Abstract
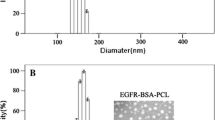
The epidermal growth factor receptor deletion variant EGFRvIII is known to be expressed in a subset of patients with glioblastoma (GBM) tumors that enhances tumorigenicity and also accounts for radiation and chemotherapy resistance. Targeting the EGFRvIII deletion mutant may lead to improved GBM therapy and better patient prognosis. Multifunctional magnetic nanoparticles serve as a potential clinical tool that can provide cancer cell targeted drug delivery, imaging, and therapy. Our previous studies have shown that an EGFRvIII-specific antibody and cetuximab (an EGFR- and EGFRvIII-specific antibody), when bioconjugated to IONPs (EGFRvIII-IONPs or cetuximab-IONPs respectively), can simultaneously provide sensitive cancer cell detection by magnetic resonance imaging (MRI) and targeted therapy of experimental GBM. In this study, we investigated whether cetuximab-IONPs can additionally allow for the radiosensitivity enhancement of GBM. Cetuximab-IONPs were used in combination with single (10 Gy × 1) or multiple fractions (10 Gy × 2) of ionizing radiation (IR) for radiosensitization of EGFRvIII-overexpressing human GBM cells in vitro and in vivo after convection-enhanced delivery (CED). A significant GBM antitumor effect was observed in vitro after treatment with cetuximab-IONPs and subsequent single or fractionated IR. A significant increase in overall survival of nude mice implanted with human GBM xenografts was found after treatment by cetuximab-IONP CED and subsequent fractionated IR. Increased DNA double strands breaks (DSBs), as well as increased reactive oxygen species (ROS) formation, were felt to represent the mediators of the observed radiosensitization effect with the combination therapy of IR and cetuximab-IONPs treatment.





Similar content being viewed by others
References
Braun S, Oppermann H, Mueller A, Renner C, Hovhannisyan A, Baran-Schmidt R, Gebhardt R, Hipkiss A, Thiery J, Meixensberger J, Gaunitz F (2012) Hedgehog signaling in glioblastoma multiforme. Cancer Biol Ther 13(7):487–495. doi:10.4161/cbt.19591
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ, Belanger K, Brandes AA, Marosi C, Bogdahn U, Curschmann J, Janzer RC, Ludwin SK, Gorlia T, Allgeier A, Lacombe D, Cairncross JG, Eisenhauer E, Mirimanoff RO (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352(10):987–996. doi:10.1056/NEJMoa043330
Omay SB, Vogelbaum MA (2009) Current concepts and newer developments in the treatment of malignant gliomas. Indian J Cancer 46(2):88–95
Parsons DW, Jones S, Zhang X, Lin JC, Leary RJ, Angenendt P, Mankoo P, Carter H, Siu IM, Gallia GL, Olivi A, McLendon R, Rasheed BA, Keir S, Nikolskaya T, Nikolsky Y, Busam DA, Tekleab H, Diaz LA Jr, Hartigan J, Smith DR, Strausberg RL, Marie SK, Shinjo SM, Yan H, Riggins GJ, Bigner DD, Karchin R, Papadopoulos N, Parmigiani G, Vogelstein B, Velculescu VE, Kinzler KW (2008) An integrated genomic analysis of human glioblastoma multiforme. Science 321(5897):1807–1812. doi:10.1126/science.1164382
Gan HK, Kaye AH, Luwor RB (2009) The EGFRvIII variant in glioblastoma multiforme. J Clin Neurosci 16(6):748–754. doi:10.1016/j.jocn.2008.12.005
Mukherjee B, McEllin B, Camacho CV, Tomimatsu N, Sirasanagandala S, Nannepaga S, Hatanpaa KJ, Mickey B, Madden C, Maher E, Boothman DA, Furnari F, Cavenee WK, Bachoo RM, Burma S (2009) EGFRvIII and DNA double-strand break repair: a molecular mechanism for radioresistance in glioblastoma. Cancer Res 69(10):4252–4259. doi:10.1158/0008-5472.can-08-4853
