Abstract
Background
The global prevalence of invasive fungal infections (IFI) is increasing, particularly within Intensive Care Units (ICU), where Candida spp. and Aspergillus spp. represent the most important pathogens. Diagnosis and management of IFIs becomes progressively challenging, with increasing antifungal resistance and the emergence of rare fungal species.
Through a consensus survey focused on assessing current views on how IFI should be managed, the aim of this project was to identify challenges around diagnosing and managing IFIs in the ICU. The current status in different countries and perceived challenges to date amongst a multidisciplinary cohort of healthcare professionals involved in the care of IFI in the ICU was assessed.
Methods
Using a modified Delphi approach, an expert panel developed 44 Likert-scale statements across 6 key domains concerning patient screening and minimal standards for diagnosis of IFIs in ICU; initiation and termination of antifungal treatments and how to minimise their side effects and insights for future research on this topic. These were used to develop an online survey which was distributed on a convenience sampling basis utilising the subscriber list held by an independent provider (M3 Global). This survey was distributed to intensivists, infectious disease specialists, microbiologists and antimicrobial/ICU pharmacists within the UK, Germany, Spain, France and Italy. The threshold for consensus was set at 75%.
Results
A total of 335 responses were received during the five-month collection period. From these, 29/44 (66%) statements attained very high agreement (≥ 90%), 11/44 (25%) high agreement (< 90% and ≥ 75%), and 4/44 (9%) did not meet threshold for consensus (< 75%).
Conclusion
The results outline the need for physicians to be aware of the local incidence of IFI and the associated rate of azole resistance in their ICUs. Where high clinical suspicion exists, treatment should start immediately and prior to receiving the results from any diagnostic test. Beta-D-glucan testing should be available to all ICU centres, with results available within 48 h to inform the cessation of empirical antifungal therapy. These consensus statements and proposed measures may guide future areas for further research to optimise the management of IFIs in the ICU.
Similar content being viewed by others
Avoid common mistakes on your manuscript.
Background
In recent years the global prevalence of invasive fungal infections (IFIs) has increased across many clinical environments but particularly within intensive care units (ICU). [1,2,3] For instance, over the past decade the rate of candidaemia has increased in some settings by up to 50% [2] with an incidence of 5.1/1000 ICU admissions. [3]
Infections from Candida species and invasive aspergillosis (IA) represent a critical issue for patients within an ICU. Candida species are responsible for approximately 80% of IFI infections [4, 5] and candidaemia, the most common clinical presentation of invasive candidiasis, has a high mortality ranging from 28–59%. [6] Indeed, candidaemia accounted for 7.7% of cases in a large multicentre epidemiological study of hospital-acquired bloodstream infection in the ICU. [7] In recent years, infections by Aspergillus spp. in the form of IA have been frequently reported in critically ill patients, especially in patients with severe COVID-19, influenza, or chronic obstructive pulmonary disease (COPD), as well as the very old and critically ill. [8,9,10,11]
Patients admitted to an ICU with severe influenza or COVID-19 introduce a new set of challenges. These patients are at a high risk of developing an IFI due to factors such as epithelial damage caused by viral infection, mechanical ventilation, and immunosuppression. [12, 13] Aside from COVID-19 associated pulmonary aspergillosis (CAPA), COVID-19 associated mucormycosis (CAM), and Pneumocystis jirovecii pneumonia (PJP) (all of which increase patient mortality) [12, 14], infections with rare moulds and rare yeast have also been reported [12] increasing difficulties in patient management.
Depending on the presence of risk factors, underlying diseases and geographic location, IA may occur in 1 to 20% of ICU patients [4], and (in the absence of viral pandemics) approximately 43–80% of these do not have a haematological malignancy. [5] Due to the absence of specific clinical and radiological findings, diagnosis remains a challenge, as shown in a 2019 autopsy study where only 40% of IA cases were diagnosed in vivo [15]. In the absence of early diagnosis, mortality for IA cases is up to 80%. [6]
The epidemiology of IFI is changing as multi-resistant strains of not only Candida but also Aspergillus are emerging. [16, 17] The observed increase of non-albicans Candida spp., often showing resistance to azoles, is also a major concern for prolonged hospitalizations. [18,19,20] There is an increasing incidence of rare mould and yeast infections which have previously been geographically restricted. [17] This is thought to be caused by advances in diagnostics, allowing further differentiation and detection of previously unknown pathogens as well as broad usage of antifungals. Finally, the concern of climate change resulting in the emergence of new fungal pathogens that are adapting to human temperature, like Candida auris [1, 21].
Being able to treat an IFI rapidly is important for survival, yet may be hampered by the complexity of obtaining an accurate diagnosis. [6, 22] Apart from candidaemia, tissue sampling with histological examination remains the gold standard but is often not possible while patients are still alive. In addition, sensitivity of culture is usually below 50%. [22] In the absence of a reliable gold standard, non-culture-based tests are increasingly used for diagnosis of IFI in the ICU. The most important tests utilized are beta-D-glucan (BDG) for invasive Candida infections (sensitivity 77%, specificity 85%) [23], and galactomannan (GM) from bronchioalveolar lavage fluid (BALF) for invasive aspergillosis including viral associated pulmonary aspergillosis (sensitivity 75–86%, specificity 94–95%). [24, 25] The availability of these tests varies across Europe, and the tests are suboptimal in terms of sensitivity and specificity. [1, 22] The patient population admitted to ICU is heterogenous, and infected individuals often present with non-specific signs, symptoms, and radiological findings. [22]
Building on previous work in this area [1, 26], the aim of this consensus survey was to: (i) gauge the views of a multidisciplinary cohort of healthcare professionals (HCPs) regarding the current clinical reality of management; (ii) describe what is the optimal care of IFIs in the ICU; and (iii) ultimately identify remaining challenges around diagnosing and managing IFIs in the ICU.
