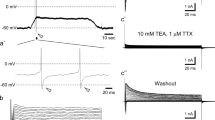
Patch clamp experiments in the whole cell configuration were performed on isolated rat prefrontal cortex neurons to study the summation of ion currents evoked by application of glutamate and GABA. Ion currents were recorded using two different pipette solutions, based on cesium chloride and fluoride. In recordings made using the cesium chloride-based solution, the peak amplitude of the current evoked by simultaneous application of GABA and glutamate (each at 200 μM) coincided with the peak amplitude of the current evoked by application of GABA alone, and was significantly smaller than the arithmetic sum of the responses to application of the two neurotransmitters individually. When the pipette solution based on cesium fluoride was used, the response to simultaneous application of glutamate and GABA was essentially the same as the arithmetic sum of the individual responses. On exposure to these neurotransmitters at saturating concentrations (5 mM), the response recorded to simultaneous application was significantly smaller than the response to application of GABA alone. These results suggest that there is a mechanism of interaction between GABAA and ionotropic glutamate receptors (AMPA and kainate). On simultaneous application of glutamate and GABA, activation of GABAA receptors evidently has a greater influence on glutamate receptors than activation of glutamate receptors of GABAA receptors.
Similar content being viewed by others
References
D. V. Amakhin and N. P. Veselkin, “Interaction of the effects of the neurotransmitters glycine and GABA in the central nervous system,” Tsitologiya, 54, No. 6, 469–477 (2012).
D. V. Amakhin and N. P. Veselkin, “Mechanisms of interaction of GABA- and glycine-mediated responses of frog spinal cord neurons,” Ros. Fiziol. Zh., 97, No. 10, 1025–1041 (2011).
D. V. Amakhin and N. P. Veselkin, “Characteristics and interaction of GABAergic and glycinergic processes in frog spinal cord neurons,” Ros. Fiziol. Zh., 95, No. 4, 313–323 (2009).
N. P. Veselkin and Yu. V. Natochin, “Principles of the organization and evolution of function-regulating systems,” Zh. Evolyuts. Biokhim. Fiziol., 46, No. 6, 495–503 (2010).
K. V. Baev, K. I. Rusin, and B. V. Safronov, “Primary receptor for inhibitory transmitters in lamprey spinal cord neurons,” Neuroscience, 46, No. 4, 931–941 (1992).
C. Barajas-Lopez, R. Espinosa-Luna, and Y. Zhu, “Functional interactions between nicotinic and P2X channels in short-term cultures of guinea-pig submucosal neurons,” J. Physiol., 513, No. 3, 671–683 (1998).
J. Q. Beltran and R. Gutierrez, “Co-release of glutamate and GABA from single, identified mossy fiber giant boutons,” 590, No. 19, 47898–4800 J. Physiol., (2012).
L. Bertollini, G. Biella, E. Wanke, et al., “Fluoride reversibly blocks HVA calcium current in mammalian thalamic neurons,” Neuroreport, 5, No. 5, 553–556 (1994).
F. Boue-Grabot, C. Barajas-Lopez, Y. Chafke, et al., “Intracellular cross talk and physical interaction between two classes of neurotransmitter-gated channels,” J. Neurosci., 23, No. 4, 1246–1253 (2003).
X. B. Gao, G. Chen, and A. N. van den Pol, “GABA-dependent firing of glutamate-evoked action potentials at AMPA/kainate receptors in developing hypothalamic neurons,” J. Neurophysiol., 79, No. 2, 716–726 (1998).
F. Grassi, “Cl(–)-mediated interaction between GABA and glycine currents in cultured rat hippocampal neurons,” Brain Res., 594, No. 1, 115–123 (1992).
U. Karlson, M. Druzin, and S. Johansson, “Cl(–) concentration changes and desensitization of GABA(A) and glycine receptors,” J. Gen. Physiol., 138, No. 6, 609–626 (2011).
B. S. Khakh, X. Zhou, J. Sydes, et al., “State-dependent cross-inhibition between transmitter-gated cation channels,” Nature, 406, No. 6794, 405–410 (2000).
Y. A. Kuryshev, A. P. Naumov, P. V. Avdonin, and G. N. Mozhayeva, “Evidence for involvement of a GTP-binding protein in activation of Ca2+ influx by epidermal growth factor in A431 cells: effects of fluoride and bacterial toxins,” Cell Signal, 5, No. 5, 555–564 (1993).
Y. Li, L. J. Wu, P. Legendre, and T. L. Xu, “Asymmetric cross-inhibition between GABAA and glycine receptors in rat spinal dorsal horn neurons,” J. Biol. Chem., 278, No. 40, 38,637–38,645 (2003).