Mendelsohn J (2002) Targeting the epidermal growth factor receptor for cancer therapy. J Clin Oncol 20(18 Suppl):1s–13s
Garrett CR, Eng C (2011) Cetuximab in the treatment of patients with colorectal cancer. Expert Opin Biol Ther 11(7):937–949. doi:10.1517/14712598.2011.582464
Cohen MH, Chen H, Shord S, Fuchs C, He K, Zhao H, Sickafuse S, Keegan P, Pazdur R (2013) Approval summary: cetuximab in combination with cisplatin or carboplatin and 5-fluorouracil for the first-line treatment of patients with recurrent locoregional or metastatic squamous cell head and neck cancer. Oncologist 18(4):460–466. doi:10.1634/theoncologist.2012-0458
Li S, Schmitz KR, Jeffrey PD, Wiltzius JJ, Kussie P, Ferguson KM (2005) Structural basis for inhibition of the epidermal growth factor receptor by cetuximab. Cancer Cell 7(4):301–311. doi:10.1016/j.ccr.2005.03.003
Martens T, Laabs Y, Gunther HS, Kemming D, Zhu Z, Witte L, Hagel C, Westphal M, Lamszus K (2008) Inhibition of glioblastoma growth in a highly invasive nude mouse model can be achieved by targeting epidermal growth factor receptor but not vascular endothelial growth factor receptor-2. Clin Cancer Res 14(17):5447–5458. doi:10.1158/1078-0432.ccr-08-0147
Belda-Iniesta C, Carpeno Jde C, Saenz EC, Gutierrez M, Perona R, Baron MG (2006) Long term responses with cetuximab therapy in glioblastoma multiforme. Cancer Biol Ther 5(8):912–914
Eller JL, Longo SL, Hicklin DJ, Canute GW (2002) Activity of anti-epidermal growth factor receptor monoclonal antibody C225 against glioblastoma multiforme. Neurosurgery 51(4):1005 discussion 1013–1004
Kaluzova M, Bouras A, Machaidze R, Hadjipanayis CG (2015) Targeted therapy of glioblastoma stem-like cells and tumor non-stem cells using cetuximab-conjugated iron-oxide nanoparticles. Oncotarget 6(11):8788–8806
Platt S, Nduom E, Kent M, Freeman C, Machaidze R, Kaluzova M, Wang L, Mao H, Hadjipanayis CG (2012) Canine model of convection-enhanced delivery of cetuximab-conjugated iron-oxide nanoparticles monitored with magnetic resonance imaging. Clin Neurosurg 59:107–113. doi:10.1227/NEU.0b013e31826989ef
Maier-Hauff K, Ulrich F, Nestler D, Niehoff H, Wust P, Thiesen B, Orawa H, Budach V, Jordan A (2011) Efficacy and safety of intratumoral thermotherapy using magnetic iron-oxide nanoparticles combined with external beam radiotherapy on patients with recurrent glioblastoma multiforme. J Neurooncol 103(2):317–324. doi:10.1007/s11060-010-0389-0
Hadjipanayis CG, Bonder MJ, Balakrishnan S, Wang X, Mao H, Hadjipanayis GC (2008) Metallic iron nanoparticles for MRI contrast enhancement and local hyperthermia. Small 4(11):1925–1929. doi:10.1002/smll.200800261
Jordan A, Scholz R, Maier-Hauff K, van Landeghem FK, Waldoefner N, Teichgraeber U, Pinkernelle J, Bruhn H, Neumann F, Thiesen B, von Deimling A, Felix R (2006) The effect of thermotherapy using magnetic nanoparticles on rat malignant glioma. J Neurooncol 78(1):7–14. doi:10.1007/s11060-005-9059-z
Mahmoudi M, Hofmann H, Rothen-Rutishauser B, Petri-Fink A (2012) Assessing the in vitro and in vivo toxicity of superparamagnetic iron oxide nanoparticles. Chem Rev 112(4):2323–2338. doi:10.1021/cr2002596
Hadjipanayis CG, Machaidze R, Kaluzova M, Wang L, Schuette AJ, Chen H, Wu X, Mao H (2010) EGFRvIII antibody-conjugated iron oxide nanoparticles for magnetic resonance imaging-guided convection-enhanced delivery and targeted therapy of glioblastoma. Cancer Res 70(15):6303–6312. doi:10.1158/0008-5472.can-10-1022
