Abstract
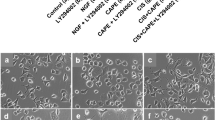
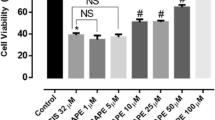
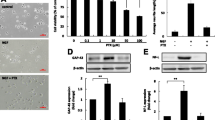
Cisplatin is the most effective and neurotoxic platinum chemotherapeutic agent. It induces a peripheral neuropathy characterized by distal axonal degeneration that might progress to degeneration of cell bodies and apoptosis. Most symptoms occur nearby distal axonal branches and axonal degeneration might induce peripheral neuropathy regardless neuronal apoptosis. The toxic mechanism of cisplatin has been mainly associated with DNA damage, but cisplatin might also affect neurite outgrowth. Nevertheless, the neurotoxic mechanism of cisplatin remains unclear. We investigated the early effects of cisplatin on axonal plasticity by using non-cytotoxic concentrations of cisplatin and PC12 cells as a model of neurite outgrowth and differentiation. PC12 cells express NGF-receptors (trkA) and respond to NGF by forming neurites, branches and synaptic vesicles. For comparison, we used a neuronal model (SH-SY5Y cells) that does not express trkA nor responds to NGF. Cisplatin did not change NGF expression in PC12 cells and decreased neurite outgrowth in both models, suggesting a NGF/trkA independent mechanism. It also reduced axonal growth (GAP-43) and synaptic (synapsin I and synaptophysin) proteins in PC12 cells, without inducing mitochondrial damage or apoptosis. Therefore, cisplatin might affect axonal plasticity before DNA damage, NGF/trkA down-regulation, mitochondrial damage or neuronal apoptosis. This is the first study to show that neuroplasticity-related proteins might be early targets of the neurotoxic action of cisplatin and their role on cisplatin-induced peripheral neuropathy should be investigated in vivo.









Similar content being viewed by others
References
Aloe L, Manni L, Properzi F, De Santis S, Fiore M (2000) Evidence that nerve growth factor promotes the recovery of peripheral neuropathy induced in mice by cisplatin: behavioral, structural and biochemical analysis. Auton Neurosci 86(1–2):84–93. doi:10.1016/S1566-0702(00)00247-2
Amptoulach S, Tsavaris N (2011) Neurotoxicity caused by the treatment with platinum analogues. Chemother Res Pract 2011:843019. doi:10.1155/2011/843019
Asbury AK (1987) Sensory neuronopathy. Semin Neurol 7(1):58–66. doi:10.1055/s-2008-1041406
Bal-Price AK, Hogberg HT, Buzanska L, Coecke S (2010) Relevance of in vitro neurotoxicity testing for regulatory requirements: challenges to be considered. Neurotoxicol Teratol 32(1):36–41. doi:10.1016/j.ntt.2008.12.003
Brouwers EE, Huitema AD, Boogerd W, Beijnen JH, Schellens JH (2009) Persistent neuropathy after treatment with cisplatin and oxaliplatin. Acta Oncologica 48(6):832–841. doi:10.1080/02841860902806609
Carozzi VA, Canta A, Chiorazzi A (2015) Chemotherapy-induced peripheral neuropathy: what do we know about mechanisms? Neurosci Lett 596:90–107. doi:10.1016/j.neulet.2014.10.014
Carozzi VA, Marmiroli P, Cavaletti G (2010) The role of oxidative stress and anti-oxidant treatment in platinum-induced peripheral neurotoxicity. Curr Cancer Drug Targets 10(7):670–682
Cavaletti G, Pezzoni G, Pisano C et al (2002) Cisplatin-induced peripheral neurotoxicity in rats reduces the circulating levels of nerve growth factor. Neurosci Lett 322(2):103–106
