Abstract
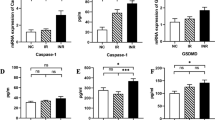
HIV infection is a global health concern. Current HIV-diagnostics provide information about the disease progression and efficacy of anti-retroviral therapies (ARVs), but this information is very limited and sometimes imprecise. Present study assessed the potential role of mononuclear cell (MNC) death, expression of caspases (1&3) and cell free mitochondrial DNA (CF mt-DNA) in HIV infected individuals. Apoptosis, cell-count, expression of caspases and CF mt-DNA were measured through flow cytometry and qPCR, respectively, in HIV infected individuals (n = 120) divided in two groups i.e. ARVs-receiving (treated, n = 87), ART-naïve (untreated, n = 37) and healthy individuals (n = 47). Data showed significant (p < 0.0001) cell death in untreated individuals than treated and healthy individuals. CD4-positive T-cell percentage declined (p < 0.0001) in untreated as compared to treated individuals. Caspase-1, an indicator of pyroptosis, and CF mt-DNA were also elevated in untreated HIV infected individuals. Untreated individuals when administered with ARVs showed improved CD4-positive T-cell percentage, lower caspase-1, CF mt-DNA and cell death. Data elucidated positive co-relation between cell death and CF mt-DNA in treated and untreated HIV infected individuals. While CD4-positive T-cell percentage was negatively correlated with caspase-1 expression and CF mt-DNA. Elevated levels of CF mt-DNA and caspase-1 in HIV infected individuals, positive correlation between cell death and CF mt-DNA, negative correlation of CD4-positive T-cell percentage with CF mt-DNA and caspase-1 expression clearly indicated the potential of CF mt-DNA and caspase-1 as a novel disease progression and ARTs effectiveness biomarkers in HIV.




Similar content being viewed by others
Data availability
All the relevant data points are included in the manuscript. For acquisition of raw data, please contact Mariam Anees (mariam@qau.edu.pk).
References
Burack JH, Barrett DC, Stall RD, Chesney MA, Ekstrand ML, Coates TJ (1993) Depressive symptoms and CD4 lymphocyte decline among HIV-infected men. JAMA 270:2568–2573. https://doi.org/10.1001/jama.1993.03510210054027
Cells CD, Laurent-crawford AG, Krust B, Rivière Y, Müller S, Kieny MP, Dauguet C, Ara G (1993) Membrane expression of HIV envelope glycoproteins triggers apoptosis in CD4 cells. AIDS Res Hum Retrovir 9:761–773
Finkel TH, Tudor-Williams G, Banda NK, Cotton MF, Curiel T, Monks C, Baba TW, Ruprecht RM, Kupfer A (1995) Apoptosis occurs predominantly in bystander cells and not in productively infected cells of HIV- and SIV-infected lymph nodes. Nat Med 1:129–134. https://doi.org/10.1038/nm0295-129
Campbell GR, Spector SA (2019) DIABLO/SMAC mimetics selectively kill HIV-1-infected resting memory CD4+ T cells: a potential role in a cure strategy for HIV-1 infection. Autophagy 15:744–746. https://doi.org/10.1080/15548627.2019.1569950
Serrano A, El HS, Moal F, Prazuck T, Legac E, Robin C, Brule F, Charpentier S, Normand T, Legrand A, Hocqueloux L, Mollet L (2018) Dysregulation of apoptosis and autophagy gene expression in peripheral blood mononuclear cells of efficiently treated HIV-infected patients. AIDS 32:1579–1587. https://doi.org/10.1097/QAD.0000000000001851
Ta V, Laubach S, Diego S, Ta V, Laubach S, Diego S (2015) Cell death by pyroptosis drives CD4 T-cell depletion in HIV-1 infection. Pediatrics 135:S13–S15. https://doi.org/10.1542/peds.2014
Yu F, Ma C, Jin X, Zhao H, Xiao J, Li L, Sing S, Zhang X, Xie X, Yang S, Tang Y, Wang L, Zhang F (2019) Increased mitochondrial mass leads to heightened pyroptosis that drives CD4+ T cell depletion in patients with HIV-1 infection. Available at SSRN: 3475569. https://doi.org/10.2139/ssrn.3475569
Frank MO (2016) Circulating cell-free DNA differentiates severity of inflammation. Biol Res Nurs 18:477–488. https://doi.org/10.1177/1099800416642571