Methods
A multidisciplinary panel (the study authors) from across Western Europe, selected based on their publications, leadership in international scientific societies, clinical expertise, and geographical representation of the countries where the survey was validated, convened in May 2022 to discuss remaining challenges in the management of IFIs in ICUs.
Using a modified Delphi methodology and a structured meeting format guided by an independent facilitator (Triducive Partners Limited, St Albans, UK), the expert panel identified 6 main domains of focus:
-
A)
Increasing IFI suspicion (patient characteristics/co-morbidities) and screening
-
B)
Accessibility and minimum standard for diagnosis
-
C)
Initiation and change of treatment
-
D)
Termination of treatment
-
E)
Managing side effects and drug-drug interactions
-
F)
Future guidance and data requirements
These domains were developed and discussed in detail by the expert panel during the initial project meeting and an initial set of statements were developed. The initial statements were then re-circulated amongst the members of the expert panel for individual ratification and amendment. A set of 44 statements were finally agreed for wider testing amongst ICU HCPs. These statements were used to form an online survey, which was distributed to ICU HCPs (intensivists, infectious disease specialists, microbiologists, and antimicrobial and/or ICU pharmacists) in five of the largest European markets (France, Germany, Italy, Spain and the UK) typically grouped as a source of data for European work.
The survey was shared on a convenience sampling basis by an independent company (M3 Global, London, UK) to their database of subscribers. Responses were sought using an incentivised methodology through standard market research approach. The invitation was unique and could not be redistributed. Each respondent was only able to complete the survey once. [27] Respondents were screened before completing the survey as to their level of experience within ICU departments and what familiarity they had with treating and diagnosing IFIs. To be eligible to take part in the survey, physicians would need a minimum of 2–5 years’ experience in their speciality as an intensivist or infectious diseases specialist, and a minimum of 5 years’ experience as a pharmacist or microbiologist.
Triducive Partners Limited independently managed meeting logistics, facilitation, structuring of information flow and survey management to ensure the anonymity of responders.
Stopping criteria were established as a minimum of 50 responses from each country surveyed, and a five-month window to collect responses (September 2022 to January 2023). A threshold for consensus was agreed at 75%. This was further defined as ‘high’ at ≥ 75% to < 90% and ‘very high’ at ≥ 90%.
The questionnaire was composed of each statement along with a 4-point Likert scale (‘strongly disagree’, ‘tend to disagree’, ‘tend to agree’, and ‘strongly agree’) to allow respondents to indicate their corresponding level of agreement. The questionnaire also captured some demographic data on country and role for further analysis. A statement of consent was included at the start of the survey, and consent was implied by completion and submission of the survey. As this study only sought the anonymous opinions of HCPs and no patient specific data was captured, ethical approval was deemed not required.
Completed surveys were anonymously collated and analysed by the independent facilitator to produce an arithmetic agreement score for each statement. The result was reviewed by the panel who subsequently proposed a set of measures aimed to address the challenges identified by the consensus survey.
Results
Following from the initial meeting held in May 2022 an initial set of 37 statements was constructed for review by the expert panel. During the independent review and approval stages, this set was refined to a final approved set of 44 statements which was then developed into the survey used within this study (Table 1).
During the five-month period to collect responses, a total of 335 responses were received. The number of responses received per country are displayed in Fig. 1A, responses according to medical specialisation in Fig. 1B, with the majority of responses received from intensivists (n = 145), followed by antimicrobial and/or ICU pharmacists (n = 95). From these responses, 29/44 (66%) statements attained very high agreement (≥ 90%), 11/44 (25%) high agreement (< 90% and ≥ 75%). The remaining 4/44 (9%) did not meet the established threshold for consensus (< 75%). The full set of statements and corresponding levels of agreement are shown in Table 1.
After review of the results, and given the high level of consensus achieved for 40/44 statements (91%) and that the stopping criteria had been met, the panel agreed that a single round of testing was sufficient for the purposes of this study. The panel agreed that statements which did not achieve consensus in this round should be reported as such and that for the purposes of this study should not be amended and resubmitted to responders.
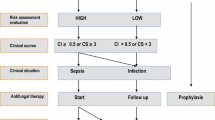
The results of the survey show a high degree of agreement across the consensus topics (Figs. 2, 3), with respondents showing strong consensus around the patient characteristics of IFI cases (Statements 1–9). HCPs also strongly agreed with the need to initiate treatment as quickly as possible where IFI is suspected, (Statements 17–30), and highlighted the need for greater evidence requirements in areas like incidence of IFIs and antifungal prophylaxis (Statements 37–44).
Upon analysis of results, clear trends emerged. When examining response by country (Fig. S1), the UK had the highest average level of agreement to the statements compared to France and Germany, which tended to be lower.
