Abstract
Purpose
Understanding the complex bidirectional interactions between neurons and glioma cells could help to identify new therapeutic targets. Herein, the techniques and application of novel neuroscience tools implemented to study the complex interactions between brain and malignant gliomas, their results, and the potential therapeutic opportunities were reviewed.
Methods
Literature search was performed on PubMed between 2001 and 2023 using the keywords “glioma”, “glioblastoma”, “circuit remodeling”, “plasticity”, “neuron networks” and “cortical networks”. Studies including grade 2 to 4 gliomas, diffuse midline gliomas, and diffuse intrinsic pontine gliomas were considered.
Results
Glioma cells are connected through tumour microtubes and form a highly connected network within which pacemaker cells drive tumorigenesis. Unconnected cells have increased invasion capabilities. Glioma cells are also synaptically integrated within neural circuitry. Neurons promote tumour growth via paracrine and direct electrochemical mechanisms, including glutamatergic AMPA-receptors. Increased glutamate release in the tumor microenvironment and loss of peritumoral GABAergic inhibitory interneurons result in network hyperexcitability and secondary epilepsy. Functional imaging, local field potentials and subcortical mapping, performed in awake patients, have defined patterns of malignant circuit remodeling. Glioma-induced remodeling is frequent in language and even motor cortical networks, depending on tumour biological parameters, and influences functional outcomes.
Conclusion
These data offer new insights into glioma tumorigenesis. Future work will be needed to understand how tumor intrinsic molecular drivers influence neuron-glioma interactions but also to integrate these results to design new therapeutic options for patients.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Introduction
Malignant gliomas are the most frequent malignant primary brain tumours in adults and are associated with a poor prognosis despite an improved understanding of tumor intrinsic drives of disease progression [1, 2]. Surgical resection, temozolomide with brain irradiation and, TTF-fields have been demonstrated to significantly prolong overall survival, yet recurrence occurs for nearly all patients and few patients survive longer than 2 years [3]. Nervous system regulation of cancer (termed Cancer Neuroscience) is an emerging discipline that focuses on defining and therapeutically targeting interactions between the nervous system and cancer [4]. There is a growing body of evidence defining the complex bidirectional interactions between neurons and malignant glial cells [4]. The central nervous system in adults maintains normal function supporting cognitive operations through cellular level neuron-to-neuron and neuron-to-glia interactions, which collectively establish local synaptic circuits and distributive cognitive networks. Therefore, while adaptive neural plasticity maintains healthy circuit dynamics through synaptic and cellular mechanisms of plasticity, these same processes may influence glioma behavior. Emerging evidence in this field may lead to new therapeutic strategies for patients. Experimental models of disease range from single cell electrophysiology of neoplastic cells and neurons to invasive and non-invasive neuronal circuit interrogation. Reviews to date on this topic focus predominantly on cellular level evidence. The aim of this study is to highlight the evidence supporting activity dependent mechanisms of glioma proliferation including both cellular and synaptic network level investigations.
Methods
Literature search was performed on the Medline electronic database between 2001 and 2023. The keywords used were “glioma”, “glioblastoma”, “circuit remodeling”, “plasticity”, “neuron networks” and “cortical networks”. The references of selected articles were also examined to identify additional studies and included studies focused on WHO grades 2–4, diffuse midline gliomas, and diffuse intrinsic pontine gliomas.
Results
Cellular level evidence of neuronal regulation of glioma proliferation and invasion
In vitro and in vivo models offer the opportunity to understand the complex bidirectional interactions between neurons and malignant glial cells, detailed in Fig. 1, within the tumour microenvironment, at the cellular and molecular levels (Table 1).
