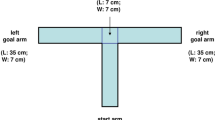
On training in an eight-channel radial maze, high-impulsivity animals found the reinforced arm and ate food more quickly than individuals capable of self-control and ambivalent rats. They made fewer errors of working memory in solving tasks linked with discriminating signal stimulus objects, though there were no differences in learning speed and the number of long-term memory errors in animals of different groups. During reversion training, when the reinforced arm of the maze was changed to another and vice versa, high-impulsivity animals spent less time exploring the reinforced maze arms than rats of the other groups. They made fewer signal working memory and long-term memory errors, though learning speed was not different between these and other groups of animals. The data obtained here provide evidence that high-impulsivity rats, as compared with low-impulsivity and ambivalent animals, showed higher levels of overall movement activity and displayed better expression of working memory.
Similar content being viewed by others
References
Albein-Urios, N., Martinez-Gonzalez, J. M., Lozano, et al., “Comparison of impulsivity and working memory in cocaine addiction and pathological gambling: Implications for cocaine-induced neurotoxicity,” Drug Alcohol Depend., 126, 1–6 (2012).
Ballard, K. and Knutson, B., “Dissociable neural representations of future reward magnitude and delay during temporal discounting,” Neuroimage, 45, 143–150 (2009).
Bickel, W. K., Jarmolowicz, D. P., Mueller, E. T., et al., “Excessive discounting of delayed reinforcers as a trans-disease process contributing to addiction and other disease-related vulnerabilities: Emerging evidence,” Pharmacol. Ther., 134, No. 3, 287–297 (2012).
Dellu-Hagedorn, E., “Relationship between impulsivity, hyperactivity and working memory: A differential analysis in the rat,” Behav. Brain Funct., 2, 10 (2006), doi:https://doi.org/10.1186/1744-9081-2-10.
Evenden, J. L. and Ryan, C. N., “The pharmacology of impulsive behaviour in rats: the effects of drugs on response choice with varying delays of reinforcement,” Psychopharmacology (Berlin), 128, No. 2, 161–170 (1996).
Franken, J. H., van Strien, J. W, Nijs, I., and Muris, P., “Impulsivity is associated with behavioral decision-making deficits,” Psychiatry Res., 158, No. 2, 155–63 (2008).
Grigor’yan, G. A. and Merzhanova, G. Kh., “Individual behavior in reinforcement prediction errors and environmental uncertainty,” Zh. Vyssh. Nerv. Deyat., 58, No. 4, 408–422 (2008).
Grigor’yan, G. A., “Serotonin and impulsivity,” Zh. Vyssh. Nerv. Deyat., 61, No. 3, 261–273 (2011).
Grigoryan, G. A., Weiss, I., and Feldon, I., “Social isolation improves working memory at reversal but not primary radial-arm learning in rats, “ Zh. Vyssh. Nerv. Deyat., 60, No. 5, 560–567 (2010).
Khurana, A., Ramer, D., Betancourt, L. M., et al., “Working memory ability predicts trajectories of early alcohol use in adolescents: The mediational role of impulsivity,” Addiction, 108, No. 3, 506–515 (2013).
Lebreton, M., Bertoux, M., Boutet, C., et al., “A critical role for the hippocampus in the valuation of imagined outcomes,” PLoS Biol., 2013, 10, e1001684 (2013), doi:https://doi.org/10.1371/journal.pbio.1001684.
Levandovskaya, A. A., Zaichenko, M. I., Merzhanova, G. Kh., and Salozhin, S. V., “Assessment of exploratory activity and anxiety in rats with different levels of impulsive behavior,” Zh. Vyssh. Nerv. Deyat., 63, No. 6, 719–729 (2013).
MacKillop, J., Amlung, M. T., Few, L. R., et al., “Delayed reward discounting and addictive behavior: A meta-analysis,” Psychopharmacology, 216, No. 3, 305–321 (2011).
Mariano, T. Y., Bannerman, D. M., McHugh, S. B., et al., “Impulsive choice in hippocampal but not orbitofrontal cortex-lesioned rats on a nonspatial decision-making maze task,” Eur. J. Neurosci., 3, 472–484 (2009).
