Abstract
Background
Abuse of addictive drugs such as methamphetamine (METH) has become a global problem, leading to many social, economic, and health disturbances, including neurological and cognitive disorders. Neuronal damage is reported in chronic METH abusers. The neuroprotective role of CoQ10 has been shown in many studies. In the present study, we aimed to assess the pre and post-efficacy of CoQ10 on the dopaminergic neurons of the Nucleus Accumbens (de Miranda et al. in Food Res Int 121:641–647, 2019) in the male adult rats treated with METH.
Methods
80 rats were randomly divided into eight groups (n = 10), including: negative control (intact), positive control (received 5 mg/kg/day METH/IP), three post-treatment groups (METH + 5, 10, 20 mg/kg CoQ10) and three pre-treatment groups (received 5, 10, 20 mg/kg CoQ10 as pre-treatment for 14 days before METH injection). The expression of Bax, Bcl-2, Bax/Bcl-2 ratio, P53, Caspase-3 and tyrosine hydroxylase in NAc studied using western blotting. Nissl staining was used to study the neuronal density of NAc.
Results
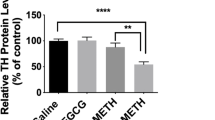
Our results showed that the different doses of CoQ10 in METH-treated animals significantly changed pro-apoptotic proteins’ expression in the benefit of neuronal survival of NAc (P < 0.05). Neuronal density in NAc were significantly lower in the METH group compared to the control and CoQ10 treated groups. Pre- and post-treatment with different doses of CoQ10 restored the neuronal damage in NAc.
Conclusions
CoQ10 could decrease the activation of pro-apoptotic proteins and reduce the neurodegenerative effects induced by METH. From a clinical point of view, it seems that certain antioxidants such as CoQ10 should receive more attention in clinical trial research. We believe that antioxidants could be the promising for drug abuse treatment in the future.




Similar content being viewed by others
References
Peacock A, Leung J, Larney S, Colledge S, Hickman M, Rehm J et al (2018) Global statistics on alcohol, tobacco and illicit drug use: 2017 status report. Addiction 113(10):1905–1926
Potvin S, Pelletier J, Grot S, Hebert C, Barr AM, Lecomte T (2018) Cognitive deficits in individuals with methamphetamine use disorder: a meta-analysis. Addict Behav 80:154–160
Turowski P, Kenny B-A (2015) The blood-brain barrier and methamphetamine: open sesame? Front NeuroSci 9:156
Thrash B, Karuppagounder SS, Uthayathas S, Suppiramaniam V, Dhanasekaran M (2010) Neurotoxic effects of methamphetamine. Neurochem Res 35(1):171–179
Rocha A, Kalivas PW (2010) Role of the prefrontal cortex and nucleus accumbens in reinstating methamphetamine seeking. Eur J Neurosci 31(5):903–909
Moszczynska A, Callan SP (2017) Molecular, behavioral, and physiological consequences of methamphetamine neurotoxicity: implications for treatment. J Pharmacol Exp Ther 362(3):474–488
Moszczynska A, Yamamoto BK (2011) Methamphetamine oxidatively damages parkin and decreases the activity of 26S proteasome in vivo. J Neurochem 116(6):1005–1017
Limanaqi F, Gambardella S, Biagioni F, Busceti CL, Fornai F (2018) Epigenetic effects induced by methamphetamine and methamphetamine-dependent oxidative stress. Oxidative Med Cell Longev. https://doi.org/10.1155/2018/4982453
Frank MG, Adhikary S, Sobesky JL, Weber MD, Watkins LR, Maier SF (2016) The danger-associated molecular pattern HMGB1 mediates the neuroinflammatory effects of methamphetamine. Brain Behav Immun 51:99–108
Halpin LE, Collins SA, Yamamoto BK (2014) Neurotoxicity of methamphetamine and 3, 4-methylenedioxymethamphetamine. Life Sci 97(1):37–44
Chen C, Qincao L, Xu J, Du S, Huang E, Liu C et al (2016) Role of PUMA in methamphetamine-induced neuronal apoptosis. Toxicol Lett 240(1):149–160
Elyasi L, Eftekhar-Vaghefi SH, Esmaeili-Mahani S (2014) Morphine protects SH-SY5Y human neuroblastoma cells against 6-hydroxydopamine–induced cell damage: involvement of anti-oxidant, calcium blocking, and anti-apoptotic properties. Rejuven Res 17(3):255–263
Nopparat C, Porter JE, Ebadi M, Govitrapong P (2010) The mechanism for the neuroprotective effect of melatonin against methamphetamine-induced autophagy. J Pineal Res 49(4):382–389
Bank G, Kagan D, Madhavi D (2011) Coenzyme Q10: clinical update and bioavailability. J Evid-Based Complement Altern Med 16(2):129–137
Yang X, Zhang Y, Xu H, Luo X, Yu J, Liu J et al (2016) Neuroprotection of coenzyme Q10 in neurodegenerative diseases. Curr Top Med Chem 16(8):858–866
Thanh HN, Minh HPT, Duc LV, Thanh TB (2016) Protective effect of coenzyme Q10 on methamphetamine-induced neurotoxicity in the mouse brain. Trends Med Res 11(1):1–10
