Abstract
Purpose
Glioma is a life-threatening malignancy where conventional therapies are ineffective. Bacterial cancer therapy has shown potential for glioma treatment, in particular, the facultative anaerobe Salmonella has been extensively studied. Meanwhile, ferroptosis is a newly characterized form of cell death. Nevertheless, the role of ferroptosis in Salmonella-induced tumour cell death remains unclear. Therefore, we aim to elucidate whether Salmonella YB1 exerts therapeutic effects via inducing ferroptosis in glioma.
Methods
Following Salmonella YB1 infection, mRNA sequencing was applied to detect ferroptosis-related gene expression and the levels of reactive oxygen species, malondialdehyde, and glutathione were quantified. Transmission electron microscopy (TEM) was then used to observe the changes in the mitochondrial morphology of glioma cells. The role of ferroptosis in the anti-tumor effect of YB1 was assessed in vivo in mouse tumor xenograft models.
Results
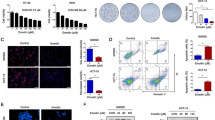
Whole-transcriptome analysis revealed that Salmonella YB1 infection alters ferroptosis-related gene expression in the U87 glioma cell line. Moreover, we found that Salmonella-induced ferroptosis is correlated with reduced levels of glutathione and glutathione peroxidase-4 (GPX4) and increased levels of reactive oxygen species and malondialdehyde in vitro. Meanwhile, TEM revealed that mitochondria are shrunken and mitochondrial membrane density increases in infected glioma cells. Experiments in vivo further showed that tumor growth in the Salmonella-treated group was significantly slower compared to the control and Fer-1 groups. However, Salmonella-induced tumor suppression can be reversed in vivo by Fer-1 treatment.
Conclusion
Salmonella YB1 inhibits GPX4 expression and induces ferroptosis to suppress glioma growth. Hence, ferroptosis regulation might represent a promising strategy to improve the efficacy of bacterial cancer therapy.
Graphical abstract








Similar content being viewed by others
Data availability
The raw sequence and processed data were deposited in the NCBI’s Sequence Read Archive (SRA) with accession number PRJNA939824. The data presented in this study are available on request from the corresponding author.
References
Wick W, Platten M, Weller M (2009) New (alternative) temozolomide regimens for the treatment of glioma. Neuro Oncol 11:69–79. https://doi.org/10.1215/15228517-2008-078
Chen JMRPL (2012) Malignant glioma: lessons from genomics, mouse models, and stem cells. Cell 1:36–47. https://doi.org/10.1016/j.cell.2012.03.009
Huang Q, Chen L, Liang J, Huang Q, Sun H (2022) Neurotransmitters: potential targets in glioblastoma. Cancers 14:3970. https://doi.org/10.3390/cancers14163970
Lu S, Wang XZ, He C, Wang L, Liang SP, Wang CC, Li C, Luo TF, Feng CS, Wang ZC, Chi GF, Ge PF (2021) ATF3 contributes to brucine-triggered glioma cell ferroptosis via promotion of hydrogen peroxide and iron. Acta Pharmacol Sin 42:1690–1702. https://doi.org/10.1038/s41401-021-00700-w
Yuan E, Liu K, Lee J, Tsung K, Chow F, Attenello FJ (2022) Modulating glioblastoma chemotherapy response: evaluating long non-coding RNA effects on DNA damage response, glioma stem cell function, and hypoxic processes. Neuro-Oncol Adv 4:c119. https://doi.org/10.1093/noajnl/vdac119
Yang L, Lin C, Wang L, Guo H, Wang X (2012) Hypoxia and hypoxia-inducible factors in glioblastoma multiforme progression and therapeutic implications. Exp Cell Res 318:2417–2426. https://doi.org/10.1016/j.yexcr.2012.07.017
Legendre C, Garcion E (2015) Iron metabolism: a double-edged sword in the resistance of glioblastoma to therapies. Trends Endocrinol Metab 26:322–331. https://doi.org/10.1016/j.tem.2015.03.008
