Abstract
Background
High-fat diets (HFD) have recently become a public health concern. We hypothesize that HFD induces exosomes biogenesis in the lung tissue of rat model.
Methods and results
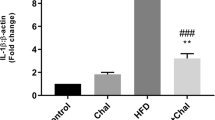
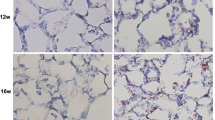
Sixteen adult male Wistar rats were fed with HFD or a regular chow diet for 3 months. The histopathological changes in lung tissues were measured by hematoxylin and eosin (H&E) staining. Bronchoalveolar lavage (BAL) was performed to assay exosomes by acetylcholinesterase enzyme (AhCE) activity. Real-time PCR (qPCR) was used to evaluate Rab27-b, Alix, and IL-1β expression, while the immunohistochemical examination was performed for CD81 expression in lung tissues. In addition, expression of IL-1β was detected by ELISA. We found pathological alterations in the lung tissue of HFD animals. AhCE activity along with the expression level of Rab27-b, Alix, and IL-1β was increased in HFD animals (p < 0.05). Immunohistochemical staining showed that expression of CD81 was increased in lung tissues of HFD animals compared with the control group (p < 0.05).
Conclusion
Hence, HFD induced exosomes biogenesis and histopathological changes with IL-1β expression in rats’ lung tissues.





Similar content being viewed by others
Data availability
The data that support the findings of this study are available on request from the corresponding author.
References
Canfora EE, Meex RC, Venema K, Blaak EE (2019) Gut microbial metabolites in obesity, NAFLD and T2DM. Nat Rev Endocrinol 15(5):261–273
Buckley J (2018) Availability of high-fat foods might drive the obesity epidemic. Nat Rev Endocrinol 14(10):574–575
Pacana T, Cazanave S, Verdianelli A, Patel V, Min H-K, Mirshahi F, Quinlivan E, Sanyal AJ (2015) Dysregulated hepatic methionine metabolism drives homocysteine elevation in diet-induced nonalcoholic fatty liver disease. PLoS ONE 10(8):e0136822
Kim Y-C, Seok S, Byun S, Kong B, Zhang Y, Guo G, Xie W, Ma J, Kemper B, Kemper JK (2018) AhR and SHP regulate phosphatidylcholine and S-adenosylmethionine levels in the one-carbon cycle. Nat Commun 9(1):540
Lee S, Norheim F, Gulseth HL, Langleite TM, Aker A, Gundersen TE, Holen T, Birkeland KI, Drevon CA (2018) Skeletal muscle phosphatidylcholine and phosphatidylethanolamine respond to exercise and influence insulin sensitivity in men. Sci Rep 8(1):6531
Blüher M (2019) Obesity: global epidemiology and pathogenesis. Nat Rev Endocrinol 15(5):288–298
Peters U, Suratt BT, Bates JH, Dixon AE (2018) Beyond BMI: obesity and lung disease. Chest 153(3):702–709
Dixon AE, Peters U (2018) The effect of obesity on lung function. Expert Rev Respir Med 12(9):755–767
Babaei M, Hasanzadeh S, Pirnejad H, Mohebbi I, Hoseini R, Niazkhani Z (2022) Socioeconomic status and severity of traffic accident injuries: a cross-sectional study. Iran Occup Health 19(1):380–392
Manna P, Jain SK (2015) Obesity, oxidative stress, adipose tissue dysfunction, and the associated health risks: causes and therapeutic strategies. Metab Syndr Relat Disord 13(10):423–444
Kim Y, Son T, Park J, Jun W, Kim OK (2022) Role of exosomes derived from adipose tissue under obese conditions in skeletal muscle and liver cells: commonalities and differences. Mol Nutr Food Res 66(23):2200358
Kwan HY, Chen M, Xu K, Chen B (2021) The impact of obesity on adipocyte-derived extracellular vesicles. Cell Mol Life Sci. https://doi.org/10.1007/s00018-021-03973-w
Maligianni I, Yapijakis C, Bacopoulou F, Chrousos G (2021) The potential role of exosomes in child and adolescent obesity. Children 8(3):196
Rezaie J, Feghhi M, Etemadi T (2022) A review on exosomes application in clinical trials: perspective, questions, and challenges. Cell Commun Signal 20(1):1–13
Feghhi M, Rezaie J, Akbari A, Jabbari N, Jafari H, Seidi F, Szafert S (2021) Effect of multi-functional polyhydroxylated polyhedral oligomeric silsesquioxane (POSS) nanoparticles on the angiogenesis and exosome biogenesis in human umbilical vein endothelial cells (HUVECs). Mater Des 197:109227
Tian Y, Cheng C, Wei Y, Yang F, Li G (2022) The role of exosomes in inflammatory diseases and tumor-related inflammation. Cells 11(6):1005
Hough K, Chanda D, Duncan S, Thannickal V, Deshane J (2017) Exosomes in immunoregulation of chronic lung diseases. Allergy 72(4):534–544
Hariri N, Thibault L (2010) High-fat diet-induced obesity in animal models. Nutr Res Rev 23(2):270–299
Alipoor SD, Mortaz E, Garssen J, Movassaghi M, Mirsaeidi M, Adcock IM (2016) Exosomes and exosomal miRNA in respiratory diseases. Mediators Inflamm https://doi.org/10.1155/2016/5628404
Almohammai A, Rahbarghazi R, Keyhanmanesh R, Rezaie J, Ahmadi M (2021) Asthmatic condition induced the activity of exosome secretory pathway in rat pulmonary tissues. J Inflamm. 18:1–9