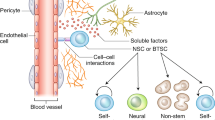
Simplified representation of the interactions between tumours cells and the bidirectional crosstalk between neurons and tumour cells (created with Biorender). A. Interactions between tumour cells. Tumor cells (green) form an extensive network, mediated by tumor microtubes that bear Connexin 43 gap-junctions, and are resistant to radio-chemotherapy. Within the network, pacemaker-like cells, thanks to KCa 3.1 channels (yellow), display rhythmic Ca2+ oscillations that are transmitted to the network and that drive tumour aggressiveness via activation of the MAPK and NF-κB pathways. Unconnected cells have increased invasion abilities. B. Neuronal activity-regulated tumour growth. First, neurons (purple) emit a paracrine signaling, notably mediated by BDNF and NLGN3, that stimulates tumour growth. NLGN3 is clived from neural cells by the ADAM10 sheddase. NLGN3 activates the PI3K-mTOR pathway and feedforwards its expression. Second, through excitatory glutamate neuro-gliomal synapses, whose establishment is eased by NLGN3, neurons activity stimulates tumors growth. BDNF binding to NTRK2 receptors increases the expression of AMPA receptors (red), which strengthen glutamate signaling. The neuron-dependent secretion of TSP1 by tumour cells contributes to tumour microtube formation and glioma progression. C. Glioma-induced neuronal activity modifications. Glutamate is released by glioma cells through the cystine/glutamate antiporter xc− (brown). Peritumoral reactive astrocytes have a decreased capability to uptake glutamate. First, the increased glutamate rate in the tumour microenvironment induces neuronal hyperexcitability. Second, glutamate-toxicity leads to the death of fast-spiking GABAergic inhibitory neurons. Third, the drop in the neuronal expression of the potassium/chloride transporter KCC2 (dark red) is responsible for the switch from inhibitory to excitatory of GABA signaling. Fourth, the disruption of perineuronal nets by glioma-secreted matrix metalloproteinases amplifies the loss of GABAergic inhibition and glutamate-induced neuronal death
Glioma-glioma networks promote glioma growth and treatment resistance
Malignant astrocytoma mouse xenografted tumors analyzed with in vivo multiphoton laser-scanning microscopy uncovered the presence of Tumour Microtubes (TM), that correspond to ultra-long membrane protrusions, stabilized by p120 catenin. Through TM, glioma cells form an extensive and non-randomly organized network mediated by Connexin 43 gap junctions [21] and exchange ions and molecules. The neuronal growth-associated protein GAP-43 conditions TM formation and function and, in concert with the membrane protein linked to neuronal development TTYH1, drives TM-mediated invasion properties and proliferation [5, 6, 22, 23]. TM also establish connections between tumor cells and non-tumoral astrocytes [7]. NOTCH1 regulates network connectivity and its downregulation induces TM extension [8]. A subpopulation of highly connected pacemaker-like glioma cells display rhythmic Ca2+ oscillations, relying on the calcium-dependent potassic channel KCa3.1. These Ca2+ oscillations are transmitted to the network and activate the frequency-dependent MAPK and NF-κB pathways [6, 24]. TM are involved in treatment failures. First, they ease repopulation of the surgical cavity after glioma resection [9]. Second, connected cells are less susceptible to radiation-induced [5] and chemotherapy-induced cytotoxicity than unconnected cells [9], but have however decreased invasion abilities [5, 7, 8, 23]. Connected and unconnected cells express a mesenchymal-like and a neural/oligodendroglial precursor-like signature, respectively [7].
Pharmacological inhibition of Connexin 43 or gap junction but also meclofenamate inhibition of intercellular cytosolic traffic via gap junctions reduced glioblastoma cell resistance to Temozolomide and Lomustine, independently of MGMT status [25,26,27]. Consistently, the genetic inactivation of Connexin 43 or GAP-43 and the senicapoc pharmacological inhibitor of potassic channel KCa3.1 slowed down xenograft progression in mice [5, 6, 9]. TTYH1 knockdown decreased the rate of invasive tumour cells harboring one or two TM but not the rate of hyperconnected cells harboring more than four TM, unveiling a functional and molecular heterogeneity among TM [23]. Consequently, TM formation and function could offer new therapeutic targets.
Neuronal activity drives malignant glioma growth
Through the use of in vivo optogenetic control of cortical neuronal activity in patient-derived pediatric glioblastoma xenograft models in mice expressing the excitatory opsin channelrhodopsin-2, it was demonstrated that neurons stimulate glioma growth through paracrine signaling mediated by Brain-Derived Neurotrophic Factor (BDNF) and the soluble synaptic adhesion protein neuroligin-3 (NLGN3) [10,11,12]. NLGN3 promotes tumour growth and feedforwards its own expression in glioma cells, through induction of the PI3K-mTOR pathway activity. Accordingly, NLGN3 expression is inversely correlated with overall survival [11] and conditions the growth of many subtypes of pediatric and adult gliomas [12]. NLGN3 is cleaved from neurons and oligodendrocytes precursor cells by the ADAM10 shedddase. ADAM10 inhibition reduces xenograft growth by preventing NLGN3 release into the tumour microenvironment and could represent the basis of new therapeutic strategies [12]. The relevance of INCB7839, an ADAM 10 and 17 inhibitor, for the treatment of pediatric high-grade gliomas is assessed in an ongoing randomized clinical trial (NTC04295759).
NLGN3 paracrine signaling upregulates the expression of several synapse-related genes [12], and promotes the establishment of aberrant synapses, localized on TM. The electrochemical communications between presynaptic neurons and postsynaptic malignant glial cells is mediated by glutamatergic α-amino-3-hydroxy-5-methyl-4-isoxazole propionic acid receptors (AMPARs) [10, 14]. AMPAR trafficking to the glioma cell membrane is promoted by docking of BDNF to the receptor NTRK2, resulting in an increased amplitude of glutamate-evoked currents in the malignant cells. Pharmacological or genetical inhibition of NTRK2 decreases synaptogenic mechanisms and prolongs survival in xenograft models [15]. A high-neural epigenetic signature associated to overexpression of synaptic genes was consistently correlated with increased connectivity according to functional Magnetic Resonance Imaging (fMRI) and Magneto-Electro-Encephalography (MEG) [28]. Additionally, the tumor cell subpopulation which overexpresses synaptic genes is mainly of oligodendroglial or neural progenitor-like signature [10, 28, 29] but the link between synaptic transmission and stemness phenotype still remains to be elucidated.