Analysis of response by speciality (Fig. S2) revealed some clear differences in agreement level, for example, response to Statement 21 (voriconazole is not a suitable treatment option for ICU patients) amongst infectious disease specialists and intensivists showed the lowest level of agreement (32% and 34% respectively) whereas microbiologists showed the highest agreement (69%).
Most respondents agreed that a BDG or GM test would take longer than 3 days in their institution (Statement 10, 68%; Statement 11, 61%). Analysis by country indicates that the UK, Italy, and France experience delays greater than 3 days for turnaround of BDG test and serum GM (BDG: 81%, 75% and 80% respectively. GM: 79%, 72% and 72% respectively).
To improve the timing and accuracy of IFI diagnosis (Statements 12, 13, and 14, all 96%), respondents strongly agreed that access to (i) serum BDG test results, (ii) serum or BAL GM test results, and (iii) PJP polymerase chain reaction results should be available in all ICU facilities within around 48 h. Furthermore, there was also strong agreement that fungal culture, species identification, and susceptibility testing should all be available in-house at every hospital. 77% of respondents agreed that a serum BDG antigen test should be taken before initiation of any antifungal treatment for patients with suspected Candida infection (Statement 20).
Strong agreement was observed that the choice of treatment for an IFI in the ICU should be based on the fungal pathogen, susceptibility testing, pharmacokinetics, co-morbidities, and the site of infection (Statements 15, 25 and 35, 90%,94% and 96%, respectively).
Discussion
In this study, an expert panel developed 44 statements across 6 key domains concerning the diagnosis and management of IFI in the ICU. These statements were then evaluated by an online survey with 335 respondents to identify unmet needs and key recommendations to improve IFI care.
Regarding identification of patients at risk of IFIs, data show a wide variation in incidence across geographies [28], therefore local incidence rates should be available for Physician use in determining individual risk. Individual patient characteristics must also be considered, as risk groups differ between IA (patients with COPD, patients who are immunocompromised, patients with viral pneumonia, liver cirrhosis, or autoimmune diseases) and invasive Candida (IC) infections (any patient with disruption of the skin and gastro-intestinal barrier, and those receiving treatments that change the composition of the gut microbiome). [16, 28,29,30]
Early reliable diagnosis of IFI is of great importance, particularly for IA due to the associated very high mortality rates following angio-invasion despite appropriate antifungal treatment [31]. The ideal sample for diagnosing IA in the ICU is a bronchoalveolar lavage (BAL) for galactomannan (GM) and culture. Additionally, treatment delay in IC infections is a major factor predicting mortality [32, 33], as echoed in the 90% agreement of respondents in this study.
A major obstacle to early diagnosis is the long turnaround time to receive results from mycological tests, and a turnaround time of longer than 3 days for BDG and GM tests was reported by the majority of respondents in this study, suggesting a clear and measurable potential metric for improvement. For the UK, turnaround time for has previously been reported for BDG (> 48 h in 87% of cases) and GM (> 48 h in 86% of cases) [34]. These data were driven by the fact that few laboratories had access to local testing for BDG (3/63, 5%) or GM (13/63, 21%), and most send samples out to reference laboratories [35]. The turnaround time for these tests may be even longer in the more resource-limited countries in Europe, as shown recently by the European Confederation of Medical Mycology (ECMM) laboratory capacities survey. [1]
These results suggest that where possible, all fungal testing should be conducted in-house to ensure a rapid turnaround time (< 48 h) and provide timely patient care in this setting where every hour counts. This recommendation could become a target for programmes such as the ECMM, International Society of Human and Animal Mycology (ISHAM), and the American Society for Microbiology (ASM) One World–One Guideline initiative [26, 36] and other initiatives. Another obstacle to early diagnosis is the lack of reliable diagnostic criteria for IA in patients who are non-neutropenic, as the established criteria for the neutropenic setting often cannot be applied to those patients who are non-neutropenic (Statement 9, 86%). Currently, efforts are underway to create improved classification criteria for the ICU setting. [37]
Early initiation of appropriate antifungal therapy is essential for a successful outcome of IFI [38], and this was recognised in the current study with strong agreement that empirical treatment should start immediately and prior to receiving the results from any diagnostic test if there is high clinical suspicion and, especially, high clinical severity.
Regarding the use of serum BDG testing prior to initiation of antifungal treatments, it is interesting to note that lower agreement levels to this statement were observed from respondents in Germany, Spain, and France (70%, 73%, and 69% respectively). These were considerably lower than responses from the UK (83%) and Italy (90%). This may be due to varying availability, turnaround time, and reimbursement of serum BDG testing. For example, guidance within the UK National Health Service recommends the use of serum BDG testing, but does not mandate it [39].
Evidence suggest that serum BDG has a use in guiding the cessation of empirical antifungal therapy via a negative result [40] because of the high negative predictive value (NPV) the test presents (of around 90–95%) [22]. Given that serum BDG tests become negative under appropriate antifungal therapy (and are therefore also used for treatment monitoring) [41] it is therefore important to obtain a sample for serum BDG testing before initiation of therapy, otherwise the NPV would decrease [42]. Combination of BDG with procalcitonin testing (negative PCT in the presence of candidemia) has shown promise, but needs further exploration, also due to the limited specific diagnostic performance of PCT in this setting[43, 44]. Receiving serum BDG results within 48 h, would allow for early discontinuation of antifungals, and prevent prolongation of unneeded antifungal treatment. In contrast, serum BDG may be less reliable as a marker driving the initiation of antifungal therapy in suspected Candida infection with positive test results. Data from a recent randomised controlled trial examining the use of antifungal therapy in ICU patients with sepsis at a high risk of Candida infection determined that, in this select group of at-risk patients, serum BDG-guided initiation of antifungal therapy did not improve 28-day mortality and may be associated with overtreatment (i.e., treating patients with antifungals without IC infection) [45].