de Souza ABF, Chírico MTT, Cartelle CT, de Paula Costa G, Talvani A, Cangussú SD, de Menezes RCA, Bezerra FS (2018) High-fat diet increases HMGB1 expression and promotes lung inflammation in mice subjected to mechanical ventilation. Oxid Med Cell Longev https://doi.org/10.1155/2018/7457054
Mafort TT, Rufino R, Costa CH, Lopes AJ (2016) Obesity: systemic and pulmonary complications, biochemical abnormalities and impairment of lung function. Multidisciplinary Respir Med 11(1) https://doi.org/10.1186/s40248-016-0066-z
Hildebrandt X, Ibrahim M, Peltzer N (2023) Cell death and inflammation during obesity: know my methods, WAT (son). Cell Death Differ 30(2):279–292
Wang F, Zuo Z, Chen K, Fang J, Cui H, Shu G, Zhou Y, Chen Z, Huang C, Liu W (2018) Histopathological changes caused by inflammation and oxidative stress in diet-induced-obese mouse following experimental lung injury. Sci Rep 8(1):14250
Jabbari N, Feghhi M, Esnaashari O, Soraya H, Rezaie J (2022) Inhibitory effects of gallic acid on the activity of exosomal secretory pathway in breast cancer cell lines: a possible anticancer impact. BioImpacts 12(6):549–559
Larios J, Mercier V, Roux A, Gruenberg J (2020) ALIX-and ESCRT-III-dependent sorting of tetraspanins to exosomes. J Cell Biol https://doi.org/10.1083/jcb.201904113
Ostrowski M, Carmo NB, Krumeich S, Fanget I, Raposo G, Savina A, Moita CF, Schauer K, Hume AN, Freitas RP (2010) Rab27a and Rab27b control different steps of the exosome secretion pathway. Nat Cell Biol 12(1):19–30
Hassanpour M, Rezaie J, Darabi M, Hiradfar A, Rahbarghazi R, Nouri M (2020) Autophagy modulation altered differentiation capacity of CD146+ cells toward endothelial cells, pericytes, and cardiomyocytes. Stem Cell Res Ther 11(1):1–14
Roucourt B, Meeussen S, Bao J, Zimmermann P, David G (2015) Heparanase activates the syndecan-syntenin-ALIX exosome pathway. Cell Res 25(4):412–428
Eguchi A, Lazic M, Armando AM, Phillips SA, Katebian R, Maraka S, Quehenberger O, Sears DD, Feldstein AE (2016) Circulating adipocyte-derived extracellular vesicles are novel markers of metabolic stress. J Mol Med 94:1241–1253
Hubal MJ, Nadler EP, Ferrante SC, Barberio MD, Suh JH, Wang J, Dohm GL, Pories WJ, Mietus-Snyder M, Freishtat RJ (2017) Circulating adipocyte‐derived exosomal MicroRNAs associated with decreased insulin resistance after gastric bypass. Obesity 25(1):102–110
Chaudhuri AD, Dastgheyb RM, Yoo S-W, Trout A, Talbot CC Jr, Hao H, Witwer KW, Haughey NJ (2018) TNFα and IL-1β modify the miRNA cargo of astrocyte shed extracellular vesicles to regulate neurotrophic signaling in neurons. Cell Death Dis 9(3):363
Villarroya-Beltri C, Gutiérrez-Vázquez C, Sánchez-Cabo F, Pérez-Hernández D, Vázquez J, Martin-Cofreces N, Martinez-Herrera DJ, Pascual-Montano A, Mittelbrunn M, Sánchez-Madrid F (2013) Sumoylated hnRNPA2B1 controls the sorting of miRNAs into exosomes through binding to specific motifs. Nat Commun 4(1):2980
Tian Y, Cheng C, Wei Y, Yang F, Li G (2022) The role of exosomes in inflammatory diseases and tumor-related inflammation. Cells. https://doi.org/10.3390/cells11061005
Console L, Scalise M, Indiveri C (2019) Exosomes in inflammation and role as biomarkers. Clin Chim Acta 488:165–171. https://doi.org/10.1016/j.cca.2018.11.009
Acknowledgements
We would like to thank the Clinical Research Development Unit of Zahra Mardani Azari children educational and treatment center, Tabriz university of medical sciences, Tabriz, Iran for their assistance in this research.
Funding
This work was approved and supported by a grant from Tabriz University of Medical Sciences (No: IR.TBZMED.VCR.REC.1399.236).
Author information
Authors and Affiliations
Contributions
M-Sh and JR conceived and designed the experiments and analyzed the data. RR prepared the figures. JR and MA wrote the manuscript. R M-H, MA, and MJ-B performed the experiments. All authors read and approved the final manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors have no competing interests to declare.
Ethical approval
The animal experimental procedures were conducted according to the principles of guidelines for the ethical use of animals in applied studies and approved by the Ethics Committee on Animal Use of Tabriz University of Medical Sciences (IR.TBZMED.VCR.REC.1399.236) in compliance with the ARRIVE guidelines.
Consent for publication
Not applicable.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Shoaran, M., Behmand, M.J., Rahbarghazi, R. et al. High-fat diet-induced biogenesis of pulmonary exosomes in an experimental rat model. Mol Biol Rep 50, 7589–7595 (2023). https://doi.org/10.1007/s11033-023-08703-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-023-08703-y