Klongpanichapak S, Govitrapong P, Sharma SK, Ebadi M (2006) Attenuation of cocaine and methamphetamine neurotoxicity by coenzyme Q 10. Neurochem Res 31(3):303–311
Gholipour F, Shams J, Zahiroddin A (2017) Protective effect of coenzyme Q10 on methamphetamine-induced apoptosis in adult male rats. Nov Biomed 5(3):127–132
Jameie SB, Masoumipoor M, Janzadeh A, Nasirinezhad F, Kerdari M, Soleimani M (2014) Combined therapeutic effects of low power laser (980 nm) and CoQ10 on Neuropathic Pain in adult male rat. Med J Islam Repub Iran 28:58
Björklund A, Dunnett SB (2019) The amphetamine induced rotation test: a re-assessment of its use as a tool to monitor motor impairment and functional recovery in rodent models of Parkinson’s disease. J Parkinson’s Dis 9(1):17–29
Thanos PK, Kim R, Delis F, Ananth M, Chachati G, Rocco MJ et al (2016) Chronic methamphetamine effects on brain structure and function in rats. PLoS One 11(6):e0155457
Hori N, Kadota M, Watanabe M, Ito Y, Akaike N, Carpenter D (2010) Neurotoxic effects of methamphetamine on rat hippocampus pyramidal neurons. Cell Mol Neurobiol 30(6):849–856
Martin TA, Jayanthi S, McCoy MT, Brannock C, Ladenheim B, Garrett T et al (2012) Methamphetamine causes differential alterations in gene expression and patterns of histone acetylation/hypoacetylation in the rat nucleus accumbens. PLoS One 7(3):e34236
Yu S, Zhu L, Shen Q, Bai X, Di X (2015) Recent advances in methamphetamine neurotoxicity mechanisms and its molecular pathophysiology. Behav Neurol. https://doi.org/10.1155/2015/103969
Zhou F, Yang Y, Xing D (2011) Bcl-2 and Bcl‐xL play important roles in the crosstalk between autophagy and apoptosis. FEBS J 278(3):403–413
Huang W, Xie W-B, Qiao D, Qiu P, Huang E, Li B et al (2015) Caspase-11 plays an essential role in methamphetamine-induced dopaminergic neuron apoptosis. Toxicol Sci 145(1):68–79
Parrish AB, Freel CD, Kornbluth S (2013) Cellular mechanisms controlling caspase activation and function. Cold Spring Harb Perspect Biol 5(6):a008672
Reiner DJ, Yu S-J, Shen H, He Y, Bae E, Wang Y (2014) 9-Cis retinoic acid protects against methamphetamine-induced neurotoxicity in nigrostriatal dopamine neurons. Neurotox Res 25(3):248–261
Bentinger M, Tekle M, Dallner G (2010) Coenzyme Q–biosynthesis and functions. Biochem Biophys Res Commun 396(1):74–79
Badawy Khair NS, Mohammed SA (2021) A comparative study on the protective role of Silymarin and Coenzyme-Q10 on the cerebellar cortex of experimentally induced atherosclerosis in adult male albino rats: a histological, immunohistochemical and biochemical study. Egypt J Histol 44(2):322–338
Mancuso M, Orsucci D, Volpi L, Calsolaro V, Siciliano G (2010) Coenzyme Q10 in neuromuscular and neurodegenerative disorders. Curr Drug Targets 11(1):111–121
Spindler M, Beal MF, Henchcliffe C (2009) Coenzyme Q10 effects in neurodegenerative disease. Neuropsychiatr Dis Treat 5:597
Vaselbehagh M, Sadegh M, Karami H, Babaie S (2021) Coenzyme Q10 modulates apoptotic effects of chronic administration of methadone on NMRI mouse hippocampus. Cell J (Yakhteh) 23:538–5435
Acknowledgements
The authors are deeply grateful to colleagues and laboratory staff of NRC/IUMS for their sincere collaboration and Vice-Chancellor of Research and Technology (Grant No. 8811215292).
Funding
The present research is supported by the Neuroscience Research Center (NRC) of the Iran University of Medical Science.
Author information
Authors and Affiliations
Contributions
SBJ prepared the study design and budget planning as the PI of the project. MF, AK, ZS, SAB were involved in experiments and wrote the paper draft. SB and MJ collected the data, wrote first draft of the manuscript and edited. All the authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared that no conflict of interest exist.
Ethical approval
The ethic committee for animal research of Neuroscience Research Center affiliated to Iran University of Medical Sciences approved all the procedures used in this study (Ethical Code IR.IUMS.REC.1396.32724).
Consent to participate
All authors participated in the informed consent.
Consent for publication
All authors approved publication.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Jameie, S.B., Kazemian, A., Sanadgol, Z. et al. Coenzyme Q10 reduces expression of apoptotic markers in adult rat nucleus accumbens dopaminergic neurons treated with methamphetamine. Mol Biol Rep 49, 2273–2281 (2022). https://doi.org/10.1007/s11033-021-07049-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-07049-7