Therefore, with the support shown by the respondents to the statements (particularly Statement 31, 80%), serum BDG should be taken at treatment initiation and the results used thereafter to help guide the discontinuation of empirical antifungal therapy for IC when there is no further evidence of current infection. Despite being supported by a strong recommendation (low quality evidence) by the European Society of Intensive Care Medicine-European Society of Clinical Microbiology and Infectious Diseases Candida infection task-force [46], there was a lack of consensus observed for Statement 32, “In treatment of suspected (and not proven) Candida infection without a clear clinical focus, if no improvement in clinical situation is seen within 4–5 days, then treatment can be stopped” (69%). This suggests a view amongst respondents that clinical findings alone are not sufficient for the discontinuation of treatment and further outlines the need for more BDG testing to support decisions on IC treatment discontinuation. The use of biomarkers can support the decision to stop antifungal treatment.
Results suggest that the choice of initial treatment the choice of treatment for an IFI in the ICU should be based on the fungal pathogen, susceptibility testing, pharmacokinetics, co-morbidities, and the site of infection. This highlights the importance of knowing the local epidemiology of Candida species and susceptibility to increase the likelihood of appropriate empiric antifungal treatment, while waiting for the microbiological results of susceptibility testing. Drug-drug interactions should also be considered (Statement 34, 82%) as low drug levels are one of the potential effects of drug interactions, hospitals should therefore consider their ability to perform therapeutic drug monitoring (TDM) when selecting an antifungal treatment. This seems particularly relevant for voriconazole treatment where TDM is strongly recommended in guidelines [47,48,49], but not always available in institutions across Europe [1]. Furthermore, a recent ECMM survey of IFI diagnostic capacity in Europe showed that availability of TDM was closely related to the gross domestic product of the country in which the institution was located [1].
In cases where culture/specific diagnostics are not possible, a broad-spectrum antifungal treatment should be selected. In cases of suspected mould infections, these broad-spectrum agents would include liposomal amphotericin B, isavuconazole, and posaconazole with activity not only against Aspergillus (for which voriconazole would be a viable option [8]) but a wide range of non-Aspergillus moulds [26]. For Candida infections, echinocandins would be the initial treatment of choice. [50] It was recognised that azole resistance in Aspergillus [51] and other fungal pathogens [18] is an emerging problem that requires future actions. HCPs agreed that in case of local azole resistance rates of > 10%, alternative antifungal treatment should be considered empirically.
In terms of special populations in the ICU, patients on extracorporeal membrane oxygenation (ECMO) were identified by respondents as a population with great unmet needs and who tend to achieve generally lower serum levels of antifungals [52].Therefore, either higher dosages or TDM with rapid dosage adjustments may be required. However, it was also acknowledged that data on antifungal treatment of patients on ECMO are limited, and further studies are needed. Renal replacement therapy in individuals with renal impairment should also be considered when selecting antifungal treatment (Statement 36, 96%).
Respondents agreed that both fungicidal mode of action in abdominal infections, and drug-drug interactions are important factors to consider when selecting a treatment. This may have contributed to the 42% agreement that voriconazole is not a suitable treatment option in ICU patients (Statement 21). However, if voriconazole is used for treatment, TDM is essential with results available within 48 h after achieving steady state (Statement 22). If TDM of voriconazole shows a failure to achieve therapeutic levels after reaching steady state, either a dose escalation or a change to another antifungal is the most appropriate strategy (Statement 24).
Our study also outlines important areas for education and evidence building. There are clear differences of opinion over the role of voriconazole treatment and antifungal prophylaxis between respondent specialities and country (Fig. S2, Statements 21 and 41). This suggests that there is an opportunity to raise awareness of what constitutes optimal treatment modalities amongst all roles. The general lack of confidence reported amongst HCPs regarding the use of antifungal agents and interpreting diagnostic results, supporting a need for greater education of these often rare and orphan diseases. Proactive formal training, with a rigorous education package around IFI, could help improve clinical confidence. This training should include available treatment methods to combat infections and provide the necessary support to implement antifungal use.
Identified Areas of IFI Management in Need of Improvement
Based on the results obtained over the course of this study and the discussion held by the expert panel to review the findings, the authors have identified the following areas requiring attention and the below measures as a way forward as to how to address these remaining IFI challenges in the ICU (Table 2).
This study has a number of strengths. The main strength of this study compared to expert statement publications is the validation of the consensus statements by a multidisciplinary cohort of healthcare professionals involved in the ICU management of IFIs in five different European countries. There was good representation across all countries included within the study with over 300 respondents, lending greater weight to what ICU healthcare professionals actually view as current status of care, optimal care and subsequent challenges faced in their clinical practice. The large study population provides further weight to the findings, especially given the distribution of results across the ICU roles tested.
The study had some limitations. Firstly, the wording of some statements may have been ambiguous and therefore have impacted the agreement displayed. Statements were developed by the expert panel and then ratified by each member individually before being used to develop the survey to reduce any potential bias that may have emerged. Delphi methodology is intended to test opinions. As such, the statements were drafted by authors with clinical and scientific expertise in the field and written as assertions. However, they could be perceived by some as encouraging agreement, although a full scale of opinion was invited from all responders.