Neuronal activity evokes excitatory post-synaptic currents but also non-synaptic activity-dependent currents, respectively calcium- and potassium-mediated, that are amplified by gap junction-mediated tumour interconnections through TM, forming an electrically coupled network [10, 14]. Synaptic and electrical integration of glioma into neural circuits, and glutamate binding to AMPARs favor tumour progression. Indeed, neuronal activity drives TM dynamics and increases the number of TM branching events via calcium signaling, but also the invasion speed of unconnected glioma cells via transient neuro-gliomal synapses with AMPARs [7, 14, 30]. Moreover, depolarization of glioma cell membranes promotes proliferation [10]. Consistently, the occurrence of gliomas is higher in brain regions which display higher intrinsic activity levels according to MEG [31]. Furthermore, high-functionally connected tumour regions are enriched in a tumor cell subpopulation with synaptogenic properties, which develops TM and proliferates on the presence of neurons, under the dependence of glioma-secreted Thrombospondin 1 (TSP1). Interestingly, the presence of high-functional connectivity areas within tumour negatively influence cognitive and survival prognosis [13]. However, remote activity-dependent glioma progression mechanisms were recently identified. They are driven by an infiltrating cell population highly-expressing axon guidance genes, notably SEMA4F, and whose activity depends on contralateral callosal projection neurons [32].
Cytostatic effects are observed following pharmacological electrochemical signaling blockade [33], using the AMPAR-blocking drug perampanel, meclofenamate [10, 14] or using gabapentin which both inhibit TSP1 and the branched-chain amino acid aminotransferase (BCAT1) thus lowering extracellular glutamate level [13, 34]. The anti-tumoral effects of glutamate signaling inhibitors, including gabapentin, meclofenamate and perampanel, are assessed in several on-going trials [16, 35, 36].
Malignant glioma proliferation promotes neuronal hyperexcitability
Electroencephalograms performed in glioma murine models demonstrate spontaneous and recurring abnormal events, compatible with progressive epileptic activity, with a concurrent enrichment in a tumor cell population expressing synaptic genes, suggesting propagation of synapse associated genes [17]. Cortical slices following malignant glioma implantation demonstrate increased glutamate release from the tumor mass, mediated by the cystine/glutamate antiporter xc− [17, 18, 30], whose expression is anticorrelated to this of p53 [19]. Moreover, peritumoral reactive astrocytes have a decreased ability to uptake glutamate and potassium. Some peritumoral astrocytes also display a depolarized resting membrane potential, further contributing to alter potassium and glutamate homeostasis [20]. A resultant glutamatergic epileptiform hyperexcitability spreading into peritumoral areas was identified by extracellular field recordings at sites distant from glioma [17], consistently with the results of human intraoperative ECoG recordings [10]. Extracellular field recordings performed on brain slices taken from xenograft models revealed that the onset latency of magnesium-free-induced epileptiform activity was shorter than in healthy slices. Moreover, the incidence of ictal-like events was higher. Blockade of the cystine/glutamate antiporter xc− and thus glutamate release from the tumour mass using sulfasalazine decreased hyperexcitability and was able to reduce not only the frequency of epileptic events in tumour-bearing mice [17] but also neuronal activity-regulated glioma growth [37].
Independently, peritumoral cortex taken from mice orthotopic xenografts also displays a distance-dependent loss of parvalbumin-positive fast-spiking GABAergic inhibitory interneurons, attributable to glutamate neurotoxicity, and a reduced neuronal expression of the potassium/chloride plasmalemmal transporter KCC2, changing response to GABA from inhibitory to excitatory potentials [33, 38, 39]. Additionally, perineuronal nets, which represent a complex lattice-like extracellular matrix, acting as an electrostatic insulator that reduces specific membrane capacitance, are degraded in a distance-dependent manner by glioma-released matrix metalloproteinases. This mechanism amplifies the loss of GABAergic inhibition but also glutamate-induced neuronal death [39].
Taken together, these results demonstrate that hyperexcitability results from peritumoral synaptic network disruption in the setting of malignant gliomas [20, 37, 38] and suggest new therapeutic targets for controlling peritumoral hyperexcitability. Interestingly, in a mouse model, it was evidenced that some hotspot-mutations of PI3KCA induced increased network hyperexcitability compared to others. Notably, neurons adjacent to C420R- and H1047R-mutant gliomas displayed enhanced synaptic imbalance, which was attributed to the secretion of glypican 3 in the case of C420R variant [40], bridging genetics with peritumoral synaptic remodeling.
