Abstract
Purpose
This study aimed to evaluate the prognostic performance of amino-acid PET in high-grade gliomas (HGG) patients at the time of temozolomide (TMZ) treatment discontinuation, after the Stupp protocol.
Methods
The analysis included consecutive HGG patients with dynamic [18F]FDOPA PET imaging within 3 months of the end of TMZ therapy, post-Stupp protocol. Static and dynamic PET parameters, responses to RANO criteria for MRI and clinical and histo-molecular factors were correlated to progression-free (PFS).
Results
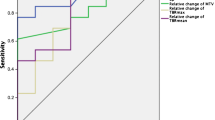
Thirty-two patients (59.4 [54.0;67.6] years old, 13 (41%) women) were included. Static PET parameters peak tumor-to-background ratio and metabolic tumor volume (respective thresholds of 1.9 and 1.5 mL) showed the best 84% accuracies for predicting PFS at 6 months (p = 0.02). These static PET parameters were also independent predictor of PFS in multivariate analysis (p ≤ 0.05).
Conclusion
In HGG patients having undergone a Stupp protocol, the absence of significant PET uptake after TMZ constitutes a favorable prognostic factor.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
The treatment of high-grade glioma (HGG) is based on total surgical resection when possible [1] followed by the Stupp protocol, which combines radiotherapy with concomitant and subsequently adjuvant chemotherapy with Temozolomide (TMZ) [2]. Although the treatment strategy is adapted to each individual, patients in good general and neurological condition are offered adjuvant TMZ maintenance cycles following the traditional Stupp protocol [3]. Nevertheless, 60–75% of glioblastoma patients derive no benefit from TMZ [4], and 15–20% develop clinically toxicity [4]. Diagnostic tools for monitoring TMZ treatment are therefore required.
Amino-acid positron emission tomography (PET) is a recommended adjunct to magnetic resonance imaging (MRI), all along the clinical course of glioma [5]. Whilst the current literature on the value of amino acid PET at initial diagnosis, treatment planning and detection of recurrences is expanding rapidly [6], studies investigating its impact on treatment follow-up remain scarce. Importantly, few studies have to date reported results of amino-acid PET planning at the time of TMZ discontinuation in patients who had undergone a Stupp protocol and none examined the amino-acid PET radiotracer, 3,4-dihydroxy-6-[18F]-fluoro-L-phenylalanine ([18F]FDOPA) [7].
This study therefore aimed to evaluate the prognostic performances of [18F]FDOPA PET at the time of TMZ discontinuation in HGG patients that completed the Stupp protocol.
Material and methods
Population
We retrospectively included consecutive HGG patients referred for dynamic [18F]FDOPA PET between December 2018 to February 2022 within 3 months after TMZ discontinuation following a Stupp protocol. For all these patients, the PET was prescribed to evaluate the clinical response to TMZ. All glioma lesions were reclassified by histo-molecular diagnostics according to the WHO 2021 brain tumor classification [8]. The decisions to treat with the Stupp protocol and to discontinue TMZ were taken at multidisciplinary neuro-oncological board meetings. Progression free survival (PFS) was defined as the time intervals from the end of TMZ treatment to progression, according to the RANO criteria [9].
[18F]FDOPA PET imaging
Patients fasted for at least 4 h before the scan on a digital PET/CT camera (Vereos; Philips®). From December 2018 to September 2020, patients received Carbidopa 1 h before the radiotracer injection to increase striatal dopaminergic activity and tumor uptake. A 30 min dynamic PET acquisition was performed following the injection of 2 MBq 18F-FDOPA/kg of body weight. One static image based on the last 20 min of acquisitions and 30 frames of 1 min each for dynamic images were reconstructed using an OSEM 3D algorithm (2 iterations, 10 subsets, 256 × 256 × 164 voxels of 1 × 1 × 1 mm3) and no post-filtering. Details of reconstruction processes are available elsewhere [10].
Semi-quantitative analysis of imaging
All dynamic [18F]FDOPA PET images were co-registered with static PET images using a rigid transform.
On static PET images, a bounding box was first delineated for each lesion using the LifeX software [11] to specifically avoid including regions with physiological uptake such as the striatum. PET segmentation was performed semi-automatically using a 1.6-fold SUVmean healthy brain threshold [12,13,14]. Tumor-to-background (TBR) and tumor-to-striatum (TSR) ratios with respectively SUVmax, SUVmean and SUVpeak of the tumor volume were determined. Time-to-peak (TTP) and slope of uptake value between the 10th and the 30th minute post injection were calculated [12]. To correct for the impact of Carbidopa, static TBR parametric images and normalized region-based time activity curves were generated [15].
