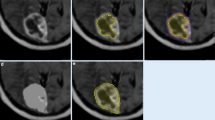
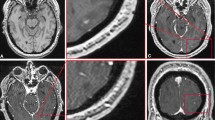
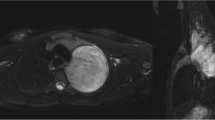
Abstract
Purpose
The Response Assessment in Neuro-Oncology Brain Metastases (RANO-BM) working group proposed a guide for treatment responses for BMs by utilizing the longest diameter; however, despite recognizing that many patients with BMs have sub-centimeter lesions, the group referred to these lesions as unmeasurable due to issues with repeatability and interpretation. In light of RANO—BM recommendations, we aimed to correlate linear and volumetric measurements in sub-centimeter BMs on contrast-enhanced MRI using intelligent automation software.
Methods
In this retrospective study, patients with BMs scanned with MRI between January 1, 2018, and December 31, 2021, were screened. Inclusion criteria were: (1) at least one sub-centimeter BM with an integer millimeter-longest diameter was noted in the MRI report; (2) patients were a minimum of 18 years of age; (3) patients with available pre-treatment three-dimensional T1-weighted spoiled gradient-echo MRI scan. The screening was terminated when there were 20 lesions in each group. Lesion volumes were measured with the help of intelligent automation software Jazz (AI Medical, Zollikon, Switzerland) by two readers. The Kruskal-Wallis test was used to compare volumetric differences.
Results
Our study included 180 patients. The agreement for volumetric measurements was excellent between the two readers. The volumes of the following groups were not significantly different: 1–2 mm, 1–3 mm, 1–4 mm, 2–3 mm, 2–4 mm, 3–4 mm, 3–5 mm, 4–5 mm, 5–6 mm, 5–7 mm, 6–7 mm, 6–8 mm, 6–9 mm, 7–8 mm, 7–9 mm, 8–9 mm.
Conclusion
Our findings indicate that the largest diameter of a lesion may not accurately represent its volume. Additional research is required to determine which method is superior for measuring radiologic response to therapy and which parameter correlates best with clinical improvement or deterioration.



Similar content being viewed by others
Data availability
The datasets generated during and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- mm:
-
Millimeter
- Q1:
-
Lower quartile
- IQR:
-
Interquartile range
- Q3:
-
Upper quartile
- SD:
-
Standard deviation
References
Lin X, DeAngelis LM (2015) Treatment of brain metastases. J Clin Oncol 33:3475–3484. https://doi.org/10.1200/JCO.2015.60.9503
Sacks P, Rahman M (2020) Epidemiology of brain metastases. Neurosurg Clin N Am 31:481–488. https://doi.org/10.1016/j.nec.2020.06.001
Preusser M, Weller M (2015) Brain metastasis research: a late awakening. Chin Clin Oncol 4:17. https://doi.org/10.3978/j.issn.2304-3865.2015.05.01
Oft D, Schmidt MA, Weissmann T et al (2020) Volumetric regression in brain metastases after stereotactic radiotherapy: time course, predictors, and significance. Front Oncol 10:590980. https://doi.org/10.3389/fonc.2020.590980
Soffietti R, Abacioglu U, Baumert B et al (2017) Diagnosis and treatment of brain metastases from solid tumors: guidelines from the European Association of Neuro-Oncology (EANO). Neuro Oncol 19:162–174. https://doi.org/10.1093/neuonc/now241
Lin NU, Lee EQ, Aoyama H et al (2015) Response assessment criteria for brain metastases: proposal from the RANO group. Lancet Oncol 16:e270–278. https://doi.org/10.1016/S1470-2045(15)70057-4
Schwartz LH, Litière S, de Vries E et al (2016) RECIST 1.1-Update and clarification: from the RECIST committee. Eur J Cancer 62:132–137. https://doi.org/10.1016/j.ejca.2016.03.081
Shah GD, Kesari S, Xu R et al (2006) Comparison of linear and volumetric criteria in assessing tumor response in adult high-grade gliomas. Neuro Oncol 8:38–46. https://doi.org/10.1215/S1522851705000529
Bauknecht H-C, Romano VC, Rogalla P et al (2010) Intra- and interobserver variability of linear and volumetric measurements of brain metastases using contrast-enhanced magnetic resonance imaging. Invest Radiol 45:49–56. https://doi.org/10.1097/RLI.0b013e3181c02ed5
Gondi V, Bauman G, Bradfield L et al (2022) Radiation therapy for brain metastases: an ASTRO clinical practice guideline. Pract Radiat Oncol 12:265–282. https://doi.org/10.1016/j.prro.2022.02.003
Minniti G, Clarke E, Lanzetta G et al (2011) Stereotactic radiosurgery for brain metastases: analysis of outcome and risk of brain radionecrosis. Radiat Oncol 6:48. https://doi.org/10.1186/1748-717X-6-48
