Abstract
A massive vaccination programme against COVID-19 infection started at the beginning of 2021. Studies show that vaccinated people are subject to reinfection, and there is uncertainty in the rate of immunity loss, the force of infection, recovery rate and vaccine efficacy. Here we study a six-dimensional stochastic epidemic model with vaccine-induced immunity loss to demonstrate the effect of vaccination in controlling the COVID-19 epidemic. It is shown that the disease persists for a long time if the stochastic basic reproduction number \(R^S_{0V}>1\) holds. We have also proved a sufficient condition for disease eradication. Our analysis shows that the disease cannot persist if \(R_{0V}^{\text {ext}}<1\). However, this latter condition may not hold if the infectivity increases and/or the vaccine-induced immunity loss increases. Indian and Italian COVID-19 data are used to demonstrate various dynamical behaviours of the system and disease persistence. A non-trivial observation is that mass vaccination cannot eradicate the disease if the vaccine-induced immunity loss is high. Disease eradication is also challenging with the ongoing immunization process if the infectivity of the virus is also high. These results decipher that the infection will last long unless a long-lasting vaccine candidate appears or a low infectious variant replaces the highly contagious COVID-19 variant.






Similar content being viewed by others
Data availability
The data sets that support the results of this study are taken from the Worldometer website (https://www.worldometers.info/coronavirus/country/india/) and from the Ourworldindata website (https://ourworldindata.org/covid-cases) which are freely available repositories.
References
Adak, D., Majumder, A., Bairagi, N.: Mathematical perspective of Covid-19 pandemic: disease extinction criteria in deterministic and stochastic models. Chaos, Solitons Fractals 142, 110381 (2021)
Anastassopoulou, C., Russo, L., Tsakris, A., Siettos, C.: Data-based analysis, modelling and forecasting of the Covid-19 outbreak. PLoS ONE 15(3), e0230405 (2020)
Bhapkar, H., Mahalle, P.N., Dey, N., Santosh, K.: Revisited Covid-19 mortality and recovery rates: are we missing recovery time period? J. Med. Syst. 44(12), 1–5 (2020)
Chen, C., Kang, Y.: The asymptotic behavior of a stochastic vaccination model with backward bifurcation. Appl. Math. Model. 40(11–12), 6051–6068 (2016)
Choudhary, O.P., Choudhary, P., Singh, I.: India’s Covid-19 vaccination drive: key challenges and resolutions. Lancet. Infect. Dis. 21(11), 1483–1484 (2021)
Croda, J., Ranzani, O.T.: Booster doses for inactivated Covid-19 vaccines: if, when, and for whom. Lancet Infect. Dis. (2021)
in Data, O.W.: The our world in data covid vaccination data. In: ourworldindata.org/covid-vaccinations? country=OWID WRL (2020)
Desmet, K., Wacziarg, R.: JUE insight: understanding spatial variation in Covid-19 across the united states. J. Urban Econom. 127, 103332 (2021)
Diekmann, O., Heesterbeek, J., Roberts, M.G.: The construction of next-generation matrices for compartmental epidemic models. J. R. Soc. Interface 7(47), 873–885 (2010)
Dolgin, E., et al.: Covid vaccine immunity is waning-how much does that matter. Nature 597(7878), 606–607 (2021)
Furuse, Y.: Simulation of future covid-19 epidemic by vaccination coverage scenarios in Japan. J. Global Health 11 (2021)
