Abstract
Background
Seeking novel anticancer agents with minimal side effects against gastric cancer is vitally important. Copper, as an important trace element, takes roles in different physiologic pathways. Also, there is a higher demand for copper in cancer cells than normal ones. Copper complexes containing a therapeutic ligand could be promising candidates for gastric cancer chemotherapy.
Methods and results
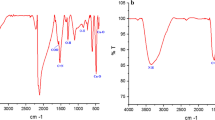
In this work, copper oxide nanoparticles were synthesized, functionalized with glutamic acid (CuO@Glu) and conjugated with thiosemicarbazone (CuO@Glu/TSC NPs). The NPs were characterized and their antiproliferative potential against AGS cancer cells was investigated using MTT, flow cytometry, Hoechst staining, and caspase 3 activation assays. The FT-IR results showed the proper binding of TSC to CuO@Glu NPs and crystallinity of the prepared NPs was confirmed by the XRD pattern. The EDX analysis confirmed the presence of Cu, N, C, O, and S elements and lack of impurities. The Hydrodynamic size and zeta potential of the CuO@Glu/TSC NPs were 168 nm and 27.5 mV, respectively. The NPs had spherical shape and were in a size range of 10 to 60 nm in diameter. This work revealed that CuO@Glu/TSC NPs efficiently inhibited the proliferation of AGS cells with significantly lower IC50 value (203 µg/mL) than normal HEK293 cells (IC50 = 435 µg/mL). Flow cytometry and Hoechst staining obviously revealed apoptosis induction among CuO@Glu/TSC treated cells, and caspase-3 activity significantly increased by 1.4 folds.
Conclusions
This study introduced CuO@Glu/TSC as an efficient anticancer against gastric cancer cells with lower toxicity toward normal cells which could be employed for cancer treatment after further studies.









Similar content being viewed by others
References
Rawla P, Barsouk A (2019) Epidemiology of gastric cancer: global trends, risk factors and prevention. Prz Gastroenterol 14(1):26. https://doi.org/10.5114/pg.2018.80001
Nagajyothi PC, Muthuraman P, Sreekanth TVM, Kim DH, Shim J (2017) Green synthesis: in-vitro anticancer activity of copper oxide nanoparticles against human cervical carcinoma cells. Arab J Chem 10(2):215–225. https://doi.org/10.1016/j.arabjc.2016.01.011
Tisato F, Marzano C, Porchia M, Pellei M, Santini C (2010) Copper in diseases and treatments, and copper-based anticancer strategies. Med Res Rev 30(4):708–749. https://doi.org/10.1002/med.20174
Qi J, Yao Q, Tian L, Wang Y (2018) Piperidylthiosemicarbazones Cu (II) complexes with a high anticancer activity by catalyzing hydrogen peroxide to degrade DNA and promote apoptosis. Eur J Med Chem 158:853–862. https://doi.org/10.1016/j.ejmech.2018.09.034
Lovejoy DB, Jansson PJ, Brunk UT, Wong J, Ponka P, Richardson DR (2011) Antitumor activity of metal-chelating compound Dp44mT is mediated by formation of a redox-active copper complex that accumulates in lysosomes. Can Res 71(17):5871–5880. https://doi.org/10.1158/0008-5472.CAN-11-1218
Tardito S, Marchio L (2009) Copper compounds in anticancer strategies. Curr Med Chem 16(11):1325–1348. https://doi.org/10.2174/092986709787846532
Ma ZY, Shao J, Bao WG, Qiang ZY, Xu JY (2015) A thiosemicarbazone copper (II) complex as a potential anticancer agent. J Coord Chem 68(2):277–294. https://doi.org/10.1080/00958972.2014.979811
Scovill JP, Klayman DL, Franchino CF (1982) 2-Acetylpyridine thiosemicarbazones. 4. Complexes with transition metals as antimalarial and antileukemic agents. J Med Chem 25(10):1261–1264. https://doi.org/10.1021/jm00352a036
Rahnama A, Gharagozlou M (2012) Preparation and properties of semiconductor CuO nanoparticles via a simple precipitation method at different reaction temperatures. Opt Quant Electron 44(6–7):313–322. https://doi.org/10.1007/s11082-011-9540-1
Shandiz SAS, Montazeri A, Abdolhosseini M, Shahrestani SH, Hedayati M, Moradi-Shoeili Z, Salehzadeh A (2018) Functionalization of Ag nanoparticles by glutamic acid and conjugation of Ag@ Glu by thiosemicarbazide enhances the apoptosis of human breast cancer MCF-7 cells. J Clust Sci 29(6):1107–1114. https://doi.org/10.1007/s10876-018-1424-0
