Abstract
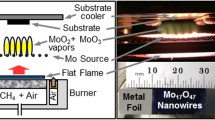
Molybdenum trioxide nanostructures attract great interest for their potential applications in areas of electrode in batteries, semiconductors, electrochromic devices, and other advanced material fields. Herein we disclosed the atmospheric, catalyst-free, scalable, rapid, and morphology-controllable flame synthesis of a series α-MoO3 nanostructures from metallic molybdenum. Controlling the morphology and size of the high-quality nanostructures by means of modulating various combustion configurations including fuel/oxygen ratio, height of growth substrates, and density of molybdenum wire mesh source were realized and discussed in detail. A series of characteristic morphologies of α-MoO3 nanostructures including layered nanobelts, nanoplates, and nanoflakes doping with nanocubics were fabricated via specific combustion conditions. These findings provide favorable access to the rapid and large-scale synthesis of high-performance nanostructure materials and may be widely used in the foreseeable future.
Graphical abstract
Flame vapor deposition (FVD) method provides a robust approach for mass-production of α-MoO3 nanostructures. The morphology of α-MoO3 layered nanostructures can be controllable fabricated by altering the combustion conditions. Herein, a series of characteristic morphology of α-MoO3 nanostructures including layered nanobelts, nanoplates, and nanoflakes doping with nanocubics were realized via specific combustion conditions.











Similar content being viewed by others
References
Pham K-C, McPhail DS, Wee ATS, Chua DHC (2017) Amorphous molybdenum sulfide on graphene–carbon nanotube hybrids as supercapacitor electrode materials. RSC Adv 7(12):6856–6864. https://doi.org/10.1039/c6ra27901e
Brezesinski T, Wang J, Tolbert SH, Dunn B (2010) Ordered mesoporous alpha-MoO3 with iso-oriented nanocrystalline walls for thin-film pseudocapacitors. Nat Mater 9(2):146–151. https://doi.org/10.1038/nmat2612
Murase S, Yang Y (2012) Solution processed MoO3 interfacial layer for organic photovoltaics prepared by a facile synthesis method. Adv Mater 24(18):2459–2462. https://doi.org/10.1002/adma.201104771
Ahmed J, Alam M, Khan MAM, Alshehri SM (2021) Bifunctional electro-catalytic performances of NiMoO4-NRs@RGO nanocomposites for oxygen evolution and oxygen reduction reactions. Journal of King Saud University – Science 33:101317–101322. https://doi.org/10.1016/j.jksus.2020.101317
Ahmed J, Ubiadullah M, Khan MAM, Alhokbany N, Alshehri SM (2021) Significant recycled efficiency of multifunctional nickel molybdenum oxide nanorods in photo-catalysis, electrochemical glucose sensing and asymmetric supercapacitors. Mater Charact 171:110741. https://doi.org/10.1016/j.matchar.2020.110741
Ahmed J, Ubaidullah M, Ahmad T, Alhokbany N, Alshehri SM (2019) Synthesis of graphite oxide/cobalt molybdenum oxide hybrid nanosheets for enhanced electrochemical performance in supercapacitors and the oxygen evolution reaction. Chem Electro Chem 6:2524–2530. https://doi.org/10.1002/celc.201900055
Ahmed J, Khan MAM, Alshehri SM (2021) Zinc molybdenum oxide sub-micron plates as electro-catalysts for hydrogen evolution reactions in acidic medium. Mater Lett 284:128996. https://doi.org/10.1016/j.matlet.2020.128996
Zhao G, Zhang N, Sun K (2013) Electrochemical preparation of porous MoO3film with a high rate performance as anode for lithium ion batteries. J Mater Chem A 1(2):221–224. https://doi.org/10.1039/c2ta00361a
Kim HS, Cook JB, Lin H, Ko JS, Tolbert SH, Ozolins V, Dunn B (2017) Oxygen vacancies enhance pseudocapacitive charge storage properties of MoO3-x. Nat Mater 16(4):454–460. https://doi.org/10.1038/nmat4810
Hu X, Zhang W, Liu X, Mei Y, Huang Y (2015) Nanostructured Mo-based electrode materials for electrochemical energy storage. Chem Soc Rev 44(8):2376–2404. https://doi.org/10.1039/c4cs00350k
Lou X, Zeng H (2002) Hydrothermal synthesis of r-MoO3 nanorods via acidification of ammonium heptamolybdate tetrahydrate. Chem Mater 14(11):4781–4789
Swiatowska-Mrowiecka J, de Diesbach S, Maurice V, Zanna S, Klein L, Briand E, Vickridge I, Marcus P (2008) Li-ion intercalation in thermal oxide thin films of MoO3 as studied by XPS, RBS, and NRA. J Phys Chem C 112(29):11050–11058. https://doi.org/10.1021/jp800147f
Labanowska M (2001) EPR monitoring of redox processes in transition metal oxide catalysts. Chem Phys Chem 2(12):712–731
Comini E, Yubao L, Brando Y, Sberveglieri G (2005) Gas sensing properties of MoO3 nanorods to CO and CH3OH. Chem Phys Lett 407(4–6):368–371. https://doi.org/10.1016/j.cplett.2005.03.116