Wang J, Liu Y, Zhang A, Yu W, Lei Q, Xiao B, Luo Z (2022) Investigational microbiological therapy for glioma. Cancers 14:5977. https://doi.org/10.1016/j.tem.2015.03.008
Wen M, Zheng JH, Choi JM, Pei J, Li C, Li S, Kim I, Lim S, Jung T, Moon K, Min J, Jung S (2018) Genetically-engineered Salmonella typhimurium expressing TIMP-2 as a therapeutic intervention in an orthotopic glioma mouse model. Cancer Lett 433:140–146. https://doi.org/10.1016/j.canlet.2018.06.031
Broekman MLD, Nieland L, Hoeben RC (2022) Virotherapy: the next addition to the standard of care for glioblastoma? Cancer Cell 40:1089–1091. https://doi.org/10.1016/j.ccell.2022.09.003
Wang Z, Ding Y, Wang X, Lu S, Wang C, He C, Wang L, Piao M, Chi G, Luo Y, Ge P (2018) Pseudolaric acid B triggers ferroptosis in glioma cells via activation of Nox4 and inhibition of xCT. Cancer Lett 428:21–33. https://doi.org/10.1016/j.canlet.2018.04.021
Song Q, Peng S, Sun Z, Heng X, Zhu X (2021) Temozolomide drives ferroptosis via a DMT1-dependent pathway in glioblastoma cells. Yonsei Med J 62:84. https://doi.org/10.3349/ymj.2021.62.9.843
Aldrovandi M, Conrad M (2020) Ferroptosis: the good, the bad and the ugly. Cell Res 30:1061–1062. https://doi.org/10.1038/s41422-020-00434-0
Chen Y, Mi Y, Zhang X, Ma Q, Song Y, Zhang L, Wang D, Xing J, Hou B, Li H, Jin H, Du W, Zou Z (2019) Dihydroartemisinin-induced unfolded protein response feedback attenuates ferroptosis via PERK/ATF4/HSPA5 pathway in glioma cells. J Exp Clin Canc Res 38:402. https://doi.org/10.1186/s13046-019-1413-7
Stockwell BR (2022) Ferroptosis turns 10: emerging mechanisms, physiological functions, and therapeutic applications. Cell 185:2401–2421. https://doi.org/10.1016/j.cell.2022.06.003
Jiang X, Stockwell BR, Conrad M (2021) Ferroptosis: mechanisms, biology and role in disease. Nat Rev Mol Cell Biol 22:266–282. https://doi.org/10.1038/s41580-020-00324-8
Zhang L, Li C, Zhang Y, Zhang J, Yang X (2022) Ophiopogonin B induces gastric cancer cell death by blocking the GPX4/xCT-dependent ferroptosis pathway. Oncol Lett 23:104. https://doi.org/10.3892/ol.2022.13224
Tang D, Chen X, Kang R, Kroemer G (2021) Ferroptosis: molecular mechanisms and health implications. Cell Res 31:107–125. https://doi.org/10.1038/s41422-020-00441-1
Zhou S, Gravekamp C, Bermudes D, Liu K (2018) Tumour-targeting bacteria engineered to fight cancer. Nat Rev Cancer 18:727–743. https://doi.org/10.1038/s41568-018-0070-z
Gurbatri CR, Arpaia N, Danino T (2022) Engineering bacteria as interactive cancer therapies. Science 378:858–864. https://doi.org/10.1126/science.add9667
Liang K, Liu Q, Li P, Luo H, Wang H, Kong Q (2019) Genetically engineered Salmonella typhimurium: recent advances in cancer therapy. Cancer Lett 448:168–181. https://doi.org/10.1016/j.canlet.2019.01.037
Guo Y, Chen Y, Liu X, Min J, Tan W, Zheng JH (2020) Targeted cancer immunotherapy with genetically engineered oncolytic Salmonella typhimurium. Cancer Lett 469:102–110. https://doi.org/10.1016/j.canlet.2019.10.033
Yu B, Yang M, Shi L, Yao Y, Jiang Q, Li X, Tang L, Zheng B, Yuen K, Smith DK, Song E, Huang J (2012) Explicit hypoxia targeting with tumor suppression by creating an obligate’’ anaerobic Salmonella typhimurium strain. Sci Rep 2:436. https://doi.org/10.1038/srep00436
Yu B, Shi L, Zhang BZ, Zhang KE, Peng X, Niu HB, Qu JL (2015) Obligate’ anaerobic Salmonella typhimurium strain YB1 treatment on xenograft tumor in immunocompetent mouse model. Oncol Lett 10:1069–1074. https://doi.org/10.3892/ol.2015.3302
Li CX, Yu B, Shi L, Geng W, Lin QB, Ling CC, Yang M, Ng KT, Huang JD, Man K (2017) Obligate’ anaerobic Salmonella strain YB1 suppresses liver tumor growth and metastasis in nude mice. Oncol Lett 13:177–183. https://doi.org/10.3892/ol.2016.5453