Ghostine, R., Gharamti, M., Hassrouny, S., Hoteit, I.: An extended SEIR model with vaccination for forecasting the Covid-19 pandemic in Saudi Arabia using an ensemble Kalman filter. Mathematics 9, 636 (2021)
Han, Q., Chen, L., Jiang, D.: A note on the stationary distribution of stochastic SEIR epidemic model with saturated incidence rate. Sci. Rep. 7(1), 1–9 (2017)
Haque, A., Pranto, T.H., Noman, A.A., Mahmood, A.: Insight about detection, prediction and weather impact of coronavirus (Covid-19) using neural network. ArXiv preprint arXiv:2104.02173 (2021)
Juno, J.A., Wheatley, A.K.: Boosting immunity to Covid-19 vaccines. Nat. Med. 27(11), 1874–1875 (2021)
Karako, K., Song, P., Chen, Y., Tang, W.: Analysis of Covid-19 infection spread in Japan based on stochastic transition model. Biosci. Trends 14, 134–138 (2020)
Khajanchi, S., Sarkar, K.: Forecasting the daily and cumulative number of cases for the Covid-19 pandemic in India. Chaos Interdiscip. J. Nonlinear Sci. 30(7), 071101 (2020)
Kurmi, S., Chouhan, U.: A multicompartment mathematical model to study the dynamic behaviour of Covid-19 using vaccination as control parameter. Nonlinear Dyn. 109, 1–17 (2022)
Majumder, A., Adak, D., Bairagi, N.: Persistence and extinction criteria of Covid-19 pandemic: India as a case study. Stoch. Anal. Appl. 1–125 (2021)
Majumder, A., Adak, D., Bairagi, N.: Persistence and extinction of species in a disease-induced ecological system under environmental stochasticity. Phys. Rev. E 103(3), 032412 (2021)
Majumder, A., Adak, D., Bairagi, N.: Phytoplankton-zooplankton interaction under environmental stochasticity: survival, extinction and stability. Appl. Math. Model. 89, 1382–1404 (2021)
Manski, C.F., Molinari, F.: Estimating the Covid-19 infection rate: anatomy of an inference problem. J. Econom. 220(1), 181–192 (2021)
Mao, X.: Stochastic Differential Equations and Applications. Elsevier, Amsterdam (2007)
Merow, C., Urban, M.C.: Seasonality and uncertainty in global Covid-19 growth rates. Proc. Natl. Acad. Sci. 117(44), 27456–27464 (2020)
Mondal, C., Adak, D., Majumder, A., Bairagi, N.: Mitigating the transmission of infection and death due to SARS-COV-2 through non-pharmaceutical interventions and repurposing drugs. ISA Trans. (2020)
Moore, S., Hill, E.M., Tildesley, M.J., Dyson, L., Keeling, M.J.: Vaccination and non-pharmaceutical interventions for Covid-19: a mathematical modelling study. Lancet. Infect. Dis. 21(6), 793–802 (2021)
Musa, M.R., Iyaniwura, S.: Assessing the potential impact of immunity waning on the dynamics of Covid-19: an endemic model of Covid-19. MedRxiv (2021)
Paul, A., Chatterjee, S., Bairagi, N.: Covid-19 transmission dynamics during the unlock phase and significance of testing. medRxiv (2020)
Paul, A., Chatterjee, S., Bairagi, N.: Prediction on Covid-19 epidemic for different countries: focusing on South Asia under various precautionary measures. Medrxiv (2020)
Perc, M., Gorišek Miksić, N., Slavinec, M., Stožer, A.: Forecasting Covid-19. Front. Phys. 8, 127 (2020)
Petrov, V.V.: On the strong law of large numbers. Theor. Probab. Appl. 14(2), 183–192 (1969)
Prem, K., Liu, Y., Russell, T.W., Kucharski, A.J., Eggo, R.M., Davies, N., Flasche, S., Clifford, S., Pearson, C.A., Munday, J.D., et al.: The effect of control strategies to reduce social mixing on outcomes of the Covid-19 epidemic in Wuhan, China: a modelling study. Lancet Public Health 5(5), e261–e270 (2020)
Pritchard, E., Matthews, P.C., Stoesser, N., Eyre, D.W., Gethings, O., Vihta, K.D., Jones, J., House, T., VanSteenHouse, H., Bell, I., et al.: Impact of vaccination on new SARS-COV-2 infections in the united kingdom. Nat. Med. 27, 1–9 (2021)