Zhou J, Lin N, Wang L, Zhang K, Zhu Y, Qian Y (2015) Synthesis of hexagonal MoO3 nanorods and a study of their electrochemical performance as anode materials for lithium-ion batteries. J Mater Chem A 3(14):7463–7468. https://doi.org/10.1039/c5ta00516g
Ranjba M, Delalat F, Salamati H (2017) Molybdenum oxide nanosheets prepared by an anodizing-exfoliation process and observation of photochromic properties. Appl Surf Sci 396:1752–1759. https://doi.org/10.1016/j.apsusc.2016.11.225
Tang W, Liu L, Tian S, Li L, Yue Y, Wu Y, Zhu K (2011) Aqueous supercapacitors of high energy density based on MoO3 nanoplates as anode material. Chem Commun (camb) 47(36):10058–10060. https://doi.org/10.1039/c1cc13474d
Hashem AM, Groult H, Mauger A, Zaghib K, Julien CM (2012) Electrochemical properties of nanofibers α-MoO3 as cathode materials for Li batteries. J Power Sources 219:126–132. https://doi.org/10.1016/j.jpowsour.2012.06.093
Nadimicherla R, Liu Y, Chen K, Chen W (2014) Electrochemical performance of new α-MoO3 nanobelt cathode materials for rechargeable Li-ion batteries. Solid State Sci 34:43–48. https://doi.org/10.1016/j.solidstatesciences.2014.05.012
Riley LA, Lee S-H, Gedvilias L, Dillon AC (2010) Optimization of MoO3 nanoparticles as negative-electrode material in high-energy lithium ion batteries. J Power Sources 195(2):588–592. https://doi.org/10.1016/j.jpowsour.2009.08.013
Zhou K, Zhou W, Liu X, Sang Y, Ji S, Li W, Lu J, Li L, Niu W, Liu H, Chen S (2015) Ultrathin MoO 3 nanocrystalsself-assembled on graphene nanosheets via oxygen bonding as supercapacitor electrodes of high capacitance and long cycle life. Nano Energy 12:510–520. https://doi.org/10.1016/j.nanoen.2015.01.017
Rao PM, Cho IS, Zheng X (2013) Flame synthesis of WO3 nanotubes and nanowires for efficient photoelectrochemical water-splitting. Proc Combust Inst 34(2):2187–2195. https://doi.org/10.1016/j.proci.2012.06.122
Xu F, Liu X, Tse SD, Cosandey F, Kear BH (2007) Flame synthesis of zinc oxide nanowires. Chem Phys Lett 449(1–3):175–181. https://doi.org/10.1016/j.cplett.2007.10.045
Strobel R, Pratsinis SE (2007) Flame aerosol synthesis of smart nanostructured materials. J Mater Chem 17 (45). https://doi.org/10.1039/b711652g
Rao PM, Zheng X (2011) Flame synthesis of tungsten oxide nanostructures on diverse substrates. Proc Combust Inst 33(2):1891–1898. https://doi.org/10.1016/j.proci.2010.06.071
Rao PM, Zheng X (2011) Unique magnetic properties of single crystal gamma-Fe2O3 nanowires synthesized by flame vapor deposition. Nano Lett 11(6):2390–2395. https://doi.org/10.1021/nl2007533
Merchan-Merchan W, Saveliev AV, Taylor AM (2008) High rate flame synthesis of highly crystalline iron oxide nanorods. Nanotechnology 19(12):125605. https://doi.org/10.1088/0957-4484/19/12/125605
Chang MT, Chou LJ, Chueh YL, Lee YC, Hsieh CH, Chen CD, Lan YW, Chen LJ (2007) Nitrogen-doped tungsten oxide nanowires: low-temperature synthesis on Si, and electrical, optical, and field-emission properties. Small 3(4):658–664. https://doi.org/10.1002/smll.200600562
Cai L, Rao PM, Zheng X (2011) Morphology-controlled flame synthesis of single, branched, and flower-like alpha-MoO3 nanobelt arrays. Nano Lett 11(2):872–877. https://doi.org/10.1021/nl104270u
Allen P, Cai L, Zhou L, Zhao C, Rao PM (2016) Rapid synthesis of thin and long Mo17O47 nanowire-arrays in an oxygen deficient flame. Sci Rep 6:27832. https://doi.org/10.1038/srep27832
Cai L, McClellan CJ, Koh AL, Li H, Yalon E, Pop E, Zheng X (2017) Rapid flame synthesis of atomically thin MoO3 down to monolayer thickness for effective hole doping of WSe2. Nano Lett 17(6):3854–3861. https://doi.org/10.1021/acs.nanolett.7b01322
Koirala R, Pratsinis SE, Baiker A (2016) Synthesis of catalytic materials in flames: opportunities and challenges. Chem Soc Rev 45(11):3053–3068. https://doi.org/10.1039/c5cs00011d
Chen J, Cheah Y, Madhavi S, Lou X (2010) Fast synthesis of r-MoO3 nanorods with controlled aspect ratios and their enhanced lithium storage capabilities. J Phys Chem C 114(18):8675–8678
Seguin L, Figlarz M, Cavagnat R, Lassegues LC (1995) Infrared and Raman spectra of MoO3 molybdenum trioxides and MoO3 · xH2O molybdenum trioxide hydrates. Sepctrochimica Acta Part A 51:1323–1344
Funding
This work was supported by the National Natural Science Foundation of China (No. 51676176, 51976207).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhong, Z., Zhang, L. Optimized rapid flame synthesis of morphology-controlled α-MoO3 layered nanoflakes. J Nanopart Res 23, 115 (2021). https://doi.org/10.1007/s11051-021-05236-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11051-021-05236-y