Secondly, responses were sought using an incentivised methodology through a standard market research approach, which may have introduced participant bias. Respondents may have felt pressured to align their answers with the perceived objectives of the research group.
Thirdly, respondents’ expertise in IFI management was likely heterogeneous. However, all respondents were screened to confirm involvement in IFI diagnosis and management and had to have a predefined number of years of experience in their roles, so responses were valid in reflecting the current situation in centres across Europe.
Considering these factors, all results were analysed by the project facilitators for potential problems. The full data set was also presented to the expert panel for ratification and to ensure that no bias was present or inadvertently introduced.
Finally, although externally validated, the proposed statements and measures have been developed through modified Delphi consensus approach and as such are expert opinions of the panel and respondents and as such are not evidence-based recommendations.
Conclusions
Based on the results obtained over the course of this study and the discussion the expert panel held to review the findings, the authors identified a number of areas where measures could be taken to address the current state of IFI management. These measures could serve as a potential path on how to improve IFI diagnosis and management, thereby helping to address the remaining IFI challenges in the ICU. These consensus statements and measures may also guide areas for further research to optimise the management of IFI in the ICU.
Availability of Data and Materials
The datasets used and/or analysed during the current study are available from the corresponding author on reasonable request.
Abbreviations
- ASM:
-
American society for microbiology
- BAL:
-
Bronchoalveolar lavage
- BDG:
-
Beta-D-Glucan
- CAPA:
-
COVID-19 Associated pulmonary aspergillosis
- CAM:
-
COVID-19 Associated pulmonary mucormycosis
- COPD:
-
Chronic obstructive pulmonary disease
- ECMM:
-
European confederation of medical mycology
- ECMO:
-
Extracorporeal membrane oxygenation
- GM:
-
Galactomannan
- HCPs:
-
Healthcare professionals
- IA:
-
Invasive aspergillosis
- IC:
-
Invasive candida
- ICU:
-
Intensive care unit(s)
- IFI:
-
Invasive fungal infection(s)
- ISHAM:
-
International society of human and animal mycology
- NPV:
-
Negative predictive value
- PJP:
-
Pneumocystis jirovecii Pneumonia
- TDM:
-
Therapeutic drug monitoring
References
Salmanton-García J, Hoenigl M, Gangneux JP, Segal E, Alastruey-Izquierdo A, Arikan Akdagli S, et al. The current state of laboratory mycology and access to antifungal treatment in europe: a european confederation of medical mycology survey. Lancet Microbe. 2023;4(1):e47-56. https://doi.org/10.1016/S2666-5247(22)00261-0. (Epub 2022 Dec 1 PMID: 36463916).
Poissy J, Rouzé A, Cornu M, Nseir S, Sendid B. The changing landscape of invasive fungal infections in ICUs: a need for risk stratification to better target antifungal drugs and the threat of resistance. J Fungi (Basel). 2022;8(9):946. https://doi.org/10.3390/jof8090946.
Koehler P, Stecher M, Cornely OA, Koehler D, Vehreschild MJGT, Bohlius J, et al. Morbidity and mortality of candidaemia in Europe: an epidemiologic meta-analysis. Clin Microbiol Infect. 2019;25(10):1200–12. https://doi.org/10.1016/j.cmi.2019.04.024. (Epub 2019 Apr 27 PMID: 31039444).
Cuenca-Estrella M, Kett DH, Wauters J. Defining standards of CARE for invasive fungal diseases in the ICU. J Antimicrob Chemother. 2019;74:9–15. https://doi.org/10.1093/jac/dkz038.
Ruiz Camps I. Aspergillosis: beyond the oncohematological patient. Enferm Infecc Microbiol Clin Engl Ed. 2020;38(1):1–3.
Lass-Flörl C. The changing face of epidemiology of invasive fungal disease in Europe. Mycoses. 2009;52(3):197–205. https://doi.org/10.1111/j.1439-0507.2009.01691.x. (PMID: 19391253).
Tabah A, Buetti N, Staiquly Q, Ruckly S, Akova M, Aslan AT, et al. Epidemiology and outcomes of hospital-acquired bloodstream infections in intensive care unit patients: the EUROBACT-2 international cohort study. Intensive Care Med. 2023;49(2):178–90.
Koehler P, Bassetti M, Chakrabarti A, Chen SCA, Colombo AL, Hoenigl M, et al. Defining and managing COVID-19-associated pulmonary aspergillosis: the 2020 ECMM/ISHAM consensus criteria for research and clinical guidance. Lancet Infect Dis. 2021;21(6):149–62. https://doi.org/10.1016/S1473-3099(20)30847-1.
Schauwvlieghe AFAD, Rijnders BJA, Philips N, Verwijs R, Vanderbeke L, Van Tienen C, et al. Invasive aspergillosis in patients admitted to the intensive care unit with severe influenza: a retrospective cohort study. Lancet Respir Med. 2018;6(10):782–92. https://doi.org/10.1016/S2213-2600(18)30274-1. (Epub 2018 Jul 31 PMID: 30076119).