All MRI images, at the beginning and at the end of the TMZ therapy. including at least T1-weighted with injection and T2-FLAIR weighted sequences, were reviewed and MRI responses classified as complete or partial, and stable or progressive disease according to the RANO criteria according to reports of experienced neuro-radiologists [9].
All PET final segmentations and MRI classifications were reviewed by an experienced physician (A.V.) to ascertain the quality of the methods applied.
Statistical analysis
Categorical variables are expressed as percentages and continuous variables as medians [first quartiles; third quartiles]. Receiver operating characteristic curves of static and dynamic PET parameters were calculated for PFS at 6 months with decision cut-offs defined as the point on the curve closest to (0,1). Univariate and multivariate Cox regression analyses were performed to test for associations between PFS and amino-acid PET uptake as well as MRI response assessment and clinical factors, including age, sex, the WHO global status, whether the patient had a biopsy or a subtotal or complete surgery, the WHO 2021 glioma grade, the IDH and the MGMT statutes. A p-value < 0.05 was considered significant. Statistical analyses were performed using R version 4.1.1 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Thirty-two patients (59.4 [54.0;67.6] years old, 13 (41%) women) having received a median of 6 [6;12] TMZ cycles were finally included. The histo-molecular diagnoses were glioblastoma IDH-wildtype (n = 27, 84.4%), astrocytoma IDH-mutant (n = 4, 12.5%) and diffuse midline glioma, H3k27M-mutant (n = 1, 3.1%). In the 29 patients with MGMT status available, 13 (45%) presented a methylation. PET scans were performed at 21 [11;37] days after the end of TMZ. During follow-up of 32.4 [22.8;37.5] months, 20 patients progressed (7 progressions at 6 months) and 11 patients died. 21 patients (66%) had a significant amino-acid PET uptake (i.e., a metabolic tumor volume, MTV) at the end of TMZ treatment. The median time interval between MRI and amino acid PET was 3 [-13.5;13.25] days. 14 patients (44%) had a complete or partial response by MRI. All detailed characteristics of patients are available in the Supplemental Table 1.
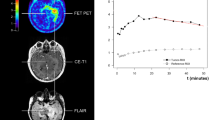
All static PET parameters, except TSRmean, were significant for the prediction of PFS at 6 months, with TBRpeak (1.9 threshold) and MTV (1.5 mL threshold) showing the best accuracies (84%, p = 0.02, Table 1). Among all clinical, MRI and PET parameters, only TBRpeak and MTV were significant in univariate Cox model analysis (p ≤ 0.05, Table 2). In multivariate analysis, TBRpeak represented the only significant predictor of PFS in the best model (HR = 3.6 [1.1–12.1], p = 0.04) and MTV in the second-best model (HR = 1.3 [1.0–1.6], p = 0.05). Figure 1 shows representative images with and without significant PET uptake. Supplemental Fig. 1 shows Kaplan Meier curves for patients with or without significant amino-acid PET uptake (i.e. with an MTV) at the end of TMZ.
(a)(c) Static [18F]FDOPA PET images; (b)(d) Co-registered PET and MRI images (fused). The two left panels are those of an 81-years-old man with an IDH-wildtype glioblastoma with respective PET parameters obtained after 22 TMZ cycles: TBRpeak: 2.3, MTV: 7.3 mL. Follow-up revealed progression at 3.9 months. The two right panels are those of a 54-years-old man with an IDH-wildtype glioblastoma with respective PET parameters obtained after 6 TMZ cycles: TBRpeak: 1.5, MTV: 0 ml. Follow-up revealed progression at 12.5 months
Discussion
The present study shows that the absence of significant uptake in [18F]FDOPA PET after cycles of adjuvant TMZ constitutes a favorable prognostic factor.
Several studies have investigated the value of amino-acid PET for treatment follow-up of gliomas [7], but none thus far with the amino-acid [18F]FDOPA radiotracer at the time of TMZ discontinuation, post Stupp protocol. The most complete study to date was performed with 18F-fluoro-ethyl-tyrosine ([18F]-FET) in a prospective cohort of 79 glioblastoma patients [16]. In this study, the MTV was a prognostic factor of survival but only prior to any treatment. Another study of 44 glioblastoma patients using 11C-methionine ([11C]-MET) showed that a positive MTV, defined as tumor uptake at least two-fold above healthy brain uptake, was an independent prognostic factor of PFS, at the time of adjuvant TMZ discontinuation [17]. This is in line with our current results where the presence of an MTV (of at least 1.5 mL), as well as significant TBRpeak PET parameter (threshold at 1.9), were predictors of PFS at 6 months at the time of TMZ discontinuation (Table 1). Moreover, TBRpeak or MTV were independent prognostic factors of PFS whilst MRI RANO criteria, clinical and histo-molecular factors were not (Table 2). It is noteworthy that application of the proposed cut-offs (near 2 for TBRpeak and 0 mL for MTV) are entirely feasible in routine practice. A positive MTV 1.6-fold greater than the SUVmean healthy brain threshold is reported in the European glioma guidelines for amino-acid PET [13] and was subsequently applied to [18F]FDOPA PET imaging [12] is associated with shorter survival albeit non-significantly mainly due to the low number of patients included (Supplemental Fig. 1). The current study also confirms that dynamic PET parameters provide less prognostic information than static PET parameters in previously treated glioma patients [18].