Rabiu, M., Iyaniwura, S.A.: Assessing the potential impact of immunity waning on the dynamics of cCovid-19 in South Africa: an endemic model of Covid-19. Nonlinear Dyn. 109, 1–21 (2022)
Roghani, A.: The influence of Covid-19 vaccine on daily cases, hospitalization, and death rate in tennessee: a case study in the United States. JMIRx Med 2(3), e29324 (2021)
Rossman, H., Shilo, S., Meir, T., Gorfine, M., Shalit, U., Segal, E.: Covid-19 dynamics after a national immunization program in Israel. Nat. Med. 27, 1–7 (2021)
Sanyaolu, A., Okorie, C., Marinkovic, A., Patidar, R., Younis, K., Desai, P., Hosein, Z., Padda, I., Mangat, J., Altaf, M.: Comorbidity and its impact on patients with Covid-19. SN Compr. Clin. Med. 1–8 (2020)
Sarkar, K., Khajanchi, S., Nieto, J.J.: Modeling and forecasting the Covid-19 pandemic in India. Chaos, Solitons Fractals 139, 110049 (2020)
De la Sen, M., Ibeas, A.: On an SE(Is)(Ih)AR epidemic model with combined vaccination and antiviral controls for Covid-19 pandemic. Adv. Differ. Equ. 2021(1), 1–30 (2021)
de Sousa, L.E., de Oliveira Neto, P.H., da Silva Filho, D.A.: Kinetic Monte Carlo model for the Covid-19 epidemic: impact of mobility restriction on a Covid-19 outbreak. Phys. Rev. E 102(3), 032133 (2020)
WHO: Covid-19 public health emergency of international concern (PHEIC). In: Global Research and Innovation Forum (2020)
WHO: who-prequalification of medical products (ivds, medicines, vaccines and immunization devices, vector control). In: COVID-19 vaccines WHO EUL issued, pp. https://extranet.who.int/pqweb/vaccines/vaccinescovid--19--vaccine--eul--issued. WHO (2021)
Yang, Q., Mao, X.: Stochastic dynamical behavior of sirs epidemic models with random perturbation. Math. Biosci. Eng. 11(4), 1003–1025 (2014)
Zhai, S., Luo, G., Huang, T., Wang, X., Tao, J., Zhou, P.: Vaccination control of an epidemic model with time delay and its application to Covid-19. Nonlinear Dyn. 106(2), 1279–1292 (2021)
Zhang, Y., You, C., Cai, Z., Sun, J., Hu, W., Zhou, X.H.: Prediction of the Covid-19 outbreak based on a realistic stochastic model. MedRxiv (2020)
Zhou, D., Liu, M., Liu, Z.: Persistence and extinction of a stochastic predator-prey model with modified Leslie–Gower and Holling-type II schemes. Adv. Differ. Equ. 2020(1), 1–15 (2020)
Zhou, Y., Zhang, W., Yuan, S.: Survival and stationary distribution of a sir epidemic model with stochastic perturbations. Appl. Math. Comput. 244, 118–131 (2014)
Acknowledgements
Research of Abhijit Majumder is supported by CSIR (File No: 09/096(0874)/2017-EMR-I). Research of N.B. is supported by SERB, India, Ref. No.: MSC/2020/000020.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Appendices
Appendices
Appendix 1
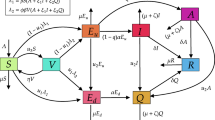
Since the coefficients of the model system (1) are locally Lipschitz continuous, for any \(\bigg (S(0),E(0),A(0),I(0),R(0),\) \(V(0)\bigg )\in {\mathbb {R}}^6_+\), there is a unique local solution \(\bigg (S(t),E(t),\) \(A(t),I(t),R(t),V(t)\bigg )\) \(\in {\mathbb {R}}^6_+\) for all \(t\in [0,\tau _e)\), where \(\tau _e\) is the explosion time [23]. We now prove \(\tau _e=\infty \) a.s. so that the solution becomes global. Let \(\kappa _0>0\) be sufficiently large for every coordinate \(\bigg (S(0),E(0),A(0),I(0),\) \(R(0),V(0)\bigg )\) lying within the interval \(\left[ \frac{1}{\kappa _0},\kappa _0\right] \). We then define, for every integer \(\kappa >\kappa _0\), the stopping time