Y. Li and T. L. Xu, “State-dependent cross-inhibition between anionic GABA(A) and glycine ionotropic receptors in rat hippocampal CA1 neurons,” Neuroreport, 13, No. 2, 223–226 (2002).
T. Narahashi, X. Zhao, T. Ikeda, et al., “Glutamate-activated chloride channels: Unique fipronil targets present in insects but not in mammals,” Pestic. Biochem. Physiol., 97, No. 2, 149–152 (2010).
J. Noh, R. P. Seal, J. A. Garver, et al., “Glutamate co-release at GABA/glycinergic synapses is crucial for the refinement of an inhibitory map,” Nat. Neurosci., 13, No. 2, 232–238 (2010).
A. S. Piper and R. J. Docherty, “One-way cross-desensitization between P2X purinoceptors and vanilloid receptors in adult rat dorsal root ganglion neurones,” J. Physiol., 523, No. 3, 685–696 (2000).
A. Poleg-Polsky and J. S. Diamond, “Imperfect space clamp permits electrotonic interactions between inhibitory and excitatory synaptic conductances, distorting voltage clamp recordings,” PLoS One, 6, No. 4, e19463 (2011).
M. Russier, I. L. Kopysova, N. Ankri, et al., “GABA and glycine corelease optimizes functional inhibition in rat brainstem motoneurons in vitro,” J. Physiol., 541, No. 1, 123–137 (2002).
N. E. Schwartz and S. Alford, “Physiological activation of presynaptic metabotropic glutamate receptors increases intracellular calcium and glutamate release,” J. Neurophysiol., 84, No. 1, 415–427 (2000).
R. P. Seal and R. H. Edwards, “Functional implications of neurotransmitter co-release: glutamate and GABA share the load,” Curr. Opin. Pharmacol., 6, No. 1, 114–119 (2006).
T. J. Searl and E. M. Silinsky, “Cross-talk between apparently independent receptors,” J. Physiol., 513, No. 3, 629–630 (1998).
A. N. Shrivastava, A. Triller, and W. Sieghart, “GABA(A) receptors: postsynaptic co-localization and cross-talk with other receptors,” Front. Cell. Neurosci., 5, No. 7, (2011).
E. Sokolova, A. Nistri, and R. Giniatullin, “Negative cross talk between anionic GABAA and cationic P2X receptors of rat dorsal root ganglion neurons,” J. Neurosci., 21, No. 14, 4958–4968 (2001).
C. H. Song, X. W. Chen, J. X. Xia, et al., “Modulatory effects of hypocretin-1/orexin-A with glutamate and gamma-aminobutyric acid on freshly isolated pyramidal neurons from the rat prefrontal cortex,” Neurosci. Lett., 399, No. 1–2, 101–105 (2006).
K. J. Staley and I. Mody, “Shunting of excitatory input to dentate gyrus granule cells by a depolarizing GABAA receptor-mediated postsynaptic conductance,” J. Neurophysiol., 68, No. 1, 197–212 (1992).
E. Toulme, D. Blais, C. Leger, et al., “An intracellular motif of P2X(3) receptors is required for functional cross-talk with GABA(A) receptors in nociceptive DRG neurons,” J. Neurochem., 102, No. 4, 1357–1368 (2007).
P. Q. Trombley, B. J. Hill, and M. S. Horning, “Interactions between GABA and glycine at inhibitory amino acid receptors on rat olfactory bulb neurons,” J. Neurophysiol., 82, No. 6, 3417–3422 (1999).
G. Vargas, T. Y. Yeh, D. K. Blumenthal, and M. T. Lucero, “Common components of patch-clamp internal recording solutions can significantly affect protein kinase A activity,” Brain Res., 828, No. 1–2, 169–173 (1999).
V. S. Vorobjev, I. N. Sharonova, and H. L. Haas, “A simple perfusion system for patch-clamp studies,” J. Neurosci. Meth., 68, No. 2, 303–307 (1996).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Rossiiskii Fiziologicheskii Zhurnal imeni I. M. Sechenova, Vol. 98, No. 12, pp. 1490–1506, December, 2012.
Rights and permissions
About this article
Cite this article
Amakhin, D.V., Popov, V.A., Malkiel’, A.I. et al. Summation of GABA- and Glutamate-Mediated Ion Currents in Isolated Rat Cerebral Cortex Neurons. Neurosci Behav Physi 44, 828–838 (2014). https://doi.org/10.1007/s11055-014-9990-6
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11055-014-9990-6