Lin Q, Rong L, Jia X, Li R, Yu B, Hu J, Luo X, Badea SR, Xu C, Fu G, Lai K, Lee M, Zhang B, Gong H, Zhou N, Chen XL, Lin S, Fu G, Huang J (2021) IFN-γ-dependent NK cell activation is essential to metastasis suppression by engineered Salmonella. Nat Commun 12:2537. https://doi.org/10.1038/s41467-021-22755-3
Guo Z, Yu B, Ning B, Chan S, Lin Q, Li JC, Huang J, Chan GC (2015) Genetically modified “obligate” anaerobic Salmonella typhimurium as a therapeutic strategy for neuroblastoma. J Hematol Oncol 8:99. https://doi.org/10.1186/s13045-015-0196-3
Wen MJSMK (2014) Targeted cancer immunotherapy with genetically engineered oncolytic Salmonella typhimurium. J Korean Neurosurg Soc 3:131–135. https://doi.org/10.1016/j.canlet.2019.10.033
Mi Z, Yao Q, Qi Y, Zheng J, Liu J, Liu Z, Tan H, Ma X, Zhou W, Rong P (2023) Salmonella-mediated blood–brain barrier penetration, tumor homing and tumor microenvironment regulation for enhanced chemo/bacterial glioma therapy. Acta Pharm Sin B 13:819–833. https://doi.org/10.1016/j.apsb.2022.09.016
Mehta A, Singh S, Ganguly NK (1998) Impairment of intestinal mucosal antioxidant defense system during Salmonella typhimurium infection. Dig Dis Sci 43:646–651. https://doi.org/10.1023/a:1018887813713
Agbor TA, Demma Z, Mrsny RJ, Castillo A, Boll EJ, McCormick BA (2014) The oxido-reductase enzyme glutathione peroxidase 4 (GPX4) governs Salmonella typhimurium-induced neutrophil transepithelial migration. Cell Microbiol 16:1339–1353. https://doi.org/10.1111/cmi.12290
Mehta A, Singh S, Ganguly NK (1998) Role of reactive oxygen species in Salmonella typhimurium-induced enterocyte damage. Scand J Gastroenterol 33:406–414. https://doi.org/10.1080/00365529850171044
Chen X, Kang R, Kroemer G, Tang D (2021) Broadening horizons: the role of ferroptosis in cancer. Nat Rev Clin Oncol 18:280–296. https://doi.org/10.1038/s41571-020-00462-0
Wang D, Wei X, Kalvakolanu DV, Guo B, Zhang L (2021) Perspectives on oncolytic Salmonella in cancer immunotherapy—a promising strategy. Front Immunol 12:615930. https://doi.org/10.3389/fimmu.2021.615930
Li B, He H, Zhang S, Zhao W, Li N, Shao R (2012) Salmonella typhimurium strain SL7207 induces apoptosis and inhibits the growth of HepG2 hepatoma cells in vitro and in vivo. Acta Pharm Sin B 2:562–5680. https://doi.org/10.1016/j.apsb.2012.10.006
Lee CH, Lin ST, Liu JJ, Chang WW, Hsieh JL, Wang WK (2014) Salmonella induce autophagy in melanoma by the downregulation of AKT/mTOR pathway. Gene Ther 21:309–316. https://doi.org/10.1038/gt.2013.86
Lei G, Zhuang L, Gan B (2022) Targeting ferroptosis as a vulnerability in cancer. Nat Rev Cancer 22:381–396. https://doi.org/10.1038/s41568-022-00459-0
Su Y, Zhao B, Zhou L, Zhang Z, Shen Y, Lv H, AlQudsy LHH, Shang P (2020) Ferroptosis, a novel pharmacological mechanism of anti-cancer drugs. Cancer Lett 483:127–136
Zhang C, Liu X, Jin S, Chen Y, Guo R (2022) Ferroptosis in cancer therapy, a novel approach to reversing drug resistance. Mol Cancer 21:47. https://doi.org/10.1016/j.canlet.2020.02.015
Zheng J, Conrad M (2020) The metabolic underpinnings of ferroptosis. Cell Metab 32:920–937. https://doi.org/10.1186/s12943-022-01530-y
Kannan R, Kumar K, Sahal D, Kukreti S, Chauhan VS (2005) Reaction of artemisinin with haemoglobin: implications for antimalarial activity. Biochem J 385:409–418. https://doi.org/10.1016/j.cmet.2020.10.011
Yi R, Wang H, Deng C, Wang X, Yao L, Niu W, Fei M, Zhaba W (2020) Dihydroartemisinin initiates ferroptosis in glioblastoma through GPX4 inhibition. Biosci Rep. https://doi.org/10.1042/BSR20193314
Kupz ACRRB (2014) In vivo IFN-c secretion by NK cells in response to Salmonella typhimurium requires NLRC4 inflammasomes. PLoS ONE 5:e97418. https://doi.org/10.1371/journal.pone.0097418
