Abstract
Background
Mortality in patients with diabetes mellitus is estimated above 65% due to cardiovascular diseases. The aim of study was to investigate the effects of high-glucose conditions on TGF-β type II receptor (TGFBR2) expression levels, cell viability, and migration rate in vascular smooth muscle cells (VSMCs).
Methods
VSMCs were incubated in 30 mM and 50 mM of glucose for 24 h, 48 h, and 72 h periods. The gene and protein expression levels were investigated by Real-time qRT-PCR and western blotting techniques, respectively. The cell viability was evaluated by MTT assay. VSMC migration rate was also studied by wound healing assay.
Results
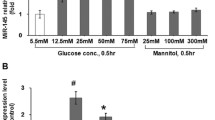
The TGFBR2 gene and protein expression levels were significantly upregulated in all the groups treated with glucose in 24 h, 48 h, and 72 h periods. The cell viability was not significantly affected in values of 30 mM and 50 mM of glucose. The increase of migration rate of VSMCs was not significant.
Conclusion
The results suggested the increased expression levels of TGFBR2 in the response to high glucose conditions may modulate the cellular events through the signaling pathway network in VSMCs.






Similar content being viewed by others
Data availability
The data sets used and/or analyzed during this study are available from the corresponding author on request.
Abbreviations
- CVD:
-
Cardiovascular disease
- VSMCs:
-
Vascular smooth muscle cells
- AGEs:
-
Advanced glycation end-products
- TGF-β:
-
Transforming growth factor-beta
- TGFBR2:
-
TGF-β type II receptor
- SMC:
-
Smooth muscle cells
- DMEM:
-
Dulbecco’s modified eagle medium
- FBS:
-
Fetal bovine serum
- cDNA:
-
Complementary DNA
- PVDF:
-
Polyvinylidene difluoride
- TBST:
-
Tris-buffered saline with 0.1% Tween®20
- ECL:
-
Enhanced chemiluminescence
- PMSF:
-
Phenylmethylsulfonyl fluoride
- PBS:
-
Phosphate-buffered saline
- CMR:
-
Cellular migration rate
- T2D:
-
Type 2 diabetes
- H-RPE:
-
Human retinal pigment epithelium
- MAPKs:
-
Mitogen-activated protein kinases
- ERK:
-
Extracellular-signal-regulated kinase
- JNK:
-
Jun N-terminal kinase
References
Low Wang CC, Hess CN, Hiatt WR, Goldfine AB (2016) Clinical update: cardiovascular disease in diabetes mellitus: atherosclerotic cardiovascular disease and heart failure in type 2 diabetes mellitus–mechanisms, management, and clinical considerations. Circulation 133(24):2459–2502
Beckman JA, Creager MA, Libby P (2002) Diabetes and atherosclerosis: epidemiology, pathophysiology, and management. JAMA 287(19):2570–2581
Gleissner CA, Galkina E, Nadler JL, Ley K (2007) Mechanisms by which diabetes increases cardiovascular disease. Drug Discov Today Dis Mech 4(3):131–140
Langlois D, Hneino M, Bouazza L, Parlakian A, Sasaki T, Bricca G et al (2010) Conditional inactivation of TGF-β type II receptor in smooth muscle cells and epicardium causes lethal aortic and cardiac defects. Transgenic Res 19(6):1069–1082
Michon IN, Penning LC, Molenaar TJ, van Berkel TJ, Biessen EA, Kuiper J (2002) The effect of TGF-β receptor binding peptides on smooth muscle cells. Biochem Biophys Res Commun 293(4):1279–1286
Kanzaki T, Tamura K, Takahashi K, Saito Y, Akikusa B, Oohashi H et al (1995) In vivo effect of TGF-β1: enhanced intimal thickening by administration of TGF-β1 in rabbit arteries injured with a balloon catheter. Arterioscler Thromb Vasc Biol 15:1951–195711
Majesky MW, Lindner V, Twardzik DR, Schwartz SM, Reidy MA (1991) Production of transforming growth factor beta 1 during repair of arterial injury. J Clin Investig 88(3):904–910
Massagué J, Gomis RR (2006) The logic of TGFβ signaling. FEBS Lett 580(12):2811–2820
Massagué J (2000) How cells read TGF-β signals. Nat Rev Mol Cell Biol 1(3):169–178
Tsai S, Hollenbeck ST, Ryer EJ, Edlin R, Yamanouchi D, Kundi R et al (2009) TGF-β through Smad3 signaling stimulates vascular smooth muscle cell proliferation and neointimal formation. Am J Physiol Heart Circ Physiol 297(2):H540–H5H9
Lien S-C, Wei S-Y, Chang S-F, Chang MD-T, Chang J-Y, Chiu J-J (2013) Activation of PPAR-α induces cell cycle arrest and inhibits transforming growth factor-β1 induction of smooth muscle cell phenotype in 10T1/2 mesenchymal cells. Cell Signal 25(5):1252–1263
Hino K, Ikeya M, Horigome K, Matsumoto Y, Ebise H, Nishio M et al (2015) Neofunction of ACVR1 in fibrodysplasia ossificans progressiva. Proc Natl Acad Sci 112(50):15438–15443
