhibited IC50 values of 6.0 and 35.6 μg · mL–1, respectively. The cytotoxicity for the mixture of 1 and 2 may be attributed to betulinic acid (1), a known cytotoxic compound.


Similar content being viewed by others
References
E. Quisumbing, Medicinal Plants of the Philippines, Bureau of Printing, Manila (1978), pp. 649 – 650.
E.-R. E. Mojica, J. R. L. Micor, Int. J. Bot., 3, 325 – 328 (2007).
M. R. Khan, A. D. Omoloso, Fitoterapia, 73, 255 – 260 (2002).
A. J. Herlt, L. N. Mander, E. Pongoh, et al., J. Nat. Prod., 65, 115 – 120 (2002).
R. A. Burton, S. G. Wood, and N. L. Owen, Arkivoc, 13, 137 – 146 (2003).
R. J. Rumampuk, E. J. Pongoh, P. Tarigan, et al., Indon. J. Chem., 3, 149 – 155 (2003).
C. Y. Ragasa, D. L. Espineli, and C.-C. Shen, Chem. Pharm. Bull., 59, 778 – 782 (2011).
C. Y. Ragasa, D. L. Espineli, and C.-C. Shen, Nat. Prod. Res., 26, 1869 – 1875 (2012).
S. Fulda, Int. J. Mol. Sci., 9, 1096 – 1107 (2008).
L. Tripathi, P. Kumar, and R. Singh, Curr. Bioact. Compd., 5, 160 – 168 (2009).
K. Karthishwaran and S. Mirunalini, Int. J. Pharmacol., 6, 836 – 843 (2010).
F. Duke, Biological Activities of Alpha-Amyrin, Dr. Duke’s Phytochemical and Ethnobotanical Databases (2011); http://www.ars-grin.gov/cgi-bin/duke/chemactivities.pl.
T. A. Smith, Exp. Opin. Invest. Drugs, 9, 1841 – 1848 (2000).
C. V. Rao, H. L. Newmark, and B. S. Reddy,. Carcinogenesis, 19, 287 – 290 (1998).
Y. S. Ravikumar, K. M. Mahadevan, H. Manjunatha, et al., Phytomedicine, 17, 513 – 518 (2010).
A. A. Baskar, S. Ignacimuthu, G. M. Paulraj, et al., BMC Comp. Alt. Med., 10, 1 – 7 (2010).
I. Freshney, Culture of Animal Cells, 3rd ed., Wiley-Liss, Inc., New York (1994).
S. D. Jacinto, E. A. C. Chun, A. S. Montuno, et al., Nat. Prod. Commun., 6, 803 – 806 (2011).
C. Y. Ragasa, A. B. Alimboyoguen, and C.-C. Shen, Philipp. Scient., 46, 78 – 87 (2009).
M. Yoshikawa, T. Murakami, S. Yoshizumi, et al., Chem. Pharm. Bull., 44, 1899 – 1907 (1996).
Y. Yang, Z. Deng, P. Proksch, et al., Pharmazie, 61: 365 – 366 (2006).
R. R. S. de Miranda, G. de Fatima Silva, L. P. Duarte, et al., Helv. Chim. Acta, 90, 652 – 658 (2007).
M. L. Narrientos, J. M. David, P. A. Pereira, et al., J. Braz. Chem. Soc., 13, 669 – 673 (2002).
L.-K. Sy and G. D. Brown, J. Nat. Prod., 61, 987 – 992 (1998).
Y. Takada and B. B. Aggarwal, J. Immunol., 171, 3278 – 3286 (2003).
E. Pisha, H. Chai, I. S. Lee, et al., Nature Med., 1, 1046 – 1051 (1995).
S. Fulda, C. Friesen, M. Los, et al., Cancer Res., 57, 4956 – 4964 (1997).
S. Fulda, I. Jeremias, H. H. Steiner, et al., Int. J. Cancer, 82, 435 – 441 (1999).
D. Thurner, D. Turhani, M. Pelzmann, et al., Head Neck, 25, 732 – 740 (2003).
V. Zuco, R. Supino, S. C. Righetti, et al., Cancer Lett., 175, 17 – 25 (2002).
H. Ehrhardt, S. Fulda, M. Fuhrer, et al., Leukemia, 18, 1406 – 1412 (2004).
Acknowledgments
A research grant from the Centennial Fund of De La Salle University — Manila is gratefully acknowledged. The MTT assay was conducted at the Institute of Biology, University of the Philippines, Diliman, Quezon City.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ragasa, C.Y., Espineli, D.L. & Shen, CC. Cytotoxic Triterpene from Barringtonia asiatica . Pharm Chem J 48, 529–533 (2014). https://doi.org/10.1007/s11094-014-1144-1
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-014-1144-1