Mehta SR, Pérez-Santiago J, Hulgan T, Day TRC, Barnholtz-Sloan J, Gittleman H, Letendre S, Ellis R, Heaton R, Patton S, Suben JD, Franklin D, Rosario D, Clifford DB, Collier AC, Marra CM, Gelman BB, McArthur J, McCutchan A, Morgello S, Simpson D, Connor J, Grant I, Kallianpur A (2017) Cerebrospinal fluid cell-free mitochondrial DNA is associated with HIV replication, iron transport, and mild HIV-associated neurocognitive impairment. J Neuroinflamm 14:1–9. https://doi.org/10.1186/s12974-017-0848-z
Ellinger J, Müller SC, Wernert N, Von Ruecker A, Bastian PJ (2008) Mitochondrial DNA in serum of patients with prostate cancer: a predictor of biochemical recurrence after prostatectomy. BJU Int 102:628–632. https://doi.org/10.1111/j.1464-410X.2008.07613.x
Nakahira K, Kyung SY, Rogers AJ, Gazourian L, Youn S, Massaro AF, Quintana C, Osorio JC, Wang Z, Zhao Y, Lawler LA, Christie JD, Meyer NJ, Causland FRM, Waikar SS, Waxman AB, Chung RT, Bueno R, Rosas IO, Fredenburgh LE, Baron RM, Christiani DC, Hunninghake GM, Choi AMK (2013) Circulating mitochondrial DNA in patients in the ICU as a marker of mortality: derivation and validation. PLoS Med 10:1–12. https://doi.org/10.1371/journal.pmed.1001577
Dwivedi DJ, Toltl LJ, Swystun LL, Pogue J, Liaw K-L, Weitz JI, Cook DJ, Fox-Robichaud AE, Liaw PC, Group the CCCTB (2012) Prognostic utility and characterization of cell-free DNA in patients with severe sepsis. Crit Care 16:R151. https://doi.org/10.1186/cc11466
Kung C-T, Hsiao S-Y, Tsai T-C, Su C-M, Chang W-N, Huang C-R, Wang H-C, Lin W-C, Chang H-W, Lin Y-J, Cheng B-C, Su BY-J, Tsai N-W, Lu C-H (2012) Plasma nuclear and mitochondrial DNA levels as predictors of outcome in severe sepsis patients in the emergency room. J Transl Med 10:130. https://doi.org/10.1186/1479-5876-10-130
Swarup V, Rajeswari MR (2007) Circulating (cell-free) nucleic acids—a promising, non-invasive tool for early detection of several human diseases. FEBS Lett 581:795–799
Boyapati RK, Rossi AG, Satsangi J, Ho GT (2016) Gut mucosal DAMPs in IBD: from mechanisms to therapeutic implications. Mucosal Immunol 9:567–582. https://doi.org/10.1038/mi.2016.14
Zhang Q, Raoof M, Chen Y, Sumi Y, Sursal T, Junger W, Brohi K, Itagaki K, Hauser CJ (2010) Circulating mitochondrial DAMPs cause inflammatory responses to injury. Nature 464:104–107. https://doi.org/10.1038/nature08780
Crepaz N, Tang T, Marks G, Medicine HH-A of I (2017) Undefined viral suppression patterns among persons in the United States with diagnosed HIV infection in 2014. Ann Intern Med 167(6):446. https://doi.org/10.7326/L17-0278
Rio DC, Ares M, Hannon GJ, Nilsen TW (2010) Purification of RNA using TRIzol (TRI reagent). Cold Spring Harb Protoc 5:1–4. https://doi.org/10.1101/pdb.prot5439
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods 25:402–408. https://doi.org/10.1006/meth.2001.1262
Sambrook J, Russell DW (2006) Purification of nucleic acids by extraction with phenol: chloroform. Cold Spring Harb Protoc 2006:4455. https://doi.org/10.1101/pdb.prot4455
Malik AN, Shahni R, Rodriguez-de-Ledesma A, Laftah A, Cunningham P (2011) Mitochondrial DNA as a non-invasive biomarker: accurate quantification using real time quantitative PCR without co-amplification of pseudogenes and dilution bias. Biochem Biophys Res Commun 412:1–7. https://doi.org/10.1016/j.bbrc.2011.06.067
Feria MG, Taborda NA, Hernandez JC, Rugeles MT (2018) HIV replication is associated to inflammasomes activation, IL-1β, IL-18 and caspase-1 expression in GALT and peripheral blood. PLoS ONE 13:1–14. https://doi.org/10.1371/journal.pone.0192845
Hunt PW (2012) HIV and inflammation: mechanisms and consequences. Curr HIV/AIDS Rep 9:139–147. https://doi.org/10.1007/s11904-012-0118-8
Stylianou E, Bjerkeli V, Yndestad A, Heggelund L, Wæhre T, Damås JK, Aukrust P, Frøland SS (2003) Raised serum levels of interleukin-18 is associated with disease progression and may contribute to virological treatment failure in HIV-1-infected patients. Clin Exp Immunol 132:462–466. https://doi.org/10.1046/j.1365-2249.2003.02179.x
Février M, Dorgham K, Rebollo A (2011) CD4 +T cell depletion in human immunodeficiency virus (HIV) infection: role of apoptosis. Viruses 3:586–612