Guinea J, Torres-Narbona M, Gijón P, Muñoz P, Pozo F, Peláez T, et al. Pulmonary aspergillosis in patients with chronic obstructive pulmonary disease: incidence, risk factors, and outcome. Clin Microbiol Infect. 2010;16(7):870–7. https://doi.org/10.1111/j.1469-0691.2009.03015.x. (Epub 2009 Nov 10 PMID: 19906275).
Hoenigl M, Seidel D, Sprute R, Cunha C, Oliverio M, Goldman GH, et al. COVID-19-associated fungal infections. Nat Microbiol. 2022;7(8):1127–40. https://doi.org/10.1038/s41564-022-01172-2.
Raffaelli F, Tanzarella ES, De Pascale G, Tumbarello M. Invasive respiratory fungal infections in COVID-19 critically ill patients. J Fungi (Basel). 2022;8(4):415. https://doi.org/10.3390/jof8040415.PMID:35448646;PMCID:PMC9025868.
Casalini G, Giacomelli A, Ridolfo A, Gervasoni C, Antinori S. Invasive fungal infections complicating COVID-19: a narrative review. J Fungi (Basel). 2021;7(11):921. https://doi.org/10.3390/jof7110921.PMID:34829210;PMCID:PMC8620819.
Egger M, Prüller F, Krause R, Prattes J, Hoenigl M. (2022) Utility of serum 1,3-β-d-glucan testing for diagnosis and prognostication in COVID-19-associated pulmonary aspergillosis. Microbiol Spectr. 2022;10(3):e0137322. https://doi.org/10.1128/spectrum.01373-22.
Tejerina EE, Abril E, Padilla R, Rodríguez Ruíz C, Ballen A, Frutos-Vivar F, et al. Invasive aspergillosis in critically ill patients: an autopsy study. Mycoses. 2019;62(8):673–9. https://doi.org/10.1111/myc.12927. (Epub 2019 Jun 9 PMID: 31177621).
Enoch DA, Yang H, Aliyu SH, Micallef C. The changing epidemiology of invasive fungal infections. Methods Mol Biol. 2017;1508:17–65. https://doi.org/10.1007/978-1-4939-6515-1_2. (PMID: 27837497).
Paramythiotou E, Frantzeskaki F, Flevari A, Armaganidis A, Dimopoulos G. Invasive fungal infections in the ICU: how to approach, how to treat. Molecules. 2014;19(1):1085–119. https://doi.org/10.3390/molecules19011085.
Pappas PG, Lionakis MS, Arendrup MC, Ostrosky-Zeichner L, Kullberg BJ. Invasive candidiasis Nat Rev Dis Primers. 2018;4:18026. https://doi.org/10.1038/nrdp.2018.26. (PMID: 29749387).
Hoenigl M, Salmanton-García J, Egger M, Gangneux JP, Bicanic T, Arikan-Akdagli S, et al. Guideline adherence and survival of patients with candidaemia in Europe: results from the ECMM Candida III multinational European observational cohort study. Lancet Infect Dis. 2023. https://doi.org/10.1016/S1473-3099(22)00872-6.
Egger M, Salmanton-García J, Barac A, Gangneux JP, Guegan H, Arsic-Arsenijevic V, et al. Predictors for prolonged hospital stay solely to complete intravenous antifungal treatment in patients with candidemia: results from the ECMM Candida III multinational european observational cohort study. Mycopathologia. 2023;188(6):983–94.
Seidel D, Wurster S, Jenks JD, Sati H, Gangneux JP, Egger M, Alastruey-Izquierdo A, Ford NP, Chowdhary A, Sprute R, Cornely O, Thompson GR 3rd, Hoenigl M, Kontoyiannis DP. Impact of climate change and natural disasters on fungal infections. Lancet Microbe. 2024. https://doi.org/10.1016/s2666-5247(24)00039-9.
Bassetti M, Azoulay E, Kullberg BJ, Ruhnke M, Shoham S, Vazquez J, et al. EORTC/MSGERC definitions of invasive fungal diseases: summary of activities of the intensive care unit working group. Clin Infect Dis. 2021;72(Suppl 2):S121–7. https://doi.org/10.1093/cid/ciaa1751. (PMID: 33709127).
Karageorgopoulos DE, Vouloumanou EK, Ntziora F, Michalopoulos A, Rafailidis PI, Falagas ME. β-D-glucan assay for the diagnosis of invasive fungal infections: a meta-analysis. Clin Infect Dis. 2011;52(6):750–70. https://doi.org/10.1093/cid/ciq206. (PMID: 21367728).
Aerts R, Autier B, Gornicec M, Prattes J, Lagrou K, Gangneux JP, et al. Point-of-care testing for viral-associated pulmonary aspergillosis. Expert Rev Mol Diagn. 2023. https://doi.org/10.1080/14737159.2023.2257597.
Mercier T, Castagnola E, Marr KA, Wheat LJ, Verweij PE, Maertens JA. Defining galactomannan positivity in the updated EORTC/MSGERC consensus definitions of invasive fungal diseases. Clin Infect Dis. 2021;72(Suppl 2):S89–94. https://doi.org/10.1093/cid/ciaa1786. (PMID: 33709125).
Hoenigl M, Salmanton-García J, Walsh TJ, Nucci M, Neoh CF, Jenks JD, et al. Global guideline for the diagnosis and management of rare mould infections: an initiative of the european confederation of medical mycology in cooperation with the international society for human and animal mycology and the american society for microbiology. Lancet Infect Dis. 2021;21(8):246–57. https://doi.org/10.1016/S1473-3099(20)30784-2.
