Abstract
Introduction Rheumatoid arthritis (RA) is a common autoimmune disease across the globe that is chronic and systemic as well. The disease is linked with autoantibodies and is inflammatory, eventually targeting several molecules along with certain modified self-epitopes. The disease majorly affects the joints of an individual. Rheumatoid arthritis is manifested clinically by polyarthritis linked with the dysfunction of the joints. This chiefly affects the synovial joint lining and is linked with progressive dysfunction, premature death, along with socioeconomic implications. The macrophage activation, along with the activation of certain defense cells, results in a response to self-epitopes that helps in providing a better understanding of the disease pathogenesis.
Material and methodology For this review article, papers have been retrieved and reviewed from database including PubMed, Scopus and Web of science. Relevant papers were taken fulfilling the criteria for writing this review article.
Results This has resulted in the establishment of several new therapeutic techniques that serve as potential inhibitors of such cells. Researchers have gained an interest in understanding this disease to provide strategies for treatment in the last two decades. This also includes recognition followed by the treatment of the disease at its early stages. Various allopathic treatment approaches often have chronic and toxic teratogenic effects. However, to avoid this issue of toxicity followed by side effects, certain medicinal plants have been used in treating RA.
Conclusion Medicinal plants possess active phytoconstituents that entail antioxidants as well as anti-inflammatory properties, making them a helpful alternative to allopathic drugs that are often linked with highly toxic effects. This review paper entails a thorough discussion of the epidemiology, pathophysiology, diagnosis, and management of RA. The paper will also focus on the use of herbal plants in the treatment of the disease to avoid the side effects that generally occur in allopathic treatment.
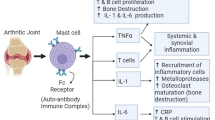
Graphical abstract




Similar content being viewed by others
Abbreviations
- ACPA:
-
Anti-citrullinated protein antibodies
- AKT:
-
Ak strain transforming
- ALT:
-
Alanine transaminase
- APC:
-
Antigen-presenting cell
- BMI:
-
Body mass index
- CCP:
-
Anti-cyclic citrullinated peptide-2
- CD4:
-
Clusters of differentiation 4
- CMV:
-
Cytomegalovirus
- COX:
-
Cyclooxygenase
- CRP:
-
c-reactive protein
- DMARDs:
-
Disease-modifying antirheumatic drugs
- DNA:
-
Deoxyribonucleic acid
- DR:
-
D-related locus
- EBV:
-
Epstein–Barr virus
- ERK:
-
Extracellular signal-regulated kinase
- ESR:
-
Erythrocyte sedimentation rate
- EXOSC1:
-
Exosome component 1
- FDA:
-
Food and Drug Administration
- FLS:
-
Fibroblast-like synoviocytes
- GM-CSF:
-
Granulocyte macrophage colony-stimulating factor
- HIV:
-
Human immunodeficiency virus
- HLA:
-
Human leukocyte antigens
- IFNs:
-
Interferons
- Ig:
-
immunoglobulin
- IL:
-
Interleukin
- iNOS:
-
Inducible nitric oxide synthase
- JNK:
-
c-Jun N-terminal kinase
- LOX:
-
Lysyl oxidase
- LTB4:
-
Leukotriene B4
- MAP Kinase:
-
Mitogen activated protein kinases
- MCP-1:
-
Monocyte chemoattractant protein-1
- MMPs:
-
Matrix metalloproteinases
- mRNA:
-
Messenger ribonucleic acid
- mTOR:
-
Mammalian target of rapamycin
- MTP:
-
Microsomal triglyceride transfer protein
- NF-kB:
-
Nuclear factor-Κb
- NO:
-
Nitric oxide
- NSAID:
-
Non-steroidal anti-inflammatory drug
- PDGF:
-
Platelet-derived growth factor
- PGE2:
-
Prostaglandin E2
- PI3Ks:
-
Phosphoinositide 3-kinases
- PIP:
-
Prolactin-induced protein
- RA:
-
Rheumatoid arthritis
- RANK:
-
Receptor activator of NF-κB
- RANKL:
-
Receptor activator of nuclear factor kappa-B ligand
- RF:
-
Rheumatoid factor
- ROS:
-
Reactive oxygen species
- STAT4:
-
Signal transducer and activator of transcription 4
- Th1:
-
T helper type 1
- Th2:
-
T helper type 2
- TNF:
-
Tumor necrosis factor
- TRAF:
-
Tumor necrosis factor receptor–associated factor
- WBC:
-
White blood cell
References
Guo Q, Wang Y, Xu D et al (2018) Rheumatoid arthritis: pathological mechanisms and modern pharmacologic therapies. Bone Res 6:1–14
Araki Y, Mimura T (2016) The mechanisms underlying chronic inflammation in rheumatoid arthritis from the perspective of the epigenetic landscape. J Immunol Res 2016:1–10. https://doi.org/10.1155/2016/6290682
van Delft MAM, Huizinga TWJ (2020) An overview of autoantibodies in rheumatoid arthritis. J Autoimmun 110:102392. https://doi.org/10.1016/J.JAUT.2019.102392
Yap HY, Tee SZY, Wong MMT et al (2018) Pathogenic role of immune cells in rheumatoid arthritis: implications in clinical treatment and biomarker development. Cells 7:161. https://doi.org/10.3390/CELLS7100161
Edilova MI, Akram A, Abdul-Sater AA (2021) Innate immunity drives pathogenesis of rheumatoid arthritis. Biomed J 44:172–182. https://doi.org/10.1016/J.BJ.2020.06.010
de Molon RS, Rossa C, Thurlings RM et al (2019) Linkage of periodontitis and rheumatoid arthritis: current evidence and potential biological interactions. Int J Mol Sci 20:4541. https://doi.org/10.3390/IJMS20184541
Sofowora A, Ogunbodede E, Onayade A (2013) The role and place of medicinal plants in the strategies for disease prevention. Afr J Tradit Complement Altern Med 10:210–229. https://doi.org/10.4314/AJTCAM.V10I5.2
Singh S, Singh TG, Mahajan K, Dhiman S (2020) Medicinal plants used against various inflammatory biomarkers for the management of rheumatoid arthritis. J Pharm Pharmacol 72:1306–1327. https://doi.org/10.1111/JPHP.13326
Anand U, Jacobo-Herrera N, Altemimi A, Lakhssassi N (2019) A comprehensive review on medicinal plants as antimicrobial therapeutics: potential avenues of biocompatible drug discovery. Metabolites 9:258. https://doi.org/10.3390/METABO9110258
Smolen JS, Aletaha D, Barton A et al (2018) Rheumatoid arthritis. Nat Rev Dis Primers 4:18001. https://doi.org/10.1038/NRDP.2018.1
Pradeepkiran JA (2019) Insights of rheumatoid arthritis risk factors and associations. J Transl Autoimmun 2:100012. https://doi.org/10.1016/J.JTAUTO.2019.100012
Svendsen AJ, Gervin K, Lyle R et al (2016) Differentially methylated DNA regions in monozygotic twin pairs discordant for rheumatoid arthritis: an epigenome-wide study. Front Immunol 7:510. https://doi.org/10.3389/fimmu.2016.00510
van Drongelen V, Holoshitz J (2017) Human leukocyte antigen-disease associations in rheumatoid arthritis. Rheum Dis Clin North Am 43:363–376. https://doi.org/10.1016/J.RDC.2017.04.003
Lim J, Kim K (2019) Genetic variants differentially associated with rheumatoid arthritis and systemic lupus erythematosus reveal the disease-specific biology. Sci Rep 9:1–7. https://doi.org/10.1038/s41598-019-39132-2
Cribbs A, Feldmann M, Oppermann U (2015) Towards an understanding of the role of DNA methylation in rheumatoid arthritis: therapeutic and diagnostic implications. Ther Adv Musculoskelet Dis 7:206–219. https://doi.org/10.1177/1759720X15598307
Ciechomska M, Roszkowski L, Maslinski W (2019) DNA methylation as a future therapeutic and diagnostic target in rheumatoid arthritis. Cells 8:953. https://doi.org/10.3390/CELLS8090953
Sokolove J, Wagner CA, Lahey LJ et al (2016) Increased inflammation and disease activity among current cigarette smokers with rheumatoid arthritis: a cross-sectional analysis of US veterans. Rheumatology 55:1969–1977. https://doi.org/10.1093/RHEUMATOLOGY/KEW285