Our patient population is highly selected because it excludes most HGG patients with recurrences occurring during TMZ adjuvant therapy as part of a Stupp protocol. However, determining the prognosis of patients at the time of TMZ discontinuation is generally a matter for debate at multidisciplinary neuro-oncology board meetings, reinforcing the critical importance of a prognostic biomarker in these types of patients.
To conclude, performing amino-acid PET in HGG patients after TMZ therapy is a useful prognostic tool which should help clinicians decide whether or not to discontinue treatment based on the significant uptake observed on PET.
Data Availability
The datasets generated during and/or analyzed during the current study are available in the Supplemental Table 1.
References
Molinaro AM, Hervey-Jumper S, Morshed RA, Young J, Han SJ, Chunduru P, Zhang Y, Phillips JJ, Shai A, Lafontaine M, Crane J, Chandra A, Flanigan P, Jahangiri A, Cioffi G, Ostrom Q, Anderson JE, Badve C, Barnholtz-Sloan J, Sloan AE, Erickson BJ, Decker PA, Kosel ML, LaChance D, Eckel-Passow J, Jenkins R, Villanueva-Meyer J, Rice T, Wrensch M, Wiencke JK, Oberheim Bush NA, Taylor J, Butowski N, Prados M, Clarke J, Chang S, Chang E, Aghi M, Theodosopoulos P, McDermott M, Berger MS (2020) Association of Maximal Extent of Resection of Contrast-Enhanced and Non–Contrast-Enhanced Tumor With Survival Within Molecular Subgroups of Patients With Newly Diagnosed Glioblastoma. JAMA Oncol 6:495. https://doi.org/10.1001/jamaoncol.2019.6143
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJB, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO, European Organisation for Research and Treatment of Cancer Brain Tumor and Radiotherapy Groups, National Cancer Institute of Canada Clinical Trials Group (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352:987–99. https://doi.org/10.1056/NEJMoa043330
Hegi ME, Diserens A-C, Gorlia T, Hamou M-F, De Tribolet N, Weller M, Kros JM, Hainfellner JA, Mason W, Mariani L, Bromberg JEC, Hau P, Mirimanoff RO, Cairncross JG, Janzer RC, Stupp R (2005) MGMT Gene Silencing and Benefit from Temozolomide in Glioblastoma. N Engl J Med 352:997–1003. https://doi.org/10.1056/NEJMoa043331
Chamberlain MC (2010) Temozolomide: therapeutic limitations in the treatment of adult high-grade gliomas. Expert Rev Neurother 10:1537–1544. https://doi.org/10.1586/ern.10.32
Albert NL, Weller M, Suchorska B, Galldiks N, Soffietti R, Kim MM, la Fougère C, Pope W, Law I, Arbizu J, Chamberlain MC, Vogelbaum M, Ellingson BM, Tonn JC (2016) Response Assessment in Neuro-Oncology working group and European Association for Neuro-Oncology recommendations for the clinical use of PET imaging in gliomas. Neuro-Oncol 18:1199–1208. https://doi.org/10.1093/neuonc/now058
Verger A, Kas A, Darcourt J, Guedj E (2022) PET Imaging in Neuro-Oncology: An Update and Overview of a Rapidly Growing Area. Cancers 14:1103. https://doi.org/10.3390/cancers14051103
Prather KY, O’Neal CM, Westrup AM, Tullos HJ, Hughes KL, Conner AK, Glenn CA, Battiste JD (2022) A systematic review of amino acid PET in assessing treatment response to temozolomide in glioma. Neuro-Oncol Adv 4:vdac008. https://doi.org/10.1093/noajnl/vdac008
Louis DN, Perry A, Wesseling P, Brat DJ, Cree IA, Figarella-Branger D, Hawkins C, Ng HK, Pfister SM, Reifenberger G, Soffietti R, von Deimling A, Ellison DW (2021) The 2021 WHO Classification of Tumors of the Central Nervous System: a summary. Neuro-Oncol 23:1231–1251. https://doi.org/10.1093/neuonc/noab106
Chukwueke UN, Wen PY (2019) Use of the Response Assessment in Neuro-Oncology (RANO) criteria in clinical trials and clinical practice. CNS Oncol 8:CNS28. https://doi.org/10.2217/cns-2018-0007
Ahrari S, Zaragori T, Zinsz A, Oster J, Imbert L, Verger A (2024) Application of PET imaging delta radiomics for predicting progression-free survival in rare high-grade glioma. Sci Rep 14:3256. https://doi.org/10.1038/s41598-024-53693-x