Nagane M, Coufal F, Lin H, Bogler O, Cavenee WK, Huang HJ (1996) A common mutant epidermal growth factor receptor confers enhanced tumorigenicity on human glioblastoma cells by increasing proliferation and reducing apoptosis. Cancer Res 56(21):5079–5086
Nishikawa R, Ji XD, Harmon RC, Lazar CS, Gill GN, Cavenee WK, Huang HJ (1994) A mutant epidermal growth factor receptor common in human glioma confers enhanced tumorigenicity. Proc Natl Acad Sci U S A 91(16):7727–7731
Jackson SP, Bartek J (2009) The DNA-damage response in human biology and disease. Nature 461(7267):1071–1078. doi:10.1038/nature08467
Kuo LJ, Yang LX (2008) Gamma-H2AX—a novel biomarker for DNA double-strand breaks. In Vivo 22(3):305–309
Yamamori T, Yasui H, Yamazumi M, Wada Y, Nakamura Y, Nakamura H, Inanami O (2012) Ionizing radiation induces mitochondrial reactive oxygen species production accompanied by upregulation of mitochondrial electron transport chain function and mitochondrial content under control of the cell cycle checkpoint. Free Radic Biol Med 53(2):260–270. doi:10.1016/j.freeradbiomed.2012.04.033
Ahamed M, Alhadlaq HA, Khan MAM, Akhtar MJ (2013) Selective killing of cancer cells by iron oxide nanoparticles mediated through reactive oxygen species via p53 pathway. J Nanopart Res 15(1):1225. doi:10.1007/s11051-012-1225-6
Lammering G, Valerie K, Lin PS, Hewit TH, Schmidt-Ullrich RK (2004) Radiation-induced activation of a common variant of EGFR confers enhanced radioresistance. Radiother Oncol 72(3):267–273. doi:10.1016/j.radonc.2004.07.004
Li B, Yuan M, Kim IA, Chang CM, Bernhard EJ, Shu HK (2004) Mutant epidermal growth factor receptor displays increased signaling through the phosphatidylinositol-3 kinase/AKT pathway and promotes radioresistance in cells of astrocytic origin. Oncogene 23(26):4594–4602. doi:10.1038/sj.onc.1207602
Lammering G, Hewit TH, Valerie K, Contessa JN, Amorino GP, Dent P, Schmidt-Ullrich RK (2003) EGFRvIII-mediated radioresistance through a strong cytoprotective response. Oncogene 22(36):5545–5553. doi:10.1038/sj.onc.1206788
Fan QW, Cheng CK, Gustafson WC, Charron E, Zipper P, Wong RA, Chen J, Lau J, Knobbe-Thomsen C, Weller M, Jura N, Reifenberger G, Shokat KM, Weiss WA (2013) EGFR phosphorylates tumor-derived EGFRvIII driving STAT3/5 and progression in glioblastoma. Cancer Cell 24(4):438–449. doi:10.1016/j.ccr.2013.09.004
Orringer DA, Koo YE, Chen T, Kopelman R, Sagher O, Philbert MA (2009) Small solutions for big problems: the application of nanoparticles to brain tumor diagnosis and therapy. Clin Pharmacol Ther 85(5):531–534. doi:10.1038/clpt.2008.296
Provenzale JM, Silva GA (2009) Uses of nanoparticles for central nervous system imaging and therapy. AJNR Am J Neuroradiol 30(7):1293–1301. doi:10.3174/ajnr.A1590
Caruso G, Caffo M, Alafaci C, Raudino G, Cafarella D, Lucerna S, Salpietro FM, Tomasello F (2011) Could nanoparticle systems have a role in the treatment of cerebral gliomas? Nanomedicine 7(6):744–752. doi:10.1016/j.nano.2011.02.008
Sun C, Veiseh O, Gunn J, Fang C, Hansen S, Lee D, Sze R, Ellenbogen RG, Olson J, Zhang M (2008) In vivo MRI detection of gliomas by chlorotoxin-conjugated superparamagnetic nanoprobes. Small 4(3):372–379. doi:10.1002/smll.200700784
Reddy GR, Bhojani MS, McConville P, Moody J, Moffat BA, Hall DE, Kim G, Koo YE, Woolliscroft MJ, Sugai JV, Johnson TD, Philbert MA, Kopelman R, Rehemtulla A, Ross BD (2006) Vascular targeted nanoparticles for imaging and treatment of brain tumors. Clin Cancer Res 12(22):6677–6686. doi:10.1158/1078-0432.ccr-06-0946