Thus, \(\tau _\kappa \) is increasing as \(\kappa \rightarrow \infty \). Set \(\lim _{\kappa \rightarrow \infty }\tau _\kappa =\tau _\infty \), when \(\tau _\infty \le \tau _e\) a.s. We now show that \(\tau _\infty =\infty \) by a contradiction. Let us assume that our claim is not true and there exist two constants \(T_2>0\) and \(\epsilon \in (0,1)\) such that \( P(\tau _\infty \le T_2)>\epsilon .\) Thus, there exists an integer \(\kappa _1\ge \kappa _0\) such that
Noticing that \(u+1-\ln {u}>0\) for all \(u>0\) and \((S(t),E(t),A(t),I(t),R(t),V(t))\in {\mathbb {R}}^6_+\), we define the following positive definite function
Applying Ito’s formula, one can have
Noting \(u\le 2(u+1-\ln {u})\) for all \(u>0\) and N is the total population, the above expression becomes
Let \(\varDelta _1=\varLambda +6\,m+q+\beta +\nu +d_i+g+ \omega +\gamma _1+\gamma +d_i+\eta +\frac{1}{2}(\sigma _3^2+\sigma _4^2)\left( 1+\left( \frac{\varLambda }{m}\right) ^2\right) +\sigma _1^2+\sigma _2^2\) and \(\varDelta _2=\max \left\{ 1, \omega ,\gamma ,\gamma _1\right\} \). Then
Defining \(\varDelta _3=\max \{\varDelta _1,\varDelta _2\}\), we have
Noticing that
we have
Since all the functions \(\frac{\sigma _1}{N}\left\{ 1-\frac{S}{E}\right\} [\kappa A+(1-\kappa )I],\) \(\frac{\sigma _2}{N}\left\{ 1-\frac{V}{E}\right\} [\kappa A+(1-\kappa )I], \sigma _3 \left\{ 1-\frac{A}{R}\right\} ,\sigma _4 \left\{ 1-\frac{I}{R}\right\} \) are continuous, bounded and non-anticipative, then for a sequence of partition of the interval \([0,\tau _{\kappa _1} \wedge T_2 ]\) with mesh size \(\varDelta t \rightarrow 0\), one have
Similarly, we have
and
Using the fact that the increment of the Brownian motion is normally distributed with mean zero and variance (\(t_{j+1}-t_j\)), we have
Integrating both sides of (19) from 0 to \(\tau _{\kappa _1} \wedge T_2\), taking the expectation and using the above fact, we obtain
Since L is an increasing function on \([0, \tau _{\kappa _1} \wedge T_2]\), for any \(t \in [0,\tau _{\kappa _1} \wedge T_2],\) \(L\bigg (S(t), E(t), A(t), I(t), R(t), V(t)\bigg )\) \( \le L\bigg (S(\tau _{\kappa _1} \wedge T_2),\) \(E(\tau _{\kappa _1} \wedge T_2), A(\tau _{\kappa _1} \wedge T_2), I(\tau _{\kappa _1} \wedge T_2),\) \(R(\tau _{\kappa _1} \wedge T_2), V(\tau _{\kappa _1} \wedge T_2)\bigg ).\)
Gronwall’s inequality then gives
Set \(\varOmega _{\kappa _1}=\{\tau _{\kappa _1}\le T_2\}\) for all \(\kappa _1\ge \kappa _2\). Thus, following (18), we get \(P(\varOmega _{\kappa _1})\ge \epsilon _3\) for all \(\omega _2\in \varOmega _{\kappa _1}\). Clearly, at least one of \(S(\tau _{\kappa _1},\omega _2), ~E(\tau _{\kappa _1},\omega _2), ~A(\tau _{\kappa _1},\omega _2) ~I(\tau _{\kappa _1},\omega _2),\) \( ~R(\tau _{\kappa _1},\omega _2), ~V(\tau _{\kappa _1},\omega _2)\) is equal to either \(\kappa _1\) or \(\frac{1}{\kappa _1}\). Hence, \(L(S(\tau _{\kappa _1}),E(\tau _{\kappa _1}),A(\tau _{\kappa _1}),I(\tau _{\kappa _1}),R(\tau {\kappa _1}),V(\tau {\kappa _1}))\) is no less than \(\min \{\kappa _1+1-\ln {\kappa _1}, ~\frac{1}{\kappa _1}+1+\ln {\kappa _1}\}\). From (18) and (20), we then obtain
where \(1_{\varOmega _{\kappa _1}}\) is the indicator function of \(\varOmega _{\kappa _1}\). Letting \(\kappa _1\rightarrow \infty \), we get \(\infty >\varDelta _4=\infty \), a contradiction. Hence, \(\tau _\infty =\infty \) a.s. Hence, the theorem is proved.