Eysenbach G. Improving the quality of web surveys: the checklist for reporting results of internet E-surveys (CHERRIES). J Med Internet Res. 2004. https://doi.org/10.2196/jmir.6.3.e34.
Bassetti M, Giacobbe DR, Vena A, Trucchi C, Ansaldi F, Antonelli M, et al. Incidence and outcome of invasive candidiasis in intensive care units (ICUs) in Europe: results of the EUCANDICU project. Crit Care. 2019;23(1):219. https://doi.org/10.1186/s13054-019-2497-3.
Prattes J, Wauters J, Giacobbe DR, Salmanton-García J, Maertens J, Bourgeois M, et al. Risk factors and outcome of pulmonary aspergillosis in critically ill coronavirus disease 2019 patients-a multinational observational study by the European confederation of medical mycology. Clin Microbiol Infect. 2022;28(4):580–7. https://doi.org/10.1016/j.cmi.2021.08.014.
Bassetti M, Vena A, Giacobbe DR, Trucchi C, Ansaldi F, Antonelli M, et al. Risk factors for intra-abdominal candidiasis in intensive care units: results from EUCANDICU study. Infect Dis Ther. 2022;11(2):827–40. https://doi.org/10.1007/s40121-021-00585-6.
Ergün M, Brüggemann RJM, Alanio A, Dellière S, van Arkel A, Bentvelsen RG, et al. aspergillus test profiles and mortality in critically ill COVID-19 patients. J Clin Microbiol. 2021;59(12):e0122921. https://doi.org/10.1128/JCM.01229-21.
Morrell M, Fraser VJ, Kollef MH. Delaying the empiric treatment of candida bloodstream infection until positive blood culture results are obtained: a potential risk factor for hospital mortality. Antimicrob Agents Chemother. 2005;49(9):3640–5. https://doi.org/10.1128/AAC.49.9.3640-3645.2005.
Garey KW, Rege M, Pai MP, Mingo DE, Suda KJ, Turpin RS, et al. Time to initiation of fluconazole therapy impacts mortality in patients with candidemia: a multi-institutional study. Clin Infect Dis. 2006;43(1):25–31. https://doi.org/10.1086/504810. (Epub 2006 May 16 PMID: 16758414).
Micallef C, Ashiru-Oredope D, Hansraj S, Denning DW, Agrawal SG, Manuel RJ, et al. An investigation of antifungal stewardship programmes in England. J Med Microbiol. 2017;66(11):1581–9. https://doi.org/10.1099/jmm.0.000612. (Epub 2017 Oct 25 PMID: 29068278).
Schelenz S, Owens K, Guy R, Rautemaa-Richardson R, Manuel RJ, Richardson M, et al. National mycology laboratory diagnostic capacity for invasive fungal diseases in 2017: Evidence of sub-optimal practice. J Infect. 2019;79(2):167–73. https://doi.org/10.1016/j.jinf.2019.06.009. (Epub 2019 Jun 21 PMID: 31233810).
ECMM. European confederation of medical mycology one world–one guideline. Available from: https://www.ecmm.info/guidelines/ Accessed 20 February 2023
Bassetti M, Scudeller L, Giacobbe DR, Lamoth F, Righi E, Zuccaro V, et al. Developing definitions for invasive fungal diseases in critically ill adult patients in intensive care units protocol of the FUNgal infections definitions in ICU patients (FUNDICU) project. Mycoses. 2019;62(4):310–9.
Chamilos G, Lewis RE, Kontoyiannis DP. Delaying amphotericin B-based frontline therapy significantly increases mortality among patients with hematologic malignancy who have zygomycosis. Clin Infect Dis. 2008;47(4):503–9. https://doi.org/10.1086/590004. (PMID: 18611163).
Lewis T. and Dymond S. Diagnosis and management of fungal infections in critical care guideline. 2020. https://www.northdevonhealth.nhs.uk/wp-content/uploads/2020/08/Antifungal-Guidelines-for-Critical-Care-Patients-v1.0-28012020.pdf Accessed 14 February 2023.
Sandhar TK, Chhina DK, Gupta V, Chaudhary J. Role of (1–3)-Β-D-glucan test in the diagnosis of invasive fungal infections among high-risk patients in a tertiary care hospital. J Lab Physicians. 2022;14(3):306–11. https://doi.org/10.1055/s-0042-1742632.
Jaijakul S, Vazquez JA, Swanson RN, Ostrosky-Zeichner L. (1,3)-β-D-glucan as a prognostic marker of treatment response in invasive candidiasis. Clin Infect Dis. 2012;55(4):521–6. https://doi.org/10.1093/cid/cis456. (Epub 2012 May 9 PMID: 22573851).
Rautemaa-Richardson R, Rautemaa V, Al-Wathiqi F, Moore CB, Craig L, Felton TW, Muldoon EG. Impact of a diagnostics-driven antifungal stewardship programme in a UK tertiary referral teaching hospital. J Antimicrob Chemother. 2018;73(12):3488–95. https://doi.org/10.1093/jac/dky360. (PMID: 30252053).
Giacobbe DR, Mikulska M, Tumbarello M, Furfaro E, Spadaro M, Losito AR, et al. Combined use of serum (1,3)-β-D-glucan and procalcitonin for the early differential diagnosis between candidaemia and bacteraemia in intensive care units. Crit Care. 2017;21(1):176. https://doi.org/10.1186/s13054-017-1763-5.
