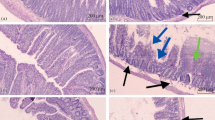
The aim of the present work was to investigate somatic pain sensitivity in conscious rats exposed to the harmful actions of indomethacin on the gastrointestinal tract (GIT). Indomethacin was given at an ulcerogenic dose (35 mg/kg, s.c.) to previously starved (24 h) and sated (not starved) rats. Somatic pain sensitivity was evaluated in terms of the latent period (LP) of the pain reaction induced by thermal stimulation of the tail (the tailflick test). LP was tested during the formation of erosions in the stomach (1–4 h after indomethacin administration) and during the formation of lesions in the small intestine (24, 48, and 72 h after drug administration). Indomethacin administration induced gastric erosions at 4 h only in starved rats, but produced lesions in the small intestine at 24, 48, and 72 h both starved and sated rats. Lesions in both the stomach and the small intestine were accompanied by increases in the LP of the pain reaction. There were no increases in LP when indomethacin administration did not have marked damaging effects on the GIT. Development of the pathological process in the GIT was accompanied by symptoms of chronic stress: a prolonged increase in the blood corticosterone level, adrenal hypertrophy, thymus involution, and weight loss. Thus, somatic pain sensitivity was reduced in conscious rats exposed to the harmful effects of indomethacin on the stomach and intestine.
Similar content being viewed by others
References
A. A. Filaretov, A. I. Bogdanov, and N. I. Yarushkina, “Stress-induced analgesia. The role of hypophyseal-adrenocortical system hormones,” Ros. Fiziol. Zh., 81, No. 2, 40–46 (1995).
N. I. Yarushkina, “The role of the hypothalamo-hypophyseal-adrenocortical system in regulating pain sensitivity,” Ros. Fiziol. Zh., 93, No. 11, 1252–1262 (2007).
N. I. Yarushkina, T. R. Bagaeva, and L. P. Filaretov, “Mechanisms of the analgesic action of corticotropin-releasing factor on pain sensitivity in rats,” Ros. Fiziol. Zh., 99, No. 7, 806–819 (2013).
L. C. Bastos and C. R. Tonussi, “PGE(2)-induced lasting nociception to heat: evidence for a selective involvement of A-delta fibres in the hyperpathic component of hyperalgesia,” Eur. J. Pain, 14, No. 2, 113–119 (2010).
J. A. DeLeo, R. W. Colburn, D. W. Coombs, and M. A. Ellis, “The differentiation of NSAIDs and prostaglandin action using a mechanical visceral pain model in the rat,” Pharmacol. Biochem. Behav., 33, No. 1, 253–255 (1989).
A. Dogrul, S. Ezgi Gulmez, S. Deveci, et al., “Anti-inflammatory drugs in the mouse radiant heat tail-flick test,” Anesth. Analg., 104, No. 4, 927–935 (2007).
L. Filaretova, “Gastroprotective role of glucocorticoids during NSAID-induced gastropathy,” Curr. Pharm. Design, 19, No. 1, 29–33 (2013).
L. Filaretova, T. Bagaeva, and G. Makara, “Aggravation of nonsteroidal antiinflammatory drug gastropathy by glucocorticoid deficiency or blockade of glucocorticoid receptors in rats,” Life Sci., 71, No. 21, 2457–2468 (2002).
L. Filaretova, T. Podvigina, T. Bagaeva, G. Makara, “Gastroprotective action of glucocorticoids during the formation and the healing of indomethacin-induced gastric erosions in rats,” Life Sci. Physiol. (Paris), 95, No. 1–6, 201–208 (2001).
L. Filaretova, A. Tanaka, and T. Miyazawa, “Mechanisms by which endogenous glucocorticoid protects against indomethacin-induced gastric injury in rats,” Am. Physiol. Gastrointest. Liver Physiol., 283, No. 5, G1082–1089 (2002).
L. P. Filaretova, T. R. Bagaeva, O. Yu. Morozova, and D. Zelena, “The healing of NSAID-induced gastric lesion may be followed by small intestinal and cardiovascular side effects,” Physiol. Pharmacol., 62, No. 6, 619–625 (2011).
N. Friese, L. Diop, E. Chevalier, et al., “Involvement of prostaglandins and CGRP-dependent sensory afferents in peritoneal irritation-induced visceral pain,” Regul. Pept., 70, No. 1, 1–7 (1997).
R. J. Hamm, J. S. Knisely, A. Watson, et al., “Hormonal mediation of the analgesia produced by food deprivation,” Physiol. Behav., 35, No. 6, 879–882 (1985).
S. K. Joshi and G. F. Gebhart, “Visceral pain,” Curr. Rev. Pain, 4, No. 6, 499–506 (2000).
J. M. Laird, L. Martinez-Caro, E. Garcia-Nicas, and F. Cervero, “A new model of visceral pain and referred hyperalgesia in the mouse,” Pain, 92, No. 3, 335–342 (2001).
T. J. Ness and G. F. Gebhart, “Interactions between visceral and cutaneous nociception in the rat. II. Noxious visceral stimuli inhibit cutaneous nociceptive neurons and reflexes,” Neurophysiol., 66, No. 1, 29–39 (1991).
M. I. Ortiz, “Blockade of the antinociception induced by diclofenac but not of indomethacin by sulfonylureas and biguanides,” Pharmacol. Biochem. Behav., 99, No. 1, 1–6 (2011).
F. Pinto-Ribeiro, A. Almeida, J. M. Pego, et al., “Chronic unpredictable stress inhibits nociception in male rats,” Neurosci. Lett., 359 No.1–2, 73–76 (2004).
K. Takayama, A. Hirose, I. Suda, et al., “Comparison of the anti-inflammatory and analgesic effects in rats of diclofenac-sodium felbinac and indomethacin patches,” Int. Biomed. Sci., 7, No. 3, 222–229 (2011).
K. Takeuchi, A. Tanaka, Y. Hayashi, and A. Yokota, “COX inhibition and NSAID-induced gastric damage – roles in various pathogenic events,” Curr. Top. Med. Chem., 5, No. 5, 475–486 (2005).
R. J. Traub and G. Wang, “Colonic inflammation decreases thermal sensitivity of the forepaw and hindpaw in the rat,” Neurosci. Lett., 359, No. 1–2, 81–84 (2004).
J. L. Wallace, “NSAID gastropathy and enteropathy: distinct pathogenesis likely necessitates distinct prevention strategies,” Br. Pharmacol., 165, No. 1, 57–74 (2012).
N. I. Yarushkina, N. I. Bogdanov, and L. P. Filaretova, “Somatic pain sensitivity during formation and healing of acetic acid-induced gastric ulcers in conscious rats,” Autonom. Neurosci. Basic and Clinical, 126–127, No. 1–2, 100–105 (2006).
Q. Q. Zhou, D. D. Price, R. M. Caudle, and G. N. Verne, “Visceral and somatic hypersensitivity in TNBS induced colitis in rats,” Dig. Dis. Sci., 53, No. 2, 429–435 (2008).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Rossiiskii Fiziologicheskii Zhurnal imeni I. M. Sechenova, Vol. 100, No. 1, pp. 73–85, January, 2014.
Rights and permissions
About this article
Cite this article
Yarushkina, N.I., Bagaeva, T.R. & Filaretova, L.P. Somatic Pain Sensitivity in Rats Exposed to the Harmful Actions of Indomethacin on the Gastrointestinal Tract. Neurosci Behav Physi 45, 780–788 (2015). https://doi.org/10.1007/s11055-015-0144-2
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-015-0144-2