Appendix 2
One can easily write the deterministic version of the stochastic model (1) as
Using the next-generation matrix method [9], the infection sub-system of the system (21), which describes the production of new infections and makes change in the states, reads
The transmission matrix (F) and the transition matrix (\( \varSigma \)) associated with the system (22) are given by
Then the deterministic basic reproduction number (DBRN) \(R_{0V}^D\) of (21) is the spectral radius of the next-generation matrix \(-{ F\varSigma ^{-1}}\), i.e. \(R_{0V}^D=\rho (-{ F\varSigma ^{-1}})\), where \({ \varSigma ^{-1}}=\)
Thus, \( R_{0V}^D\)=\(\frac{ \omega (\beta m+\eta q)\{\kappa \delta (\gamma +m+d_i)+(1-\kappa )\delta \nu +(1-\kappa )(1-\delta )(\nu +\gamma _1+m)\}}{(q+m)(\gamma +m+d_i)(\nu +\gamma _1+m)(\omega +m)}.\)
If \(R_{0V}^D>1\), then the disease is established in the system.
COVID-19 data fitting with the parameter values and noise intensities as in the Table 2. The first row provides the cumulative actual COVID-19 data (red-coloured curve) of the confirmed, recovered and vaccinated cases in Italy for the period 11 October 2021 to 18 January 2022. The solution (blue-coloured curve) of the stochastic model (1) is the fitted curve with the parameter values of the first row of Table 2. The other rows represent the same with the consecutive periods mentioned in Table 2
Appendix 3
Parameter estimation has been done in two steps [20]. First, we fitted the COVID-19 data with the corresponding deterministic system (21) and next the optimal noise intensities are determined to find the best-fitted parameter set for the stochastic system (1). In order to find the best-fitted parameter values of the deterministic system, we used a MATLAB embedded function, lsqcurvefit, which is a nonlinear solver that minimizes the sum of squared difference between the model output and a given data set. Here, a curve \(h=g(x, \omega )\), parameterized by \(\omega =(\omega _1, \omega _2,..., \omega _m)\), is fitted with the data points \((x_1,h_1), (x_2, h_2),...(x_m, h_m)\). The nonlinear least-squares method finds the certain value of the parameters such that \(\varSigma _{i=1}^m \left( g(x_i, \omega )-h_i\right) ^2\) becomes minimum. With this best-fitted parameter set, we then find the optimum noise intensity for the stochastic system (1). Assuming 10,000 random values of \(\sigma _1, \sigma _2, \sigma _3\) and \(\sigma _4\) between 0 and 1, the stochastic system (2) is simulated 1000 times for each of these four tuples \((\sigma _1, \sigma _2, \sigma _3, \sigma _4)\). We then take the mean of those 1000 evolutions to determine the corresponding r-squared value. The particular value of \(\sigma _1, \sigma _2, \sigma _3\) and \(\sigma _4\) for which the r-squared value is closest to 1 is our required noise intensity.
Appendix 4
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Majumder, A., Bairagi, N. Is large-scale vaccination sufficient for controlling the COVID-19 pandemic with uncertainties? A model-based study. Nonlinear Dyn 112, 2349–2366 (2024). https://doi.org/10.1007/s11071-023-09077-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11071-023-09077-3