Cho T, Tiffany-Castiglioni E (2004) Neurofilament 200 as an indicator of differences between mipafox and paraoxon sensitivity in Sy5Y neuroblastoma cells. J Toxicol Environ Health A 67(13):987–1000. doi:10.1080/15287390490447287
Costa LG, Giordano G, Guizzetti M (2011) In vitro neurotoxicology: an introduction. Methods Mol Biol 758:1–9. doi:10.1007/978-1-61779-170-3_1
Das KP, Freudenrich TM, Mundy WR (2004) Assessment of PC12 cell differentiation and neuritc growth: a comparison of morphological and neurochemical measures. Neurotoxicol Teratol 26(3):397–406. doi:10.1016/j.ntt.2004.02.006
De Santis S, Pace A, Bove L et al (2000) Patients treated with antitumor drugs displaying neurological deficits are characterized by a low circulating level of nerve growth factor. Clin Cancer Res 6(1):90–95
Decatris MP, Sundar S, O’Byrne KJ (2004) Platinum-based chemotherapy in metastatic breast cancer: current status. Cancer Treat Rev 30(1):53–81. doi:10.1016/S0305-7372(03)00139-7
dos Santos NA, Martins NM, Silva Rde B, Ferreira RS, Sisti FM, dos Santos AC (2014) Caffeic acid phenethyl ester (CAPE) protects PC12 cells from MPP+ toxicity by inducing the expression of neuron-typical proteins. Neurotoxicology 45:131–138. doi:10.1016/j.neuro.2014.09.007
Geuna S, Fornaro M, Raimondo S, Giacobini-Robecchi MG (2010) Plasticity and regeneration in the peripheral nervous system. Italian J Anat Embryol 115(1–2):91–94
Greene LA, Tischler AS (1976) Establishment of a noradrenergic clonal line of rat adrenal pheochromocytoma cells which respond to nerve growth factor. Proc Natl Acad Sci USA 73(7):2424–2428
Grothey A (2003) Oxaliplatin-safety profile: neurotoxicity. Semin Oncol 30(4 Suppl 15):5–13
Hamers FP, Plantinga LC, Verhaagen J, Neijt JP, Gispen WH (1996) Upregulation of B50/GAP-43 protein mRNA in rat dorsal root ganglia during cisplatin intoxication. J Neurosci Res 44(2):142–148. doi:10.1002/(SICI)1097-4547(19960415)44:2<142::AID-JNR6>3.0.CO;2-D
Hansen MB, Nielsen SE, Berg K (1989) Re-examination and further development of a precise and rapid dye method for measuring cell growth/cell kill. J Immunol Methods 119(2):203–210
Harrill JA, Mundy WR (2011) Quantitative assessment of neurite outgrowth in PC12 cells. In: Costa LG, Giordano G, Guizzetti M (eds) In vitro neurotoxicology: methods and protocols. Methods in molecular biology, vol 758. Humana Press, New York, pp 331–348
Hausheer FH, Schilsky RL, Bain S, Berghorn EJ, Lieberman F (2006) Diagnosis, management, and evaluation of chemotherapy-induced peripheral neuropathy. Semin Oncol 33(1):15–49. doi:10.1053/j.seminoncol.2005.12.010
Jamsa A, Hasslund K, Cowburn RF, Backstrom A, Vasange M (2004) The retinoic acid and brain-derived neurotrophic factor differentiated SH-SY5Y cell line as a model for Alzheimer’s disease-like tau phosphorylation. Biochem Biophys Res Commun 319(3):993–1000. doi:10.1016/j.bbrc.2004.05.075
Kelley MR, Jiang Y, Guo C, Reed A, Meng H, Vasko MR (2014) Role of the DNA base excision repair protein, APE1 in cisplatin, oxaliplatin, or carboplatin induced sensory neuropathy. PloS One 9(9):e106485. doi:10.1371/journal.pone.0106485
Klein R, Brown D, Turnley AM (2007) Phenoxodiol protects against Cisplatin induced neurite toxicity in a PC-12 cell model. BMC Neurosci 8:61. doi:10.1186/1471-2202-8-61
Krarup-Hansen A, Helweg-Larsen S, Schmalbruch H, Rorth M, Krarup C (2007) Neuronal involvement in cisplatin neuropathy: prospective clinical and neurophysiological studies. Brain 130(Pt 4):1076–1088. doi:10.1093/brain/awl356
