Abstract
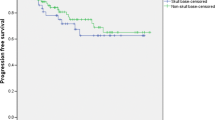
Secretory meningioma (SM) is a rare histological subtype of the meningioma family. Few reports investigating SM have been published due to its extremely low incidence; thus, the current understanding of this disease is poor. We analyzed the incidence and clinical, radiological, pathological, and prognostic features of SM. Approximately 12,380 intracranial meningiomas were surgically resected at Beijing Tiantan Hospital between April 2008 and January 2017. All pathologically confirmed SM cases were identified. SMs accounted for approximately 1.2% of the intracranial meningiomas (149 of 12,380). The patients with SM had a mean age of 51.0 years and were predominantly female (112 female and 37 male). Radiologically, peritumoral brain edema was observed in 49 (32.9%) patients. Gross total resection was achieved in 115 (77.2%) cases. At the 35-months median follow-up (range 4–109 months), six patients had tumor recurrence, and one patient died from the tumor recurrence. The 5-year progression-free survival rates were 95.9%, and the 5-year overall survival rate was 99.3%. A skull base location and a tumor size ≥ 3.5 cm were significantly associated with poor short-term outcomes, and a skull base location was significantly associated with an increased risk of poor long-term outcomes (P < 0.05). A skull base location (OR 3.797; 95% CI 1.071–13.468; P = 0.039) and tumor size ≥ 3.5 cm (OR 2.616; 95% CI 1.107–6.181; P = 0.028) were independent risk factors for non-gross total resection. A son-skull base location (OR 0.070; 95% CI 0.028–0.177; P = 0.001) was the only independent risk factor that correlated with more severe peritumoral brain edema. SM is a rare subtype of meningiomas with a female predominance and low recurrence. Our results highlight the risk factors for short- and long-term outcomes, which can be useful for selecting treatments and predicting prognosis. Microsurgical treatment of a skull base SM remains a formidable challenge due to a large tumor size and critical neurovascular structure encasement.
Similar content being viewed by others
References
Ostrom QT, Gittleman H, Farah P, Ondracek A, Chen Y, Wolinsky Y, Stroup NE, Kruchko C, Barnholtz-Sloan JS (2013) CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2006–2010. Neuro-oncology 15(Suppl 2):ii1–56. https://doi.org/10.1093/neuonc/not151
Whittle IR, Smith C, Navoo P, Collie D (2004) Meningiomas Lancet 363:1535–1543. https://doi.org/10.1016/S0140-6736(04)16153-9
Dolecek TA, Dressler EV, Thakkar JP, Liu M, Al-Qaisi A, Villano JL (2015) Epidemiology of meningiomas post-Public Law 107–206: The Benign Brain Tumor Cancer Registries Amendment Act. Cancer 121:2400–2410. https://doi.org/10.1002/cncr.29379
Cea-Soriano L, Wallander MA, Garcia Rodriguez LA (2012) Epidemiology of meningioma in the United Kingdom. Neuroepidemiology 39:27–34. https://doi.org/10.1159/000338081
Baldi I, Engelhardt J, Bonnet C, Bauchet L, Berteaud E, Gruber A, Loiseau H (2014) Epidemiology of meningiomas. Neurochirurgie. https://doi.org/10.1016/j.neuchi.2014.05.006
Louis DN, Perry A, Reifenberger G, von Deimling A, Figarella-Branger D, Cavenee WK, Ohgaki H, Wiestler OD, Kleihues P, Ellison DW (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131:803–820. https://doi.org/10.1007/s00401-016-1545-1
Riemenschneider MJ, Perry A, Reifenberger G (2006) Histological classification and molecular genetics of meningiomas. Lancet Neurol 5:1045–1054. https://doi.org/10.1016/S1474-4422(06)70625-1