Network level evidence of nervous system regulation of glioma proliferation and invasion
Resting state and task-based investigations are available to indirectly and macroscopically probe functional plasticity mechanisms and circuit remodeling caused by malignant gliomas (detailed in Fig. 2 and Table 2). Network level understanding of glioma development has recently come into clearer view because of emerging technologies.
Functional MRI (fMRI)
Four different glioma-induced remodeling patterns in patients have been identified using fMRI with the appearance of additional activation sites within (1) the tumor, (2) the ipsilateral peritumoral cortex, (3) the distant ipsilateral cortex, and (4) the contralateral normal appearing cortex [41]. Indeed, in a series of patients with gliomas located at an average distance of 3.9 ± 3.5 cm from the hand motor region, 62% and 46% of patients exhibited an ipsilateral and contralateral recruitment, respectively. Ipsilateral recruitment decreased as tumour volume increased and distance from primary motor cortex decreased and vice versa [42]. However, patients with gliomas located in or near motor areas, but without any motor deficit, had a significant reduction in inter-hemispheric functional connectivity between bilateral primary motor cortices, compared to age-matched healthy controls [43]. Regarding the language network, right-handed patients with a left hemisphere glioma also had a global reduction of bilateral functional connectivity, compared to healthy controls. The most affected node was the left temporo-parietal junction [44].
According to resting-state fMRI, compared to healthy controls, patients with glioma had a decreased functional connectivity concerning the whole-brain, and not restricted to the lesional hemisphere [45, 46]. The importance of these alterations was correlated with high tumor grade, negative IDH status and decreased neuropsychological performances but not with tumour size or location [45]. Besides, Default Mode Network connectivity was modified in patients managed for left-sided gliomas, with increased and decreased integration in hippocampal and prefrontal areas, respectively [47]. Finally, a higher intra-network functional connectivity strength within glioblastoma was found to be independent of tumour size but predicted a better overall survival [48].
Positron Emission Tomography (PET)
One of the first studies demonstrating remodeled network connectivity caused by chronic disease of the central nervous system was identified using PET imaging of amyloid beta in patients with Alzheimer’s disease [49]. PET of gliomas located in the hand motor region showed that, compared to the unaffected side, the activations were shifted by 20 ± 13 mm (SD), either along the mediolateral body representation of motor cortex or into premotor or parietal somatosensory cortex [50]. Additional activation of the supplementary motor area was occasionally present [50]. Regarding the plasticity of the language network, two compensatory mechanisms, whose occurrence depended on tumour location, were identified. First, at the intra-hemispheric level, left fronto-lateral regions other than classical language areas can be recruited. Second, at the inter-hemispheric level, fronto-lateral activation can appear in the right nondominant hemisphere, especially in patients with frontal or temporal posterior tumours, possibly representing a loss of transcallosal collateral inhibition [51].
MagnetoEncephaloGraphy (MEG)
The comparison of whole-brain activation motor maps performed in the same patients at initial diagnosis and compared with first recurrence has demonstrated a shift in activation peaks in the ipsilateral and contralateral motor cortices [52]. Specifically, motor activity following glioma progression is associated with contralesional hemisphere activation for speech and motor tasks. Tumor location, presence of a motor impairment and longer time lapse were associated with greater cortical remodeling [52]. The same methodology applied to language mapping highlighted a shift in language laterality index in about 30% of patients. Magnitude and relative direction of the shift depended on tumor location and initial language dominance, as shift was greater in patients with increased lateralization compared to those with bilateral representation [53]. However, these studies not only reflected glioma-induced but also surgical-induced plasticity mechanisms.
Electrocorticography (ECoG)
In awake patients with malignant gliomas, at resting state and during speech, an increased high-gamma band range power is detectable outside of the necrotic tumour core, in glioma-infiltrated brain compared to healthy-appearing brain, consistent with cortical hyperexcitability [10, 13]. Furthermore, intraoperative subdural local field potentials using ECoG recordings before awake resection of dominant hemisphere malignant gliomas proved that glioma-infiltrated cortex is able to engage in synchronous activity during task performance, similarly to normal-appearing cortex, but recruits a widened spatial network [13, 54], as observed with imaging techniques [51]. Glioma-infiltrated cortex has decreased entropy compared to normal-appearing cortex and may therefore be less efficient to encode information during nuanced tasks as production of monosyllabic and polysyllabic words. Consistently, in glioma-infiltered cortex, signals corresponding to monosyllabic and polysyllabic words were indistinguishable, conversely to those arising from normal-appearing cortex [54].