Komanduri KV, Viswanathan MN, Wieder ED, Schmidt DK, Bredt BM, Jacobson MA, McCune MJ (1998) Restoration of cytomegalovirus-specific CD4+ T-lymphocyte responses after ganciclovir and highly active antiretroviral therapy in individuals infected with HIV-1. Nat Med 4(953):956
Autran B, Carcelain G, Li TS, Blanc C, Mathez D, Tubiana R, Katlama C, Debré P, Leibowitch J (1997) Positive effects of combined antiretroviral therapy on CD4+ T cell homeostasis and function in advanced HIV disease. Science 277:112–116. https://doi.org/10.1126/science.277.5322.112
Wensing AM, Calvez V, Ceccherini-Silberstein F, Charpentier C, Günthard HF, Paredes R, Shafer RW, Richman DD (2019) 2019 update of the drug resistance mutations in HIV-1. Top Antivir Med 27:111–121
Uppal SS, Verma S, Dhot PS (2003) Normal values of CD4 and CD8 lymphocyte subsets in healthy indian adults and the effects of sex, age, ethnicity, and smoking. Cytometry 52B:32–36. https://doi.org/10.1002/cyto.b.10011
Jiang W, Kang L, Lu H-Z, Pan X, Lin Q, Pan Q, Xue Y, Weng X, Tang Y-W (2004) Normal values for CD4 and CD8 lymphocyte subsets in healthy Chinese adults from Shanghai. Clin Diagn Lab Immunol 11:811–813. https://doi.org/10.1128/CDLI.11.4.811-813.2004
Wallace MR, Moss RB, Beecham HJIII, Grace CJ, Hersh EM, Peterson E, Murphy R, Shepp DH, Siegal FP, Turner JL, Safrin S, Carlo DJ, Levine AM (1996) Early clinical markers and CD4 percentage in subjects with human immunodeficiency virus infection. JAIDS J Acquir Immune Defic Syndr 12:358–362
Le TP, Tribble DR, Zhou SY, Malone L, Chung RC, Rusnak JM, Wagner KF (1997) Clinical significance of discordant CD4 count and CD4 percentage in HIV-infected individuals. AIDS 11:1395–1396
Cai R, Liu L, Luo B, Wang J, Shen J, Shen Y, Zhang R, Chen J, Lu H (2017) Caspase-1 activity in CD4 T cells is downregulated following antiretroviral therapy for HIV-1 infection. AIDS Res Hum Retrovir 33:164–171. https://doi.org/10.1089/aid.2016.0234
Kohler C, Radpour R, Barekati Z, Asadollahi R, Bitzer J, Wight E, Bürki N, Diesch C, Holzgreve W, Zhong XY (2009) Levels of plasma circulating cell free nuclear and mitochondrial DNA as potential biomarkers for breast tumors. Mol Cancer 8:105. https://doi.org/10.1186/1476-4598-8-105
Arnalich F, Maldifassi MC, Ciria E, Codoceo R, Renart J, Fernández-Capitán C, Herruzo R, Garcia-Rio F, López-Collazo E, Montiel C (2013) Plasma levels of mitochondrial and nuclear DNA in patients with massive pulmonary embolism in the emergency department: a prospective cohort study. Crit Care. https://doi.org/10.1186/cc12735
Ali Z, Waseem S, Anis RA, Anees M (2020) Assessment of cell free mitochondrial DNA as a biomarker of disease severity in different viral infections. Pak J Med Sci 36:860–866. https://doi.org/10.12669/pjms.36.5.2476
Jacotot E, Ravagnan L, Loeffler M, Ferri KF, Vieira HLA, Zamzami N, Costantini P, Druillennec S, Hoebeke J, Briand JP, Irinopoulou T, Daugas E, Susin SA, Cointe D, Xie ZH, Reed JC, Roques BP, Kroemer G (2000) The HIV-1 viral protein R induces apoptosis via a direct effect on the mitochondrial permeability transition pore. J Exp Med 191:33–45. https://doi.org/10.1084/jem.191.1.33
Arshad O, Gadawska I, Sattha B, Côté HCF, Hsieh AYY (2018) Elevated cell-free mitochondrial DNA in filtered plasma is associated with HIV infection and inflammation. J Acquir Immune Defic Syndr 78(1):111–118
Acknowledgements
We are thankful to Mr. Asghar Satti, Mr. Uzair Tariq, Miss Kalsoom Zahra and Mr. Ramzan from Association of People Living with HIV (APLHIV) Pakistan for their kind help in sampling and provision of necessary data.
Funding
The project has been supported financially by Higher Education Commission Grant No. NRPU 4481 to M. A. Research associate funding was provided to ZA in the same project.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ali, Z., Waseem, S., Shahzadi, I. et al. Association of cell free mitochondrial DNA and caspase-1 expression with disease severity and ARTs efficacy in HIV infection. Mol Biol Rep 48, 3327–3336 (2021). https://doi.org/10.1007/s11033-021-06313-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-021-06313-0