Wang W, Green M, Choi JE, Gijon M, Kennedy PD, Johnson JK, Liao P, Lang X, Kryczek I, Sell A, Xia H, Zhou J, Li G, Li J, Li W, Wei S, Vatan L, Zhang H, Szeliga W, Gu W, Liu R, Lawrence TS, Lamb C, Tanno Y, Cieslik M, Stone E, Georgiou G, Chan TA, Chinnaiyan A, Zou W (2019) CD8(+) T cells regulate tumour ferroptosis during cancer immunotherapy. Nature 569:270–274. https://doi.org/10.1038/s41586-019-1170-y
Patra S, Pradhan B, Nayak R, Behera C, Panda KC, Das S, Jena M, Bhutia SK (2021) Apoptosis and autophagy modulating dietary phytochemicals in cancer therapeutics: current evidences and future perspectives. Phytother Res 35:4194–4214. https://doi.org/10.1002/ptr.7082
Yang S, Fan W, Li Y, Liu Q, He H, Huang F (2021) Autophagy in tooth: physiology, disease and therapeutic implication. Cell Biochem Funct 39:702–712. https://doi.org/10.1002/cbf.3636
Dixon SJ, Stockwell BR (2019) The hallmarks of ferroptosis. Annual Rev Cancer Biol 3:35–54. https://doi.org/10.1146/annurev-cancerbio-030518-055844
Hou W, Xie Y, Song X, Sun X, Lotze MT, Zeh HJ, Kang R, Tang D (2016) Autophagy promotes ferroptosis by degradation of ferritin. Autophagy 12:1425–1428. https://doi.org/10.1080/15548627.2016.1187366
Wu Z, Geng Y, Lu X, Shi Y, Wu G, Zhang M, Shan B, Pan H, Yuan J (2019) Chaperone-mediated autophagy is involved in the execution of ferroptosis. Proc Natl Acad Sci USA 116:2996–3005. https://doi.org/10.1073/pnas.1819728116
Acknowledgements
The authors are grateful to all staff at the Laboratory Animal Center of Zhujiang Hospital of Southern Medical University, and all staff at the Laboratory Center of Zhujiang Hospital of Southern Medical University. We thank Prof. Xun Sun and Dr. Wenjun Li for helpful discussions and insightful comments. We also thank Editage (www.editage.com) for English language editing.
Funding
This work was supported by the National Natural Science Foundation of China (Grant Number: 82072762 and 32001040); National Key Research and Development Program of China (Grant Number: 2021YFA0910700 and 2018YFA0902701), and Guangdong Science and Technology Department (Grant Number: 2020B1212030004); Guangdong Introducing Innovative and Entrepreneurial Teams (Grant Number: 2019BT02Y198) and Shenzhen Key Laboratory for Cancer Metastasis and Personalized Therapy (Grant Number: ZDSYS20210623091811035); Guangdong Basic and Applied Basic Research Foundation (Grant Number: 2023A1515030045) and Presidential Foundation of Zhujiang Hospital, Southern Medical University (Grant Number: yzjj2022ms4).
Author information
Authors and Affiliations
Contributions
Conceptualization: HS, YK and JDH; Methodology: TL; Software: JZ; Validation: JC; Formal analysis: JC; Investigation: NZ, YH; Resources: CM; Data curation: JZ; Writing—original draft preparation: JC; Writing—review and editing: MZ, JJ; Visualization: JC; Supervision: HS, YK, and JDH; Project administration: JC; Funding acquisition: HS, YK, and JDH. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Ethical approval
This study was approved by the Animal Ethics Committee of Southern Medical University and performed in line with the principles of the Declaration of Helsinki and according to the National Institutes of Health (Approval ID: LAEC-2022-068FS).
Consent to participate
Not applicable.
Consent for publication
Not applicable.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Chen, J., Li, T., Zhou, N. et al. Engineered Salmonella inhibits GPX4 expression and induces ferroptosis to suppress glioma growth in vitro and in vivo. J Neurooncol 163, 607–622 (2023). https://doi.org/10.1007/s11060-023-04369-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-023-04369-5