Mazur, J. E., “An adjusting procedure for studying delayed reinforcement,” in: Quantitative Analyses of Behavior: The Effect of Delay and of Intervening Events on Reinforcement Value: Quantitative Analyses of Behavior, Commons, M. L. et al. (eds.), Lawrence Erlbaum, Hillsdale, N. J. (1987), Vol. 5, pp. 55–73.
McHugh, S. B., Campbell, T. G., Taylor, A. M., et al., “A role for dorsal and ventral hippocampus in inter-temporal choice cost-benefit decision making,” Behav. Neurosci., 122, No. 1, 1–8 (2008).
Merzhanova, G. Kh., Dolbakyan, E. E., and Khikhlova, V. N., “Organization of fronto-hippocampal neural networks in cats in different types of purposive behavior,” Zh. Vyssh. Nerv. Deyat., 54, No. 4, 508–518 (2004).
Merzhanova, G. Kh., “Impulsivity and Self-control in animals and humans during choice of behavioral strategy,” in: Psychology of Impulsivity, Cyders, M. A. (ed.), Nova (2012), Ch. 6, pp, 183–208.
Patros, C. H., Alderson, R. M., Lea, S. E., et al., “Visuospatial working memory underlies choice-impulsivity in boys with attention deficit/hyperactivity disorder,” Res. Dev. Disabil., 38, 134–144 (2015).
Perry, J. L., Nelson, S. E., and Carroll, M. E., “Impulsive choice as a predictor of acquisition of cocaine self- administration and reinstatement of cocaine-seeking behavior in male and female rats,” Exp. Clin. Psychopharm., 16, No. 2, 165–177 (2008).
Pigareva, M. L., “Facilitation of conditioned reflex switching of different types of conditioned reflexes in rats after hippocampal injury,” Zh. Vyssh. Nerv. Deyat., 20, No. 5, 932–940 (1970).
Renda, C. R., Stein, J. S., Madden, G. J., “Impulsive choice predicts poor working memory in male rats,” PloS One, 9, No. 4, e93263 (2014), doi:https://doi.org/10.1371/journal.pone.0093263.
Romer, D., Betancourt, L., Giannetta, J. M., et al., “Executive cognitive functions and impulsivity as correlates of risk taking and problem behavior in preadolescents,” Neuropsychologia, 47, 2916–2926 (2009).
Simonov, P. V., Lectures on the Operation of the Brain. An Informational-Needs Theory of Nervous Activity, Institute of Psychology, Russian Academy of Sciences, Moscow (1998).
Torregrossa, M. M., Xie, M., and Taylor, J. R., “Chronic corticosterone ex posure during adolescence reduces impulsive action but increases impulsive choice and sensitivity to yohimbine in male Sprague–Dawley rats,” Neuropsychopharmacology, 37, No. 7, 1656–1670 (2012).
Zaichenko, M. I. and Merzhanova, G. Kh., “Studies of the manifestations of impulsivity in rats in a food reinforcement selection situation,” Zh. Vyssh. Nerv. Deyat., 60, No. 1, 56–64 (2010).
Zaichenko, M. I., Bazhenova, D. A., Grigor’yan, G. A., and Merzhanova, G. Kh., “Does the property of impulsivity affect the operation of working and long-term memory in rats?” Zh. Vyssh. Nerv. Deyat., 66, No. 1, 82–91 (2016).
Zaichenko, M. I., Vanetsian, G. L., and Merzhanova, G. Kh., “Differences in the behavior of impulsive and self-controlled rats in studies using an open field test and a light/dark chamber,” Zh. Vyssh. Nerv. Deyat., 61, No. 3, 340–345 (2011).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Zhurnal Vysshei Nervnoi Deyatel’nosti imeni I. P. Pavlova, Vol. 66, No. 5, pp. 600–610, September–October, 2016.
Rights and permissions
About this article
Cite this article
Zaichenko, M.I., Sharkova, A.V., Grigor’yan, G.A. et al. Memory in an Eight-Channel Radial Maze is Better in High-Impulsivity Rats Than Low-Impulsivity Animals. Neurosci Behav Physi 48, 109–117 (2018). https://doi.org/10.1007/s11055-017-0538-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-017-0538-4