Koziel J, Mydel P, Potempa J (2014) The link between periodontal disease and rheumatoid arthritis: an updated review. Curr Rheumatol Rep 16:408–408. https://doi.org/10.1007/S11926-014-0408-9
Jeong Y, Kim JW, You HJ et al (2019) Gut microbial composition and function are altered in patients with early rheumatoid arthritis. J Clin Med 8:693. https://doi.org/10.3390/JCM8050693
Novella-Navarro M, Plasencia-Rodríguez C, Nuño L, Balsa A (2021) Risk factors for developing rheumatoid arthritis in patients with undifferentiated arthritis and inflammatory arthralgia. Front Med 8:668898. https://doi.org/10.3389/fmed.2021.668898
Chen J, Wright K, Davis JM et al (2016) An expansion of rare lineage intestinal microbes characterizes rheumatoid arthritis. Genome Med 8:43. https://doi.org/10.1186/S13073-016-0299-7
Jagpal A, Navarro-Millán I (2018) Cardiovascular co-morbidity in patients with rheumatoid arthritis: a narrative review of risk factors, cardiovascular risk assessment and treatment. BMC Rheumatol 2:10. https://doi.org/10.1186/S41927-018-0014-Y
Bustamante MF, Garcia-Carbonell R, Whisenant KD, Guma M (2017) Fibroblast-like synoviocyte metabolism in the pathogenesis of rheumatoid arthritis. Arthritis Res Ther 19:110. https://doi.org/10.1186/S13075-017-1303-3
Kim JM, Lin C, Stavre Z et al (2020) Osteoblast-osteoclast communication and bone homeostasis. Cells 9:2073. https://doi.org/10.3390/CELLS9092073.
Fang Q, Zhou C, Nandakumar KS (2020) Molecular and cellular pathways contributing to joint damage in rheumatoid arthritis. Mediators Inflamm 2020:3830212. https://doi.org/10.1155/2020/3830212
Steffen U, Schett G, Bozec A (2019) How autoantibodies regulate osteoclast induced bone loss in rheumatoid arthritis. Front Immunol 10:1483. https://doi.org/10.3389/FIMMU.2019.01483
Breedveld FC (2002) Current and future management approaches for rheumatoid arthritis. Arthritis Res Ther 4(2):1–6. https://doi.org/10.1186/AR548
Rantapää-Dahlqvist S, De Jong BAW, Berglin E et al (2003) Antibodies against cyclic citrullinated peptide and IgA rheumatoid factor predict the development of rheumatoid arthritis. Arthritis Rheum 48:2741–2749. https://doi.org/10.1002/ART.11223
Nielen MMJ, Van Schaardenburg D, Reesink HW et al (2004) Specific autoantibodies precede the symptoms of rheumatoid arthritis a study of serial measurements in blood donors. Arthritis Rheum 50:380–386. https://doi.org/10.1002/art.20018
Jørgensen KT, Wiik A, Pedersen M et al (2008) Cytokines, autoantibodies and viral antibodies in premorbid and postdiagnostic sera from patients with rheumatoid arthritis: case–control study nested in a cohort of norwegian blood donors. Ann Rheum Dis 67:860–866. https://doi.org/10.1136/ARD.2007.073825
Silman AJ, Hennessy E, Ollier B (1992) Incidence of rheumatoid arthritis in a genetically predisposed population. Rheumatology 31:365–368. https://doi.org/10.1093/RHEUMATOLOGY/31.6.365
Bos WH, Wolbink GJ, Boers M et al (2010) Arthritis development in patients with arthralgia is strongly associated with anti-citrullinated protein antibody status: a prospective cohort study. Ann Rheum Dis 69:490–494. https://doi.org/10.1136/ARD.2008.105759
Sokolove J, Bromberg R, Deane KD et al (2012) Autoantibody epitope spreading in the pre-clinical phase predicts progression to rheumatoid arthritis. PLoS ONE 7:e35296. https://doi.org/10.1371/JOURNAL.PONE.0035296
Suwannalai P, Van De Stadt LA, Radner H et al (2012) Avidity maturation of anti-citrullinated protein antibodies in rheumatoid arthritis. Arthritis Rheum 64:1323–1328. https://doi.org/10.1002/ART.33489