Diffusion Tensor Imaging (DTI)
Tumor invasion may interact with white-matter tracts in three distinct patterns: infiltration, disruption, and displacement [41]. In patients with glioma, DTI sequences demonstrate a global decreased in fractional anisotropy and decreased axial, mean and radial diffusivity in the ipsilateral hemisphere, compared to the contralateral hemisphere [55]. Interestingly, preoperative tracking in patients with a left-sided glioma highlighted that patients with symmetric or right-lateralized posterior segment of the arcuate fasciculi had no language impairments. These data suggests that right homologs of structural language-associated pathways at the subcortical level could be supportive for language functions [56]. In clinical practice, glioma-network interrogation using DTI has prognostic significance for patients in whom awake resection is indicated, and to help in surgical planning [57].The data presented above suggest that glioma induced network remodeling is influenced by tumour biology which ultimately influences onco-functional outcomes.
Navigated Transcranial Magnetic Stimulation
Whereas direct electrostimulation is the gold standard method to detect functional areas, navigated Transcranial Magnetic Stimulation (nTMS) represents a unique non-invasive technique. As imaging techniques, nTMS objectivated an ipsilateral recruitment with a spread of motor areas in the post-central gyrus but also in the superior and middle frontal gyri, depending on tumor location compared to motor cortex [58]. Two studies assessed motor cortex remodeling before and after glioma resection, with the same limitations as studies previously described [52, 53]. The centers of gravity were shifted from 12.3 ± 14.3 mm [59], 4.6 ± 0.8 mm on the mediolateral axis, and 8.7 ± 1.5 mm on the anteroposterior axis [60]. Complete postoperative motor recovery was observed exclusively in patients with cortical remodeling [59]. nTMS targeting the right hemisphere induced language disturbances in right-handed patients with left-sided gliomas but not in healthy controls, confirming an underlying bilateral remodeling of the language network [57].
Direct Electrostimulation
Causal evidence supporting glioma-induced cortical function remodeling has been demonstrated using direct electrical stimulation mapping. In patients who underwent repeated stimulation mappings during glioma resection, of 22 initially identified eloquent sites, 13 (59.1%) remained positive, while 9 (40.9%) had become negative although no neurological impairment was noted. These findings suggest that neurological function may be preserved through neural circuit remodeling or activation of latent functional pathways. Patients in who cortical function was lost were noted to have a smaller tumour volume compared to other patients but there were no differences regarding demographical, pathological or treatment-related features [61]. In another series of 42 patients who underwent repeated cortical and subcortical mappings during the awake resection of a glioma, patients with high-level plasticity (displacement of ≥ 2 eloquent sites) and low-level plasticity (displacement < 2 eloquent sites) were distinguished. In the high-level plasticity group, various rates of eloquent sites were gained or lost, including displacement of primary motor sites [62]. While initially thought to be static, cortical plasticity of primary sensorimotor sites has been reported with gained or lost function without corresponding neurologic impairments in 32/51 (62.7%) of stimulated sites [63]. These data refine the current understanding of glioma-induced modifications in neurocognitive processing and could guide new strategies including neuromodulation [54].
Conclusion
Whereas functional imaging and per-operative acquisitions have defined patterns of malignant circuit remodeling at a macroscopic level, various in vitro and animal models have deciphered the complex bidirectional crosstalk between neurons and glioma cells at a microscopic level, leading to the emergence of new potential therapeutic targets. Future challenges will be to better understand how tumor genetics modulate neuron-glioma interactions and to integrate these data to design new therapeutic options likely to improve both oncological and functional outcomes.
Data Availability
No datasets were generated or analysed during the current study.
References
Louis DN, Perry A, Wesseling P et al (2021) The 2021 WHO Classification of Tumors of the Central Nervous System: a summary. Neuro Oncol 23:1231–1251. https://doi.org/10.1093/neuonc/noab106