Kepes J (1961) Observations on the formation of psammoma bodies and pseudopsammoma bodies in meningiomas. J Neuropathol Exp Neurol 20:255–262
Alguacil-Garcia A, Pettigrew NM, Sima AA (1986) Secretory meningioma. A distinct subtype of meningioma. Am J Surg Pathol 10:102–111
Kleihues P, Burger PC, Scheithauer BW (1993) The new WHO classification of brain tumours. Brain Pathol 3:255–268
Mohme M, Emami P, Regelsberger J, Matschke J, Lamszus K, Westphal M, Eicker SO (2016) Secretory meningiomas: increased prevalence of seizures secondary to edema formation in a rare histologic subtype. World Neurosurg 92:418–425. https://doi.org/10.1016/j.wneu.2016.05.046
Tsuzuki N, Nakau H, Sugaya M, Hashizume K, Matsukuma S, Wada R, Kuwabara N (1997) Secretory meningioma with severe perifocal edema: case report. Neurologia Medico-chirurgica 37:620–623
Nishio S, Morioka T, Suzuki S, Hirano K, Fukui M (2001) Secretory meningioma: clinicopathologic features of eight cases. J Clin Neurosci 8:335–339. https://doi.org/10.1054/jocn.2000.0793
Probst-Cousin S, Villagran-Lillo R, Lahl R, Bergmann M, Schmid KW, Gullotta F (1997) Secretory meningioma: clinical, histologic, and immunohistochemical findings in 31 cases. Cancer 79:2003–2015
Colakoglu N, Demirtas E, Oktar N, Yuntem N, Islekel S, Ozdamar N (2003) Secretory meningiomas. J Neuro-oncol 62:233–241
Regelsberger J, Hagel C, Emami P, Ries T, Heese O, Westphal M (2009) Secretory meningiomas: a benign subgroup causing life-threatening complications. Neuro-oncology 11:819–824. https://doi.org/10.1215/15228517-2008-109
Tirakotai W, Mennel HD, Celik I, Hellwig D, Bertalanffy H, Riegel T (2006) Secretory meningioma: immunohistochemical findings and evaluation of mast cell infiltration. Neurosurg Rev 29:41–48. https://doi.org/10.1007/s10143-005-0402-9
Cenacchi G, Ferri GG, Salfi N, Tarantino L, Modugno GC, Ceroni AR, Martinelli GN (2008) Secretory meningioma of the middle ear: a light microscopic, immunohistochemical and ultrastructural study of one case. Neuropathology 28:69–73. https://doi.org/10.1111/j.1440-1789.2007.00821.x
Kamp MA, Beseoglu K, Eicker S, Steiger HJ, Hanggi D (2011) Secretory meningiomas: systematic analysis of epidemiological, clinical, and radiological features. Acta Neurochirurgica 153:457–465. https://doi.org/10.1007/s00701-010-0914-0
Wang DJ, Xie Q, Gong Y, Wang Y, Cheng HX, Mao Y, Zhong P, Huang FP, Zheng K, Wang YF, Bao WM, Yang BJ, Chen H, Xie LQ, Zheng MZ, Tang HL, Zhu HD, Chen XC, Zhou LF (2013) Secretory meningiomas: clinical, radiological and pathological findings in 70 consecutive cases at one institution. Int J Clin Exp Pathol 6:358–374
Simpson D (1957) The recurrence of intracranial meningiomas after surgical treatment. J Neurol Neurosurg Psychiatry 20:22–39
Louis DN, Ohgaki H, Wiestler OD, Cavenee WK, Burger PC, Jouvet A, Scheithauer BW, Kleihues P (2007) The 2007 WHO classification of tumours of the central nervous system. Acta Neuropathol 114:97–109. https://doi.org/10.1007/s00401-007-0243-4
Buccoliero AM, Gheri CF, Castiglione F, Ammannati F, Gallina P, Taddei A, Garbini F, Degl’Innocenti DR, Arganini L, Di Lorenzo N, Mennonna P, Taddei GL (2007) Merlin expression in secretory meningiomas: evidence of an NF2-independent pathogenesis? Immunohistochemical study. Appl Immunohistochem Mol Morphol 15:353–357. https://doi.org/10.1097/01.pai.0000213114.27978.3a