Gvozdenović E, Dirven L, Van Den Broek M et al (2014) Intra articular injection with corticosteroids in patients with recent onset rheumatoid arthritis: subanalyses from the BeST study. Clin Rheumatol 33:263–267. https://doi.org/10.1007/S10067-013-2465-2
Sandler RD, Dunkley L (2018) Osteoarthritis and the inflammatory arthritides. Surgery (United Kingdom) 36:21–26. https://doi.org/10.1016/j.mpsur.2017.10.004
Wilson A, Yu HT, Goodnough LT, Nissenson AR (2004) Prevalence and outcomes of anemia in rheumatoid arthritis: a systematic review of the literature. Am J Med 116:50–57. https://doi.org/10.1016/j.amjmed.2003.12.012
Ingegnoli F, Castelli R, Gualtierotti R (2013) Rheumatoid factors: clinical applications. Dis Markers 35:727–734. https://doi.org/10.1155/2013/726598
Lerkvaleekul B, Jaovisidha S, Sungkarat W et al (2017) The comparisons between thermography and ultrasonography with physical examination for wrist joint assessment in juvenile idiopathic arthritis. Physiol Meas 38:691–700. https://doi.org/10.1088/1361-6579/AA63D8
Laurent L, Anquetil F, Clavel C et al (2015) IgM rheumatoid factor amplifies the inflammatory response of macrophages induced by the rheumatoid arthritis-specific immune complexes containing anticitrullinated protein antibodies. Ann Rheum Dis 74:1425–1431. https://doi.org/10.1136/ANNRHEUMDIS-2013-204543
Muehleman C, Green J, Williams JM et al (2002) The effect of bone remodeling inhibition by zoledronic acid in an animal model of cartilage matrix damage. Osteoarthr Cartil 10:226–233. https://doi.org/10.1053/joca.2001.0506
Grigor C, Capell H, Stirling A et al (2004) Effect of a treatment strategy of tight control for rheumatoid arthritis (the TICORA study): a single-blind randomised controlled trial. Lancet 364:263–269. https://doi.org/10.1016/S0140-6736(04)16676-2
St.Clair EW, Van Der Heijde DMFM, Smolen JS et al (2004) Combination of infliximab and methotrexate therapy for early rheumatoid arthritis: a randomized, controlled trial. Arthritis Rheum 50:3432–3443. https://doi.org/10.1002/art.20568
Yoo DH, Prodanovic N, Jaworski J et al (2017) Efficacy and safety of CT-P13 (biosimilar infliximab) in patients with rheumatoid arthritis: comparison between switching from reference infliximab to CT-P13 and continuing CT-P13 in the PLANETRA extension study. Ann Rheum Dis 76:355–363. https://doi.org/10.1136/ANNRHEUMDIS-2015-208786
Roubille C, Richer V, Starnino T et al (2015) The effects of tumour necrosis factor inhibitors, methotrexate, non-steroidal anti-inflammatory drugs and corticosteroids on cardiovascular events in rheumatoid arthritis, psoriasis and psoriatic arthritis: a systematic review and meta-analysis. Ann Rheum Dis 74:480–489. https://doi.org/10.1136/ANNRHEUMDIS-2014-206624
Rathbun AM, Harrold LR, Reed GW (2016) A prospective evaluation of the effects of prevalent depressive symptoms on disease activity in rheumatoid arthritis patients treated with biologic response modifiers. Clin Ther 38:1759–1772. https://doi.org/10.1016/J.CLINTHERA.2016.06.007
Storgard CM, Stupack DG, Jonczyk A et al (1999) Decreased angiogenesis and arthritic disease in rabbits treated with an αvβ3 antagonist. J Clin Invest 103:47–54. https://doi.org/10.1172/JCI3756
O’Dell JR, Haire CE, Erikson N et al (1996) Treatment of rheumatoid arthritis with methotrexate alone, sulfasalazine and hydroxychloroquine, or a combination of all three medications. N Engl J Med 334:1287–1291. https://doi.org/10.1056/NEJM199605163342002
Shea B, Swinden MV, Tanjong Ghogomu E et al (2013) Folic acid and folinic acid for reducing side effects in patients receiving methotrexate for rheumatoid arthritis. Cochrane Database Syst Rev 2013:CD000951. https://doi.org/10.1002/14651858.CD000951.pub2