L’Episcopo F, Serapide MF, Tirolo C et al (2011) A Wnt1 regulated Frizzled-1/beta-Catenin signaling pathway as a candidate regulatory circuit controlling mesencephalic dopaminergic neuron-astrocyte crosstalk: therapeutical relevance for neuron survival and neuroprotection. Mol Neurodegener 6:49. doi:10.1186/1750-1326-6-49
Lehky TJ, Leonard GD, Wilson RH, Grem JL, Floeter MK (2004) Oxaliplatin-induced neurotoxicity: acute hyperexcitability and chronic neuropathy. Muscle Nerve 29(3):387–392. doi:10.1002/mus.10559
Lobner D (2000) Comparison of the LDH and MTT assays for quantifying cell death: validity for neuronal apoptosis? J Neurosci Methods 96(2):147–152
McDonald ES, Randon KR, Knight A, Windebank AJ (2005) Cisplatin preferentially binds to DNA in dorsal root ganglion neurons in vitro and in vivo: a potential mechanism for neurotoxicity. Neurobiol Dis 18(2):305–313. doi:10.1016/j.nbd.2004.09.013
Park SB, Goldstein D, Krishnan AV et al (2013) Chemotherapy-induced peripheral neurotoxicity: a critical analysis. CA Cancer J Clin 63(6):419–437. doi:10.3322/caac.21204
Pisano C, Pratesi G, Laccabue D et al (2003) Paclitaxel and Cisplatin-induced neurotoxicity: a protective role of acetyl-l-carnitine. Clin Cancer Res 9(15):5756–5767
Podratz JL, Knight AM, Ta LE et al (2011) Cisplatin induced mitochondrial DNA damage in dorsal root ganglion neurons. Neurobiol Dis 41(3):661–668. doi:10.1016/j.nbd.2010.11.017
Quasthoff S, Hartung HP (2002) Chemotherapy-induced peripheral neuropathy. J Neurol 249(1):9–17
Raffa RB, Pergolizzi JV Jr (2013) Cancer chemotherapy-induced neuropathic pain. The underlying peripheral neuropathy. In: Raffa RB, Langford R, Joseph V, Pergolizzi J, Porreca F, Tallarida RJ (eds) Chemotherapy-induced neuropathic pain. CRC Press, Boca Raton, pp 113–135
Rasband WS (1997–2014) ImageJ, U.S. National Institutes of Health. Bethesda, Maryland, USA. http://imagej.nih.gov/ij/
Ray AL (2015) Neuroplasticity, sensitization, and pain In: Deer TR, Leong MS, Ray AL (eds) Treatment of chronic pain by integrative approaches. Springer, New York, pp 15–24
Slotkin TA, Seidler FJ (2010) Oxidative stress from diverse developmental neurotoxicants: antioxidants protect against lipid peroxidation without preventing cell loss. Neurotoxicol Teratol 32(2):124–131. doi:10.1016/j.ntt.2009.12.001
Tsoulfas P, Stephens RM, Kaplan DR, Parada LF (1996) TrkC isoforms with inserts in the kinase domain show impaired signaling responses. J Biol Chem 271(10):5691–5697
Wang D, Lippard SJ (2005) Cellular processing of platinum anticancer drugs. Nat Rev Drug Discov 4(4):307–320. doi:10.1038/nrd1691
Zhang W, Wang GM, Wang PJ, Zhang Q, Sha SH (2014) Effects of neural stem cells on synaptic proteins and memory in a mouse model of Alzheimer’s disease. J Neurosci Res 92(2):185–194. doi:10.1002/jnr.23299
Acknowledgments
The authors would like to thank CNPq (Conselho Nacional de Desenvolvimento Científico e Tecnológico) for the financial support (Grant Number 140106/2015-4).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ferreira, R.S., dos Santos, N.A.G., Martins, N.M. et al. Non-cytotoxic Concentration of Cisplatin Decreases Neuroplasticity-Related Proteins and Neurite Outgrowth Without Affecting the Expression of NGF in PC12 Cells. Neurochem Res 41, 2993–3003 (2016). https://doi.org/10.1007/s11064-016-2019-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-016-2019-5