Nioche C, Orlhac F, Boughdad S, Reuzé S, Goya-Outi J, Robert C, Pellot-Barakat C, Soussan M, Frouin F, Buvat I (2018) LIFEx: A Freeware for Radiomic Feature Calculation in Multimodality Imaging to Accelerate Advances in the Characterization of Tumor Heterogeneity. Cancer Res 78:4786–4789. https://doi.org/10.1158/0008-5472.CAN-18-0125
Zaragori T, Oster J, Roch V, Hossu G, Chawki MB, Grignon R, Pouget C, Gauchotte G, Rech F, Blonski M, Taillandier L, Imbert L, Verger A (2022) 18F-FDOPA PET for the Noninvasive Prediction of Glioma Molecular Parameters: A Radiomics Study. J Nucl Med Off Publ Soc Nucl Med 63:147–157. https://doi.org/10.2967/jnumed.120.261545
Law I, Albert NL, Arbizu J, Boellaard R, Drzezga A, Galldiks N, la Fougère C, Langen K-J, Lopci E, Lowe V, McConathy J, Quick HH, Sattler B, Schuster DM, Tonn J-C, Weller M (2019) Joint EANM/EANO/RANO practice guidelines/SNMMI procedure standards for imaging of gliomas using PET with radiolabelled amino acids and [18F]FDG: version 1.0. Eur J Nucl Med Mol Imaging 46:540–557. https://doi.org/10.1007/s00259-018-4207-9
Albert NL, Galldiks N, Ellingson BM, Van Den Bent MJ, Chang SM, Cicone F, De Groot J, Koh E-S, Law I, Le Rhun E, Mair MJ, Minniti G, Rudà R, Scott AM, Short SC, Smits M, Suchorska B, Tolboom N, Traub-Weidinger T, Tonn J-C, Verger A, Weller M, Wen PY, Preusser M (2024) PET-based response assessment criteria for diffuse gliomas (PET RANO 1.0): a report of the RANO group. Lancet Oncol 25:e29–e41. https://doi.org/10.1016/S1470-2045(23)00525-9
Bros M, Zaragori T, Rech F, Blonski M, Hossu G, Taillandier L, Marie P-Y, Verger A (2021) Effects of Carbidopa Premedication on 18F-FDOPA PET Imaging of Glioma: A Multiparametric Analysis. Cancers 13:5340. https://doi.org/10.3390/cancers13215340
Suchorska B, Jansen NL, Linn J, Kretzschmar H, Janssen H, Eigenbrod S, Simon M, Pöpperl G, Kreth FW, la Fougere C, Weller M, Tonn JC, Network GG (2015) Biological tumor volume in 18FET-PET before radiochemotherapy correlates with survival in GBM. Neurology 84:710–719. https://doi.org/10.1212/WNL.0000000000001262
Hirono S, Hasegawa Y, Sakaida T, Uchino Y, Hatano K, Iuchi T (2019) Feasibility study of finalizing the extended adjuvant temozolomide based on methionine positron emission tomography (Met-PET) findings in patients with glioblastoma. Sci Rep 9:17794. https://doi.org/10.1038/s41598-019-54398-2
Rozenblum L, Zaragori T, Tran S, Morales-Martinez A, Taillandier L, Blonski M, Rech F, Galanaud D, Kas A, Verger A (2022) Differentiating high-grade glioma progression from treatment-related changes with dynamic [18F]FDOPA PET: a multicentric study. Eur Radiol 33:2548–2560. https://doi.org/10.1007/s00330-022-09221-4
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
All authors participated either to: 1) study conception and design or analysis and interpretation of the data, or both (AZ; SA; JB; AM; VR; LD; MS; TZ; AV) 2) the drafting of the manuscript or revising it critically for important intellectual content (AZ; TZ; AV) or 3) final approval of the submitted manuscript (SA; JB; AM; VR; LD; MS; MB; LT; TZ; AV).
Corresponding author
Ethics declarations
Ethical approval
The institutional ethics committee (Comité d’Ethique du CHRU de Nancy) approved the evaluation of retrospective patient data and the trial was registered at ClinicalTrials.gov (NCT04469244) and complied with the principles of the Declaration of Helsinki.
Consent to participate
Informed consent was obtained from all individual participants included in the study.
Consent to publish
Not applicable.
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Zinsz, A., Ahrari, S., Becker, J. et al. Amino-acid PET as a prognostic tool after post Stupp protocol temozolomide therapy in high-grade glioma patients. J Neurooncol (2024). https://doi.org/10.1007/s11060-024-04722-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11060-024-04722-2