Smolen JS, Cohen SB, Tony HP et al (2017) A randomised, double-blind trial to demonstrate bioequivalence of GP2013 and reference rituximab combined with methotrexate in patients with active rheumatoid arthritis. Ann Rheum Dis 76:1598–1602. https://doi.org/10.1136/ANNRHEUMDIS-2017-211281
Jones G, Ding C (2010) Tocilizumab: a review of its safety and efficacy in rheumatoid arthritis. Clin Med Insights Arthritis Musculoskelet Disord 3:81–89. https://doi.org/10.4137/CMAMD.S4864
Bermas BL (2014) Non-steroidal anti inflammatory drugs, glucocorticoids and disease modifying anti-rheumatic drugs for the management of rheumatoid arthritis before and during pregnancy. Curr Opin Rheumatol 26:334–340. https://doi.org/10.1097/BOR.0000000000000054
Wen H-Y, Chiang C-C, Chen R-Y et al (2023) Immunosensing for early detection of rheumatoid arthritis biomarkers: anti-cyclic citrullinated peptide antibodies based on tilted-fiber bragg grating biosensor. Bioengineering 10:261. https://doi.org/10.3390/bioengineering10020261
Hasegawa T, Kaneko Y, Izumi K, Takeuchi T (2017) Efficacy of denosumab combined with bDMARDs on radiographic progression in rheumatoid arthritis. Joint Bone Spine 84:379–380. https://doi.org/10.1016/J.JBSPIN.2016.05.010
Felson DT, Anderson JJ, Meenan RF (1990) The comparative efficacy and toxicity of second-line drugs in rheumatoid arthritis results of two metaanalyses. Arthritis Rheum 33:1449–1461. https://doi.org/10.1002/ART.1780331001
Di Giuseppe D, Wallin A, Bottai M et al (2014) Long-term intake of dietary long-chain n-3 polyunsaturated fatty acids and risk of rheumatoid arthritis: a prospective cohort study of women. Ann Rheum Dis 73:1949–1953. https://doi.org/10.1136/ANNRHEUMDIS-2013-203338
Blanco FJ, Möricke R, Dokoupilova E et al (2017) Secukinumab in active rheumatoid arthritis: a phase III randomized, double-blind, active comparator: and placebo-controlled study. Arthritis Rheumatol 69:1144–1153. https://doi.org/10.1002/ART.40070
Fleischmann R, Cutolo M, Genovese MC et al (2012) Phase IIb dose-ranging study of the oral JAK inhibitor tofacitinib (CP-690,550) or adalimumab monotherapy versus placebo in patients with active rheumatoid arthritis with an inadequate response to disease-modifying antirheumatic drugs. Arthritis Rheum 64:617–629. https://doi.org/10.1002/art.33383
Pannell WC, Savin DD, Scott TP et al (2015) Trends in the surgical treatment of lumbar spine disease in the United States. Spine J 15:1719–1727. https://doi.org/10.1016/J.SPINEE.2013.10.014
Momohara S, Inoue E, Ikari K et al (2014) Recent trends in orthopedic surgery aiming to improve quality of life for those with rheumatoid arthritis: data from a large observational cohort. J Rheumatol 41:862–866. https://doi.org/10.3899/JRHEUM.131018
Kanbe K, Chiba J, Inoue Y et al (2015) Analysis of clinical factors related to the efficacy of shoulder arthroscopic synovectomy plus capsular release in patients with rheumatoid arthritis. Eur J Orthop Surg Traumatol 25:451–455. https://doi.org/10.1007/S00590-014-1570-5
Eversden L, Maggs F, Nightingale P, Jobanputra P (2007) A pragmatic randomised controlled trial of hydrotherapy and land exercises on overall well being and quality of life in rheumatoid arthritis. BMC Musculoskelet Disord 8:23. https://doi.org/10.1186/1471-2474-8-23
Gehrmann R, Harten R, Renard RL et al (2016) Biomechanical evaluation of patellar tendon repair techniques: comparison of double krackow stitch with and without cerclage augmentation. Orthop Rheumatol 5:00163. https://doi.org/10.15406/MOJOR.2016.05.00163
Köhler BM, Günther J, Kaudewitz D, Lorenz HM (2019) Current therapeutic options in the treatment of rheumatoid arthritis. J Clin Med 8:938. https://doi.org/10.3390/JCM8070938