Buhl R, Hugo HH, Mihajlovic Z, Mehdorn HM (2001) Secretory meningiomas: clinical and immunohistochemical observations. Neurosurgery 48:297–301 (discussion 301–292)
Bi WL, Abedalthagafi M, Horowitz P, Agarwalla PK, Mei Y, Aizer AA, Brewster R, Dunn GP, Al-Mefty O, Alexander BM, Santagata S, Beroukhim R, Dunn IF (2016) Genomic landscape of intracranial meningiomas. J Neurosurg 125:525–535. https://doi.org/10.3171/2015.6.JNS15591
Hartmann C, Sieberns J, Gehlhaar C, Simon M, Paulus W, von Deimling A (2006) NF2 mutations in secretory and other rare variants of meningiomas. Brain Pathol 16:15–19
Reuss DE, Piro RM, Jones DT, Simon M, Ketter R, Kool M, Becker A, Sahm F, Pusch S, Meyer J, Hagenlocher C, Schweizer L, Capper D, Kickingereder P, Mucha J, Koelsche C, Jäger N, Santarius T, Tarpey PS, Stephens PJ, Andrew Futreal P, Wellenreuther R, Kraus J, Lenartz D, Herold-Mende C, Hartmann C, Mawrin C, Giese N, Eils R, Collins VP, König R, Wiestler OD, Pfister SM, von Deimling A (2013) Secretory meningiomas are defined by combined KLF4 K409Q and TRAF7 mutations. Acta Neuropathol 125: 351–358. https://doi.org/10.1007/s00401-013-1093-x
Acknowledgements
We would like to thank all the patients who trusted us and all the physicians and staff who helped this study.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors report no conflict of interest concerning the materials or methods used in this study or the findings described in this paper.
Ethical standards
This retrospective study was approved by Institutional Review Board of Beijing Tiantan Hospital, Capital Medical University, Beijing, China
Electronic supplementary material
Below is the link to the electronic supplementary material.
11060_2017_2671_MOESM1_ESM.tif
Supplementary Fig. 1. Radiologic presentation and quantification of peritumoral edema formation in SM. CT and MRI of a representative SM case, pressing on the right temporal lobe with extensive perifocal edema and pronounced midline shift. Plain CT scan (A), plain T1-weighted (B) and T2-weighted (C) MR images, and ADC images (D) depict widespread edema of the white matter. (E) Comparison of peritumoral edema formation for skull base and non-skull base tumors using Regelsberger Score [<link rid="bib16">16</link>]. (F) Comparison of tumor location for peritumoral tumor edema and non-edema (TIF 705 KB)
11060_2017_2671_MOESM2_ESM.tif
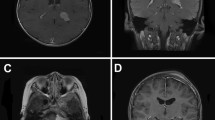
Supplementary Fig. 2. Four cases with respective grade 0 to 3 peritumoral brain edema, manifested by axial T2 weighted images. A: grade 0 (cases 31); B: grade 1 (cases 85); C: grade 2 (cases 72); D: grade 3 (cases 63) (TIF 1988 KB)
11060_2017_2671_MOESM4_ESM.tif
Supplementary Fig. 4. Pathological features of SM. Histological section showed typical focal epithelial differentiation, glandular lumina containing secretory globules, and pseudopsammoma bodies (A, B and C). In the immunohistochemical analysis, tumor cells were positive for CEA (D), EMA (E), CK (F) and vimentin (G). Original magnification in (A, C-G) is 200 ×. (B) is 400 × (TIF 4687 KB)
Rights and permissions
About this article
Cite this article
Tao, X., Wang, K., Dong, J. et al. Clinical features, surgical management, and prognostic factors of secretory meningiomas: a single-center case series of 149 patients. J Neurooncol 136, 515–522 (2018). https://doi.org/10.1007/s11060-017-2671-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-017-2671-x