Diaz Miqueli A, Rolff J, Lemm M, Fichtner I, Perez R, Montero E (2009) Radiosensitisation of U87MG brain tumours by anti-epidermal growth factor receptor monoclonal antibodies. Br J Cancer 100(6):950–958. doi:10.1038/sj.bjc.6604943
Eller JL, Longo SL, Kyle MM, Bassano D, Hicklin DJ, Canute GW (2005) Anti-epidermal growth factor receptor monoclonal antibody cetuximab augments radiation effects in glioblastoma multiforme in vitro and in vivo. Neurosurgery 56(1):155 discussion 162
Mikkelsen RB, Wardman P (2003) Biological chemistry of reactive oxygen and nitrogen and radiation-induced signal transduction mechanisms. Oncogene 22(37):5734–5754. doi:10.1038/sj.onc.1206663
Zimmer C, Weissleder R, Poss K, Bogdanova A, Wright SC Jr, Enochs WS (1995) MR imaging of phagocytosis in experimental gliomas. Radiology 197(2):533–538. doi:10.1148/radiology.197.2.7480707
Moore A, Marecos E, Bogdanov A Jr, Weissleder R (2000) Tumoral distribution of long-circulating dextran-coated iron oxide nanoparticles in a rodent model. Radiology 214(2):568–574. doi:10.1148/radiology.214.2.r00fe19568
Klein S, Sommer A, Distel LV, Neuhuber W, Kryschi C (2012) Superparamagnetic iron oxide nanoparticles as radiosensitizer via enhanced reactive oxygen species formation. Biochem Biophys Res Commun 425(2):393–397. doi:10.1016/j.bbrc.2012.07.108
Villanueva A, Canete M, Roca AG, Calero M, Veintemillas-Verdaguer S, Serna CJ, Morales Mdel P, Miranda R (2009) The influence of surface functionalization on the enhanced internalization of magnetic nanoparticles in cancer cells. Nanotechnology 20(11):115103. doi:10.1088/0957-4484/20/11/115103
Hadjipanayis CG, Fellows-Mayle W, Deluca NA (2008) Therapeutic efficacy of a herpes simplex virus with radiation or temozolomide for intracranial glioblastoma after convection-enhanced delivery. Mol Ther 16(11):1783–1788. doi:10.1038/mt.2008.185
Sampson JH, Akabani G, Archer GE, Berger MS, Coleman RE, Friedman AH, Friedman HS, Greer K, Herndon JE 2nd, Kunwar S, McLendon RE, Paolino A, Petry NA, Provenzale JM, Reardon DA, Wong TZ, Zalutsky MR, Pastan I, Bigner DD (2008) Intracerebral infusion of an EGFR-targeted toxin in recurrent malignant brain tumors. Neuro Oncol 10(3):320–329. doi:10.1215/15228517-2008-012
Bobo RH, Laske DW, Akbasak A, Morrison PF, Dedrick RL, Oldfield EH (1994) Convection-enhanced delivery of macromolecules in the brain. Proc Natl Acad Sci USA 91(6):2076–2080
Acknowledgments
We thank Dr. Robert C. Long for his significant contribution in acquiring and processing the MRI data and for helpful scientific image interpretations discussions. We also thank the Pathology Core Lab of Winship Cancer Institute of Emory University for helping with all the histology data. This work was supported by grants from the NIH (NS053454), Southeastern Brain Tumor Foundation (SBTF), Georgia Cancer Coalition Distinguished Cancer Clinicians and Scientific Program, Robbins Scholar Award from the Winship Cancer Institute of Emory University, AANS/CNS Section on Tumors/BrainLab International Research Fellowship and Dana Foundation.
Conflict of interest
The authors declare no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bouras, A., Kaluzova, M. & Hadjipanayis, C.G. Radiosensitivity enhancement of radioresistant glioblastoma by epidermal growth factor receptor antibody-conjugated iron-oxide nanoparticles. J Neurooncol 124, 13–22 (2015). https://doi.org/10.1007/s11060-015-1807-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11060-015-1807-0