Choudhary M, Kumar V, Malhotra H, Singh S (2015) Medicinal plants with potential anti-arthritic activity. J Intercult Ethnopharmacol 4:147. https://doi.org/10.5455/JICE.20150313021918
Feng Y, Zhang R, Zhao Z et al (2023) Efficacy and safety of electroacupuncture combined with medication for rheumatoid arthritis: a systematic review and meta-analysis. Heliyon 9:e14014. https://doi.org/10.1016/j.heliyon.2023.e14014
Farzaei MH, Farzaei F, Abdollahi M et al (2016) A mechanistic review on medicinal plants used for rheumatoid arthritis in traditional persian medicine. J Pharm Pharmacol 68:1233–1248. https://doi.org/10.1111/jphp.12606
Nimesh S (2018) Herbal drug is better than allopathic drug in the treatment of rheumatoid arthritis. Int J Pharmacogn 5(9):539–545. https://doi.org/10.13040/IJPSR.0975-8232.IJP.5
Chiu HY, Huang HL, Li CH et al (2015) Increased risk of chronic kidney disease in rheumatoid arthritis associated with cardiovascular complications: a national population-based cohort study. PLoS ONE 10:1–13. https://doi.org/10.1371/journal.pone.0136508
Bhupinder K, Reena G, Gupta M (2017) Natural products in treatment of rheumatoid arthritis. Int J Green Pharm 11:356–363
Teles KA, Medeiros-Souza P, Lima FAC et al (2017) Cyclophosphamide administration routine in autoimmune rheumatic diseases: a review. Rev Brasi de Reumatol (English Edition) 57:596–604. https://doi.org/10.1016/j.rbre.2016.09.008
Grainger R, Walker J (2014) Rheumatologists’ opinions towards complementary and alternative medicine: a systematic review. Clin Rheumatol 33:3–9. https://doi.org/10.1007/s10067-013-2379-z
Ekor M (2014) The growing use of herbal medicines: issues relating to adverse reactions and challenges in monitoring safety. Front Pharmacol 4:1–10. https://doi.org/10.3389/FPHAR.2013.00177
Venkatesha SH, Dudics S, Astry B, Moudgil KD (2016) Control of autoimmune inflammation by celastrol, a natural triterpenoid. Pathog Dis 74:1–12. https://doi.org/10.1093/FEMSPD/FTW059
Che CT, Wong MS, Lam CWK, McPhee DJ (2016) Natural products from chinese medicines with potential benefits to bone health. Molecules 21:239. https://doi.org/10.3390/MOLECULES21030239
Kaur A, Nain P, Nain J (2012) Herbal plants used in treatment of rheumatoid arthritis: a review. Int J Pharm Pharm Sci 4:44–57
Arya V, Kumar Gupta V, Kaur R, Pharm M (2011) A review on plants having anti-arthritic potential morphological and microscopic studies of aerial parts of ceylon leadwort view project Vikrant Arya. Int J Pharm Sci Rev Res 7:239
Goel A, Kulshrestha S (2021) Review on anti-rheumatoid arthritis potential of medicinal plants. Int J Curr Res Rev 13:16–32. https://doi.org/10.31782/IJCRR.2021.13303
Shashank D, Rajendra S, Mistry A (2018) An overview of phytoconstituents and pharmacological activities of Celastrus paniculatus Willd. J Pharm Res 16:307–313
Li M, He J, Jiang LL et al (2013) The anti-arthritic effects of Aconitum vilmorinianum, a folk herbal medicine in Southwestern China. J Ethnopharmacol 147:122–127. https://doi.org/10.1016/j.jep.2013.02.018
Gokhale AB, Damre AS, Kulkarni KR, Saraf MN (2002) Preliminary evaluation of anti-inflammatory and anti-arthritic activity of S. lappa, A. speciosa and A. aspera. Phytomedicine 9:433–437. https://doi.org/10.1078/09447110260571689
Pharm IJ, Res P, Bk M et al (2012) Hepatoprotective potency of Achyranthes aspera: an in-vivo study. Int J Pharm Phytopharmacol Res 1:387–390
Singh B, Bani S, Gupta DK et al (2003) Anti-inflammatory activity of ‘TAF’ an active fraction from the plant Barleria prionitis Linn. J Ethnopharmacol 85:187–193. https://doi.org/10.1016/S0378-8741(02)00358-6