Molinaro AM, Taylor JW, Wiencke JK, Wrensch MR (2019) Genetic and molecular epidemiology of adult diffuse glioma. Nat Rev Neurol 15:405–417. https://doi.org/10.1038/s41582-019-0220-2
Nishikawa R, Yamasaki F, Arakawa Y et al (2023) Safety and efficacy of tumour-treating fields (TTFields) therapy for newly diagnosed glioblastoma in Japanese patients using the Novo-TTF System: a prospective post-approval study. Jpn J Clin Oncol 53:371–377. https://doi.org/10.1093/jjco/hyad001
Winkler F, Venkatesh HS, Amit M et al (2023) Cancer neuroscience: State of the field, emerging directions. Cell 186:1689–1707. https://doi.org/10.1016/j.cell.2023.02.002
Osswald M, Jung E, Sahm F et al (2015) Brain tumour cells interconnect to a functional and resistant network. Nature 528:93–98. https://doi.org/10.1038/nature16071
Hausmann D, Hoffmann DC, Venkataramani V et al (2023) Autonomous rhythmic activity in glioma networks drives brain tumour growth. Nature 613:179–186. https://doi.org/10.1038/s41586-022-05520-4
Venkataramani V, Yang Y, Schubert MC et al (2022) Glioblastoma hijacks neuronal mechanisms for brain invasion. Cell 185:2899-2917.e31. https://doi.org/10.1016/j.cell.2022.06.054
Jung E, Osswald M, Ratliff M et al (2021) Tumor cell plasticity, heterogeneity, and resistance in crucial microenvironmental niches in glioma. Nat Commun 12:1014. https://doi.org/10.1038/s41467-021-21117-3
Weil S, Osswald M, Solecki G et al (2017) Tumor microtubes convey resistance to surgical lesions and chemotherapy in gliomas. Neuro Oncol 19:1316–1326. https://doi.org/10.1093/neuonc/nox070
Venkatesh HS, Morishita W, Geraghty AC et al (2019) Electrical and synaptic integration of glioma into neural circuits. Nature 573:539–545. https://doi.org/10.1038/s41586-019-1563-y
Venkatesh HS, Johung TB, Caretti V et al (2015) Neuronal Activity Promotes Glioma Growth through Neuroligin-3 Secretion. Cell 161:803–816. https://doi.org/10.1016/j.cell.2015.04.012
Venkatesh HS, Tam LT, Woo PJ et al (2017) Targeting neuronal activity-regulated neuroligin-3 dependency in high-grade glioma. Nature 549:533–537. https://doi.org/10.1038/nature24014
Krishna S, Choudhury A, Keough MB, et al (2023) Glioblastoma remodelling of human neural circuits decreases survival. Nature 1–9. https://doi.org/10.1038/s41586-023-06036-1
Venkataramani V, Tanev DI, Strahle C et al (2019) Glutamatergic synaptic input to glioma cells drives brain tumour progression. Nature 573:532–538. https://doi.org/10.1038/s41586-019-1564-x
Taylor KR, Barron T, Hui A et al (2023) Glioma synapses recruit mechanisms of adaptive plasticity. Nature 623:366–374. https://doi.org/10.1038/s41586-023-06678-1
Mastall M, Roth P, Bink A et al (2024) A phase Ib/II randomized, open-label drug repurposing trial of glutamate signaling inhibitors in combination with chemoradiotherapy in patients with newly diagnosed glioblastoma: the GLUGLIO trial protocol. BMC Cancer 24:82. https://doi.org/10.1186/s12885-023-11797-z
Buckingham SC, Campbell SL, Haas BR et al (2011) Glutamate release by primary brain tumors induces epileptic activity. Nat Med 17:1269–1274. https://doi.org/10.1038/nm.2453
Ye ZC, Sontheimer H (1999) Glioma cells release excitotoxic concentrations of glutamate. Cancer Res 59:4383–4391
Umans RA, Martin J, Harrigan ME et al (2021) Transcriptional Regulation of Amino Acid Transport in Glioblastoma Multiforme. Cancers (Basel) 13:6169. https://doi.org/10.3390/cancers13246169
Campbell SC, Muñoz-Ballester C, Chaunsali L et al (2020) Potassium and glutamate transport is impaired in scar-forming tumor-associated astrocytes. Neurochem Int 133:104628. https://doi.org/10.1016/j.neuint.2019.104628
Lin JHC, Takano T, Cotrina ML et al (2002) Connexin 43 enhances the adhesivity and mediates the invasion of malignant glioma cells. J Neurosci 22:4302–4311. https://doi.org/10.1523/JNEUROSCI.22-11-04302.2002
Gritsenko PG, Atlasy N, Dieteren CEJ et al (2020) p120-catenin-dependent collective brain infiltration by glioma cell networks. Nat Cell Biol 22:97–107. https://doi.org/10.1038/s41556-019-0443-x
Jung E, Osswald M, Blaes J et al (2017) Tweety-Homolog 1 Drives Brain Colonization of Gliomas. J Neurosci 37:6837–6850. https://doi.org/10.1523/JNEUROSCI.3532-16.2017
Weaver AK, Bomben VC, Sontheimer H (2006) Expression and function of calcium-activated potassium channels in human glioma cells. Glia 54:223–233. https://doi.org/10.1002/glia.20364
Murphy SF, Varghese RT, Lamouille S et al (2016) Connexin 43 Inhibition Sensitizes Chemoresistant Glioblastoma Cells to Temozolomide. Cancer Res 76:139–149. https://doi.org/10.1158/0008-5472.CAN-15-1286