Cortegiani A, Misseri G, Ippolito M, Bassetti M, Giarratano A, Martin-Loeches I, et al. Procalcitonin levels in candidemia versus bacteremia: a systematic review. Crit Care. 2019;23(1):190. https://doi.org/10.1186/s13054-019-2481-y.
Bloos F, Held J, Kluge S, Simon P, Kogelmann K, de Heer G, et al. (1 → 3)-β-D-glucan-guided antifungal therapy in adults with sepsis: the CandiSep randomized clinical trial. Intensive Care Med. 2022;48(7):865–75. https://doi.org/10.1007/s00134-022-06733-x.
Martin-Loeches I, Antonelli M, Cuenca-Estrella M, Dimopoulos G, Einav S, De Waele JJ, et al. ESICM/ESCMID task force on practical management of invasive candidiasis in critically ill patients. Intensive Care Med. 2019;45(6):789–805. https://doi.org/10.1007/s00134-019-05599-w. (Epub 2019 Mar 25 PMID: 30911804).
Ullmann AJ, Aguado JM, Arikan-Akdagli S, Denning DW, Groll AH, Lagrou K, et al. Diagnosis and management of Aspergillus diseases: executive summary of the 2017 ESCMID-ECMM-ERS guideline. Clin Microbiol Infect. 2018;24(Suppl 1):e1-38. https://doi.org/10.1016/j.cmi.2018.01.002. (Epub 2018 Mar 12 PMID: 29544767).
Tissot F, Agrawal S, Pagano L, Petrikkos G, Groll AH, Skiada A, et al. ECIL-6 guidelines for the treatment of invasive candidiasis, aspergillosis and mucormycosis in leukemia and hematopoietic stem cell transplant patients. Haematologica. 2017;102(3):433–44. https://doi.org/10.3324/haematol.2016.152900.
Jenks JD, Mehta SR, Hoenigl M. Broad spectrum triazoles for invasive mould infections in adults: which drug and when? Med Mycol. 2019;57(2):S168-S178S.
Cornely OA, Bassetti M, Calandra T, Garbino J, Kullberg BJ, Lortholary O, et al. ESCMID* guideline for the diagnosis and management of Candida diseases 2012: non-neutropenic adult patients. Clin Microbiol Infect. 2012;18(Suppl 7):19–37. https://doi.org/10.1111/1469-0691.12039. (PMID: 23137135).
Verweij PE, Chowdhary A, Melchers WJ, Meis JF. Azole resistance in aspergillus fumigatus: can we retain the clinical use of mold-active antifungal azoles? Clin Infect Dis. 2016;62(3):362–8.
Kriegl L, Hatzl S, Zurl C, Reisinger AC, Schilcher G, Eller P, Gringschl Y, et al. Isavuconazole plasma concentrations in critically ill patients during extracorporeal membrane oxygenation. J Antimicrob Chemother. 2022;77(9):2500–5. https://doi.org/10.1093/jac/dkac196.
Acknowledgements
Medical writing, analysis of data, and editorial support in the preparation of this manuscript was provided by Tim Warren and Thomas Scoble of Triducive Partners Limited and funded by Gilead Sciences Europe Ltd.
Funding
Open access funding provided by Medical University of Graz. Gilead Sciences Europe Ltd (Gilead) initiated and funded the consensus research, including the selection and contracting of the faculty of experts who were tasked to write the consensus statement survey. Gilead could provide editorial assistance for the manuscript but had no control over the final content. None of the authors were paid by Gilead for writing the manuscript.
Author information
Authors and Affiliations
Contributions
The author group, led by Professor Martin Höenigl, has jointly prepared the manuscript. All authors participated in discussions during the expert panel meetings, contributed to and reviewed consensus survey statements, contributed to subsequent drafts of the manuscript, including literature searches and approved the final manuscript
Corresponding author
Ethics declarations
Conflict of interest
MH received research funding and/or speaker and consultancy fees from Gilead, Astellas, MSD, Euroimmune, IMMY, Pulmocide, Mundipharma, Scynexis, F2G, and Pfizer. DAE received fees for lectures and consultancy from Tillots and Gilead. DW reports consultant and lecture fees from 3 M, Advants, AMEOS, Eumedica, EUSA, Gilead, InfectoPharm, Kite, Lilly, MSD, Novartis, Pfizer, and Shionogi. DWy has received speaker and consultancy fees from Gilead and Shionogi. AC received fees for lectures or consultancy by Gilead, MSD, Mundipharma, and Pfizer.
Ethical Approval
A statement of consent was included at the start of the survey and consent was implied by completion and submission of the survey. Consent was therefore sought from the participating HCPs.
Consent for Publication
Participants were informed that the aim was to publish aggregated data from the survey in a peer-reviewed journal. A statement of consent was included at the start of the survey and consent was implied by completion and submission of the survey.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Handling Editor: Jean-Pierre Gangneux
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hoenigl, M., Enoch, D.A., Wichmann, D. et al. Exploring European Consensus About the Remaining Treatment Challenges and Subsequent Opportunities to Improve the Management of Invasive Fungal Infection (IFI) in the Intensive Care Unit. Mycopathologia 189, 41 (2024). https://doi.org/10.1007/s11046-024-00852-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11046-024-00852-3