Jazz. In: AI Medical. https://www.ai-medical.ch/jazz. Accessed 9 Mar 2023
R Core Team (2022) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. https://www.R-project.org/
Koo TK, Li MY (2016) A Guideline of selecting and reporting intraclass correlation coefficients for reliability research. J Chiropr Med 15:155–163. https://doi.org/10.1016/j.jcm.2016.02.012
Plotly Technologies Inc. Collaborative data science. Montréal, QC (2015) https://plot.ly
Ostrom QT, Wright CH, Barnholtz-Sloan JS (2018) Brain metastases: epidemiology. Handb Clin Neurol 149:27–42. https://doi.org/10.1016/B978-0-12-811161-1.00002-5
Le Rhun E, Guckenberger M, Smits M et al (2021) EANO-ESMO clinical practice guidelines for diagnosis, treatment and follow-up of patients with brain metastasis from solid tumours. Ann Oncol 32:1332–1347. https://doi.org/10.1016/j.annonc.2021.07.016
Yoo SK, Kim TH, Chun J et al (2022) Deep-learning-based automatic detection and segmentation of brain metastases with small volume for stereotactic ablative radiotherapy. Cancers. https://doi.org/10.3390/cancers14102555
Huang YX, Bert C, Sommer P et al (2022) Deep learning for brain metastasis detection and segmentation in longitudinal MRI data. Med Phys 49:5773–5786. https://doi.org/10.1002/mp.15863
Li R, Guo Y, Zhao Z et al (2023) MRI-based two-stage deep learning model for automatic detection and segmentation of brain metastases. Eur Radiol. https://doi.org/10.1007/s00330-023-09420-7
Ozkara BB, Chen MM, Federau C et al (2023) Deep learning for detecting brain metastases on MRI: a systematic review and Meta-analysis. Cancers (Basel) 15:334. https://doi.org/10.3390/cancers15020334
Yi D, Grovik E, Tong E et al (2021) MRI pulse sequence integration for deep-learning-based brain metastases segmentation. Med Phys 48:6020–6035. https://doi.org/10.1002/mp.15136
Grovik E, Yi D, Iv M et al (2021) Handling missing MRI sequences in deep learning segmentation of brain metastases: a multicenter study. Npj Digit Med. https://doi.org/10.1038/s41746-021-00398-4. 4:
Yang Z, Chen ML, Kazemimoghadam M et al (2022) Deep-learning and radiomics ensemble classifier for false positive reduction in brain metastases segmentation. Phys Med Biol. https://doi.org/10.1088/1361-6560/ac4667
Liang Y, Lee K, Bovi JA et al (2022) Deep learning-based automatic detection of brain metastases in heterogenous multi-institutional magnetic resonance imaging sets: an exploratory analysis of NRG-CC001. Int J Radiat Oncol Biol Phys 114:529–536. https://doi.org/10.1016/j.ijrobp.2022.06.081
Am B, Kd M, Py C et al (2013) Tumor volume as a predictor of survival and local control in patients with brain metastases treated with Gamma Knife surgery. J Neurosurg. https://doi.org/10.3171/2013.7.JNS13431
Routman DM, Bian SX, Diao K et al (2018) The growing importance of lesion volume as a prognostic factor in patients with multiple brain metastases treated with stereotactic radiosurgery. Cancer Med 7:757–764. https://doi.org/10.1002/cam4.1352
Wolchok JD, Hoos A, O’Day S et al (2009) Guidelines for the evaluation of immune therapy activity in solid tumors: immune-related response criteria. Clin Cancer Res 15:7412–7420. https://doi.org/10.1158/1078-0432.CCR-09-1624
Funding
The authors declare that no funds, grants, or other support were received during the preparation of this manuscript.
Author information
Authors and Affiliations
Contributions
Conceptualization: BBO, CF, FEU, MW; Data curation: BBO, SAD, DP, FEU, MMC; Formal Analysis: BBO, MW; Investigation: CF, FEU, MMC, MW; Methodology: BBO, FEU, MW; Software: BBO, CF; Validation: CF, FEU, MMC, MW; Visualization: BBO, CF; Supervision: FEU, MMC, MW; Writing—original draft: BBO, CF; Writing—review & editing: All authors.
Corresponding author
Ethics declarations
Competing interests
The authors have no relevant financial or non-financial interests to disclose.
Ethical approval
This study was performed in line with the principles of the Declaration of Helsinki. Approval was granted by the Institutional Review Board at The University of Texas MD Anderson Cancer Center.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ozkara, B.B., Federau, C., Dagher, S.A. et al. Correlating volumetric and linear measurements of brain metastases on MRI scans using intelligent automation software: a preliminary study. J Neurooncol 162, 363–371 (2023). https://doi.org/10.1007/s11060-023-04297-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-023-04297-4