Potthoff A-L, Heiland DH, Evert BO et al (2019) Inhibition of Gap Junctions Sensitizes Primary Glioblastoma Cells for Temozolomide. Cancers (Basel) 11:858. https://doi.org/10.3390/cancers11060858
Schneider M, Potthoff A-L, Evert BO et al (2021) Inhibition of Intercellular Cytosolic Traffic via Gap Junctions Reinforces Lomustine-Induced Toxicity in Glioblastoma Independent of MGMT Promoter Methylation Status. Pharmaceuticals (Basel) 14:195. https://doi.org/10.3390/ph14030195
Drexler R, Khatri R, Sauvigny T, et al (2023) Epigenetic neural glioblastoma enhances synaptic integration and predicts therapeutic vulnerability. bioRxiv 2023.08.04.552017. https://doi.org/10.1101/2023.08.04.552017
Filbin MG, Tirosh I, Hovestadt V et al (2018) Developmental and oncogenic programs in H3K27M gliomas dissected by single-cell RNA-seq. Science 360:331–335. https://doi.org/10.1126/science.aao4750
Takano T, Lin JH, Arcuino G et al (2001) Glutamate release promotes growth of malignant gliomas. Nat Med 7:1010–1015. https://doi.org/10.1038/nm0901-1010
Numan T, Breedt LC, de Maciel B, APC, et al (2022) Regional healthy brain activity, glioma occurrence and symptomatology. Brain 145:3654–3665. https://doi.org/10.1093/brain/awac180
Huang-Hobbs E, Cheng Y-T, Ko Y et al (2023) Remote neuronal activity drives glioma progression through SEMA4F. Nature 619:844–850. https://doi.org/10.1038/s41586-023-06267-2
Soroceanu L, Manning TJ, Sontheimer H (1999) Modulation of glioma cell migration and invasion using Cl(-) and K(+) ion channel blockers. J Neurosci 19:5942–5954. https://doi.org/10.1523/JNEUROSCI.19-14-05942.1999
Tönjes M, Barbus S, Park YJ et al (2013) BCAT1 promotes cell proliferation through amino acid catabolism in gliomas carrying wild-type IDH1. Nat Med 19:901–908. https://doi.org/10.1038/nm.3217
Zeyen T, Potthoff A-L, Nemeth R et al (2022) Phase I/II trial of meclofenamate in progressive MGMT-methylated glioblastoma under temozolomide second-line therapy-the MecMeth/NOA-24 trial. Trials 23:57. https://doi.org/10.1186/s13063-021-05977-0
Heuer S, Burghaus I, Gose M et al (2024) PerSurge (NOA-30) phase II trial of perampanel treatment around surgery in patients with progressive glioblastoma. BMC Cancer 24:135. https://doi.org/10.1186/s12885-024-11846-1
Campbell SL, Buckingham SC, Sontheimer H (2012) Human glioma cells induce hyperexcitability in cortical networks. Epilepsia 53:1360–1370. https://doi.org/10.1111/j.1528-1167.2012.03557.x
Campbell SL, Robel S, Cuddapah VA et al (2015) GABAergic disinhibition and impaired KCC2 cotransporter activity underlie tumor-associated epilepsy. Glia 63:23–36. https://doi.org/10.1002/glia.22730
Tewari BP, Chaunsali L, Campbell SL et al (2018) Perineuronal nets decrease membrane capacitance of peritumoral fast spiking interneurons in a model of epilepsy. Nat Commun 9:4724. https://doi.org/10.1038/s41467-018-07113-0
Yu K, Lin C-CJ, Hatcher A et al (2020) PIK3CA variants selectively initiate brain hyperactivity during gliomagenesis. Nature 578:166–171. https://doi.org/10.1038/s41586-020-1952-2
Krishna S, Kakaizada S, Almeida N et al (2021) Central Nervous System Plasticity Influences Language and Cognitive Recovery in Adult Glioma. Neurosurgery 89:539–548. https://doi.org/10.1093/neuros/nyaa456
Kong NW, Gibb WR, Badhe S et al (2020) Plasticity of the Primary Motor Cortex in Patients with Primary Brain Tumors. Neural Plast 2020:3648517. https://doi.org/10.1155/2020/3648517
Niu C, Zhang M, Min Z et al (2014) Motor network plasticity and low-frequency oscillations abnormalities in patients with brain gliomas: a functional MRI study. PLoS ONE 9:e96850. https://doi.org/10.1371/journal.pone.0096850
Briganti C, Sestieri C, Mattei PA et al (2012) Reorganization of functional connectivity of the language network in patients with brain gliomas. AJNR Am J Neuroradiol 33:1983–1990. https://doi.org/10.3174/ajnr.A3064
Stoecklein VM, Stoecklein S, Galiè F et al (2020) Resting-state fMRI detects alterations in whole brain connectivity related to tumor biology in glioma patients. Neuro Oncol 22:1388–1398. https://doi.org/10.1093/neuonc/noaa044
Park JE, Kim HS, Kim SJ et al (2016) Alteration of long-distance functional connectivity and network topology in patients with supratentorial gliomas. Neuroradiology 58:311–320. https://doi.org/10.1007/s00234-015-1621-6
Esposito R, Mattei PA, Briganti C et al (2012) Modifications of default-mode network connectivity in patients with cerebral glioma. PLoS ONE 7:e40231. https://doi.org/10.1371/journal.pone.0040231