Jarestan M, Khalatbari K, Pouraei A, Sadat Shandiz SA, Beigi S, Hedayati M, Salehzadeh A (2020) Preparation, characterization, and anticancer efficacy of novel cobalt oxide nanoparticles conjugated with thiosemicarbazide. 3 Biotech 10:1–9. https://doi.org/10.1007/s13205-020-02230-4
Bejarbaneh M, Moradi-Shoeili Z, Jalali A, Salehzadeh A (2020) Synthesis of cobalt hydroxide nano-flakes functionalized with glutamic acid and conjugated with thiosemicarbazide for anticancer activities against human breast cancer cells. Biol Trace Elem Res. https://doi.org/10.1007/s12011-020-02049-3
Khan S, Ansari AA, Khan AA, Abdulla M, Al-Obaid O, Ahmad R (2017) In vitro evaluation of cytotoxicity, possible alteration of apoptotic regulatory proteins, and antibacterial activity of synthesized copper oxide nanoparticles. Colloids Surf B 153:320–326. https://doi.org/10.1016/j.colsurfb.2017.03.005
Rehana D, Mahendiran D, Kumar RS, Rahiman AK (2017) Evaluation of antioxidant and anticancer activity of copper oxide nanoparticles synthesized using medicinally important plant extracts. Biomed Pharmacother 89:1067–1077. https://doi.org/10.1016/j.biopha.2017.02.101
Habibi A, Shandiz SA, Salehzadeh A, Moradi-Shoeili Z (2020) Novel pyridinecarboxaldehyde thiosemicarbazone conjugated magnetite nanoparticulates (MNPs) promote apoptosis in human lung cancer A549 cells. J Biol Inorg Chem 25(1):13–22. https://doi.org/10.1007/s00775-019-01728-4
Marzano C, Pellei M, Tisato F, Santini C (2009) Copper complexes as anticancer agents. Anti-Cancer Agents Med Chem 9(2):185–211. https://doi.org/10.2174/187152009787313837
Halliwell B, Aruoma OI (1991) DNA damage by oxygen-derived species its mechanism and measurement in mammalian systems. FEBS Lett 281(1–2):9–19. https://doi.org/10.1016/0014-5793(91)80347-6
Schnitzer E, Pinchuk I, Lichtenberg D (2007) Peroxidation of liposomal lipids. Eur Biophys J 36(4–5):499–515. https://doi.org/10.1007/s00249-007-0146-2
Lodish H, Berk A, Kaiser CA, Krieger M, Scott MP, Bretscher A et al (2008). Molecular cell biology. Macmillan
Marques AJ, Palanimurugan R, Matias AC, Ramos PC, Dohmen RJ (2009) Catalytic mechanism and assembly of the proteasome. Chem Rev 109(4):1509–1536. https://doi.org/10.1021/cr8004857
Weder JE, Dillon CT, Hambley TW, Kennedy BJ, Lay PA, Biffin JR et al (2002) Copper complexes of non-steroidal anti-inflammatory drugs: an opportunity yet to be realized. Coord Chem Rev 232(1–2):95–126. https://doi.org/10.1016/S0010-8545(02)00086-3
Easmon J, Pürstinger G, Heinisch G, Roth T, Fiebig HH, Holzer W, Hofmann J (2001) Synthesis, cytotoxicity, and antitumor activity of copper (II) and iron (II) complexes of 4 N-azabicyclo [3.2.2] nonane thiosemicarbazones derived from acyl diazines. J Med Chem 44(13):2164–2171. https://doi.org/10.1021/jm000979z
Siddiqui MA, Alhadlaq HA, Ahmad J, Al-Khedhairy AA, Musarrat J, Ahamed M (2013) Copper oxide nanoparticles induced mitochondria mediated apoptosis in human hepatocarcinoma cells. PLoS ONE 8(8):e69534. https://doi.org/10.1371/journal.pone.0069534
Ahamed M, Siddiqui MA, Akhtar MJ, Ahmad I, Pant AB, Alhadlaq HA (2010) Genotoxic potential of copper oxide nanoparticles in human lung epithelial cells. Biochem Biophys Res Commun 396(2):578–583. https://doi.org/10.1016/j.bbrc.2010.04.156
Fuentes-Prior P, Salvesen GS (2004) The protein structures that shape caspase activity, specificity, activation and inhibition. Biochem J 384(2):201–232. https://doi.org/10.1042/BJ20041142
Acknowledgements
We would like to appreciate Dr. Zeinab Moradi-Shoeili for collaborate on the interpretation of chemical spectra.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
Conceptualization: MB and AS; Methodology: AS; Formal analysis and investigation: MB, FSH, SHJ, SRM, MJ, HF and MH; Writing—original draft preparation: AS; Editing: AS; Resources: MB, FSH, SHJ, SRM, MJ, TGH, HF and MH; Supervision: AS.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Badrooh, M., Shokrollahi, F., Javan, S. et al. Trigger of apoptosis in adenocarcinoma gastric cell line (AGS) by a complex of thiosemicarbazone and copper nanoparticles. Mol Biol Rep 49, 2217–2226 (2022). https://doi.org/10.1007/s11033-021-07043-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-07043-z