Nazir N, Koul S, Qurishi MA et al (2007) Immunomodulatory effect of bergenin and norbergenin against adjuvant-induced arthritis—a flow cytometric study. J Ethnopharmacol 112:401–405. https://doi.org/10.1016/j.jep.2007.02.023
Fan AY, Lao L, Zhang RX et al (2005) Effects of an acetone extract of Boswellia carterii Birdw. (Burseraceae) gum resin on adjuvant-induced arthritis in Lewis rats. J Ethnopharmacol 101:104–109. https://doi.org/10.1016/j.jep.2005.03.033
Wu SQ, Otero M, Unger FM et al (2011) Anti-inflammatory activity of an ethanolic Caesalpinia sappan extract in human chondrocytes and macrophages. J Ethnopharmacol 138:364–372. https://doi.org/10.1016/j.jep.2011.09.011
Kumar VL, Roy S (2007) Calotropis procera latex extract affords protection against inflammation and oxidative stress in Freund’s complete adjuvant-induced monoarthritis in rats. Mediat Inflamm 2007:047523. https://doi.org/10.1155/2007/47523
Jeyadevi R, Sivasudha T, Rameshkumar A, Kumar LD (2013) Anti-arthritic activity of the indian leafy vegetable Cardiospermum halicacabum in Wistar rats and UPLC-QTOF- MS/MS identification of the putative active phenolic components. Inflamm Res 62:115–126. https://doi.org/10.1007/s00011-012-0558-z
Mukhopadhyay MK, Nath D (2011) Phytochemical screening and toxicity study of Saraca asoca bark methanolic extract. Int J Phytomed 3:498–505
Agarwal T, Singh R, Shukla AD et al (2012) Comparative analysis of antibacterial activity of four piper betel varieties. Pelagia Res Libr 3:698–705
Radu AF, Bungau SG (2021) Management of rheumatoid arthritis: an overview. Cells 10:2857. https://doi.org/10.3390/CELLS10112857
Zhou L, Xiong JY, Chai YQ et al (2022) Possible antidepressant mechanisms of omega-3 polyunsaturated fatty acids acting on the central nervous system. Front Psychiatry 13:1991. https://doi.org/10.3389/FPSYT.2022.933704/BIBTEX
Kowalska K (2011) Natural compounds involved in adipose tissue mass control in in vitro studies. Postepy higieny i medycyny doswiadczalnej (Online) 65:515–523. https://doi.org/10.5604/17322693.955499
Schroeder FC, Gronquist M (2006) Extending the scope of NMR spectroscopy with microcoil probes. Angew Chem Int Ed 45:7122–7131. https://doi.org/10.1002/ANIE.200601789
Ross IA (2005) Medicinal plants of the world. Med Plants World 3:1–623. https://doi.org/10.1007/978-1-59259-887-8/COVER
Chan K, Shaw D, Simmonds MSJ et al (2012) Good practice in reviewing and publishing studies on herbal medicine, with special emphasis on traditional chinese medicine and chinese materia medica. J Ethnopharmacol 140:469–475. https://doi.org/10.1016/J.JEP.2012.01.038
Makunga NP, Philander LE, Smith M (2008) Current perspectives on an emerging formal natural products sector in South Africa. J Ethnopharmacol 119:365–375. https://doi.org/10.1016/J.JEP.2008.07.020
Jianzhang M, Ke R, Kun C (2013) Research and practice on biodiversity in situ conservation in China: pro-gress and prospect. Biodivers Sci 20:551–558. https://doi.org/10.3724/SP.J.1003.2012.08118
Acknowledgements
Authors are grateful to the GLA University, management for providing support.
Funding
None.
Author information
Authors and Affiliations
Contributions
AS: Original draft preparation, AG: Supervision and Editing.
Corresponding author
Ethics declarations
Conflict of interest
All authors declared that there are no conflicts of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sharma, A., Goel, A. Pathogenesis of rheumatoid arthritis and its treatment with anti-inflammatory natural products. Mol Biol Rep 50, 4687–4706 (2023). https://doi.org/10.1007/s11033-023-08406-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-023-08406-4