Daniel AGS, Park KY, Roland JL et al (2021) Functional connectivity within glioblastoma impacts overall survival. Neuro Oncol 23:412–421. https://doi.org/10.1093/neuonc/noaa189
Buckner RL, Snyder AZ, Shannon BJ et al (2005) Molecular, structural, and functional characterization of Alzheimer’s disease: evidence for a relationship between default activity, amyloid, and memory. J Neurosci 25:7709–7717. https://doi.org/10.1523/JNEUROSCI.2177-05.2005
Wunderlich G, Knorr U, Herzog H et al (1998) Precentral glioma location determines the displacement of cortical hand representation. Neurosurgery 42:18–26. https://doi.org/10.1097/00006123-199801000-00005
Thiel A, Herholz K, Koyuncu A et al (2001) Plasticity of language networks in patients with brain tumors: a positron emission tomography activation study. Ann Neurol 50:620–629. https://doi.org/10.1002/ana.1253
Bulubas L, Sardesh N, Traut T et al (2020) Motor Cortical Network Plasticity in Patients With Recurrent Brain Tumors. Front Hum Neurosci 14:118. https://doi.org/10.3389/fnhum.2020.00118
Traut T, Sardesh N, Bulubas L et al (2019) MEG imaging of recurrent gliomas reveals functional plasticity of hemispheric language specialization. Hum Brain Mapp 40:1082–1092. https://doi.org/10.1002/hbm.24430
Aabedi AA, Lipkin B, Kaur J et al (2021) Functional alterations in cortical processing of speech in glioma-infiltrated cortex. Proc Natl Acad Sci U S A 118:e2108959118. https://doi.org/10.1073/pnas.2108959118
Ormond DR, D’Souza S, Thompson JA (2017) Global and Targeted Pathway Impact of Gliomas on White Matter Integrity Based on Lobar Localization. Cureus 9:e1660. https://doi.org/10.7759/cureus.1660
Jehna M, Becker J, Zaar K et al (2017) Symmetry of the arcuate fasciculus and its impact on language performance of patients with brain tumors in the language-dominant hemisphere. J Neurosurg 127:1407–1416. https://doi.org/10.3171/2016.9.JNS161281
Rösler J, Niraula B, Strack V et al (2014) Language mapping in healthy volunteers and brain tumor patients with a novel navigated TMS system: evidence of tumor-induced plasticity. Clin Neurophysiol 125:526–536. https://doi.org/10.1016/j.clinph.2013.08.015
Bulubas L, Sabih J, Wohlschlaeger A et al (2016) Motor areas of the frontal cortex in patients with motor eloquent brain lesions. J Neurosurg 125:1431–1442. https://doi.org/10.3171/2015.11.JNS152103
Barz A, Noack A, Baumgarten P et al (2018) Motor Cortex Reorganization in Patients with Glioma Assessed by Repeated Navigated Transcranial Magnetic Stimulation-A Longitudinal Study. World Neurosurg 112:e442–e453. https://doi.org/10.1016/j.wneu.2018.01.059
Conway N, Wildschuetz N, Moser T et al (2017) Cortical plasticity of motor-eloquent areas measured by navigated transcranial magnetic stimulation in patients with glioma. J Neurosurg 127:981–991. https://doi.org/10.3171/2016.9.JNS161595
Southwell DG, Hervey-Jumper SL, Perry DW, Berger MS (2016) Intraoperative mapping during repeat awake craniotomy reveals the functional plasticity of adult cortex. J Neurosurg 124:1460–1469. https://doi.org/10.3171/2015.5.JNS142833
Picart T, Herbet G, Moritz-Gasser S, Duffau H (2019) Iterative Surgical Resections of Diffuse Glioma With Awake Mapping: How to Deal With Cortical Plasticity and Connectomal Constraints? Neurosurgery 85:105–116. https://doi.org/10.1093/neuros/nyy218
Gibb WR, Kong NW, Tate MC (2020) Direct Evidence of Plasticity within Human Primary Motor and Somatosensory Cortices of Patients with Glioblastoma. Neural Plast 2020:8893708. https://doi.org/10.1155/2020/8893708
Acknowledgements
Dr Thiébaud Picart thanks the Hospices Civils de Lyon (France), the Ligue Nationale Contre le Cancer (France), the Philippe Foundation and the French Society of Neurosurgery (SFNC) for their financial support. Dr Shawn Hervey-Jumper is supported by NIH grants K08NS110919 and P50CA097257 for the completion of this work.
Funding
No specific funding was received for this review.
Author information
Authors and Affiliations
Contributions
Conceptualization, S.H.J and T.P—Original Draft Preparation, T.P. – Writing, Review and Editing, S.H.J. All authors have read and agreed to the published version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Picart, T., Hervey-Jumper, S. Central nervous system regulation of diffuse glioma growth and invasion: from single unit physiology to circuit remodeling. J Neurooncol 169, 1–10 (2024). https://doi.org/10.1007/s11060-024-04719-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-024-04719-x