Zhou JW, Wang M, Sun NX, Qing Y, Yin TF, Li C et al (2019) Sulforaphane–induced epigenetic regulation of Nrf2 expression by DNA methyltransferase in human Caco–2 cells. Oncol Lett 18(3):2639–2647
Ismail N, Abdullah N, Abdul Murad NA, Jamal R, Sulaiman SA (2021) Long non-coding RNAs (lncRNAs) in cardiovascular disease complication of type 2 diabetes. Diagnostics 11(1):145
Pirri D, Fragiadaki M, Evans PC (2020) Diabetic atherosclerosis: is there a role for the hypoxia-inducible factors? Biosci Rep 40(8):BSR20200026
Selvin E, Coresh J, Shahar E, Zhang L, Steffes M, Sharrett AR (2005) Glycaemia (haemoglobin A1c) and incident ischaemic stroke: the Atherosclerosis Risk in Communities (ARIC) Study. Lancet Neurol 4(12):821–826
Khaw K-T, Wareham N, Bingham S, Luben R, Welch A, Day N (2004) Association of hemoglobin A1c with cardiovascular disease and mortality in adults: the European prospective investigation into cancer in Norfolk. Ann Intern Med 141(6):413–420
Gerstein H, Pogue J, Mann J, Lonn E, Dagenais G, McQueen M et al (2005) The relationship between dysglycaemia and cardiovascular and renal risk in diabetic and non-diabetic participants in the HOPE study: a prospective epidemiological analysis. Diabetologia 48(9):1749–1755
Azevedo PS, Polegato BF, Minicucci MF, Paiva SA, Zornoff LA (2016) Cardiac remodeling: concepts, clinical impact, pathophysiological mechanisms and pharmacologic treatment. Arq Bras Cardiol 106(1):62–69
Farnoodian M, Halbach C, Slinger C, Pattnaik BR, Sorenson CM, Sheibani N (2016) High glucose promotes the migration of retinal pigment epithelial cells through increased oxidative stress and PEDF expression. Am J Physiol-Cell Physiol 311(3):C418-CC36
Fei J, Xiao C, Yang M, Zhou X, Gong P (2021) Inhibition of SNCG suppresses the proliferation of lung cancer cells induced by high glucose. Mol Med Rep 23(2):1
Yuan H, Fan Y, Wang Y, Gao T, Shao Y, Zhao B et al (2019) Calcium–sensing receptor promotes high glucose–induced myocardial fibrosis via upregulation of the TGF–β1/Smads pathway in cardiac fibroblasts. Mol Med Rep 20(2):1093–1102
Wu F, Wang F, Yang Q, Zhang Y, Cai K, Liu L et al (2020) Upregulation of miRNA-23a-3p rescues high glucose-induced cell apoptosis and proliferation inhibition in cardiomyocytes. In Vitro Cell Dev Biol-Anim 56(10):866–877
Ye D, Lou GH, Li AC, Dong FQ, Chen GP, Xu WW et al (2020) MicroRNA–125a–mediated regulation of the mevalonate signaling pathway contributes to high glucose–induced proliferation and migration of vascular smooth muscle cells. Mol Med Rep 22(1):165–174
Zhou D-M, Ran F, Ni H-Z, Sun L-L, Xiao L, Li X-Q et al (2020) Metformin inhibits high glucose-induced smooth muscle cell proliferation and migration. Aging 12(6):5352
Zhang W, Chen S, Zhang Z, Wang C, Liu C (2017) FAM3B mediates high glucose-induced vascular smooth muscle cell proliferation and migration via inhibition of miR-322-5p. Sci Rep 7(1):2298
Qin D, Jiang Y-R (2020) Tangeretin inhibition of high-glucose-induced IL-1β, IL-6, TGF-β1, and VEGF expression in human RPE cells. J Diabetes Res. https://doi.org/10.1155/2020/9490642
Nickel J, Ten Dijke P, Mueller TD (2018) TGF-β family co-receptor function and signaling. Acta Biochim Biophys Sin 50(1):12–36
Massagué J, Chen Y-G (2000) Controlling TGF-β signaling. Genes Dev 14(6):627–644
Frutkin AD, Shi H, Otsuka G, Levéen P, Karlsson S, Dichek DA (2006) A critical developmental role for tgfbr2 in myogenic cell lineages is revealed in mice expressing SM22-Cre, not SMMHC-Cre. J Mol Cell Cardiol 41(4):724–731
Acknowledgements
No.
Funding
The study is supported by Iran University of Medical Sciences, Grant No. 14440.
Author information
Authors and Affiliations
Contributions
MN designed research; GG, FZG, AAS and AM performed experiments; MN and GG analyzed data; MN and GG wrote the manuscript. All authors reviewed and approved the manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest, financial or otherwise.
Ethical approval
The Ethical Committee of Iran University of Medical Sciences, Tehran, Iran, approves the study (14440).
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ghasempour, G., Mohammadi, A., Zamani-Garmsiri, F. et al. Upregulation of TGF-β type II receptor in high glucose-induced vascular smooth muscle cells. Mol Biol Rep 49, 2869–2875 (2022). https://doi.org/10.1007/s11033-021-07100-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-07100-7