Abstract
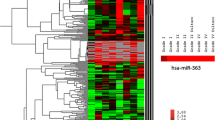
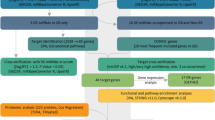
Glioblastoma (GBM) is the most common primary malignant neoplasm of the central nervous system and, despite the standard therapy; the patients’ prognoses remain dismal. The miRNA expression profiles have been associated with patient prognosis, suggesting that they may be helpful for tumor diagnosis and classification as well as predictive of tumor response to treatment. We described the microRNA expression profile of 29 primary GBM samples (9 pediatric GBMs) and 11 non-neoplastic white matter samples as controls (WM) by microarray analysis and we performed functional in vitro assays on these 2 most differentially expressed miRNAs. Hierarchical clustering analysis showed 3 distinct miRNA profiles, two of them in the GBM samples and a group consisting only of cerebral white matter. When adult and pediatric GBMs were compared to WM, 37 human miRNAs were found to be differentially expressed, with miR-10b-5p being the most overexpressed and miR-630 the most underexpressed. The overexpression of miR-630 was associated with reduced cell proliferation and invasion in the U87 GBM cell line, whereas the inhibition of miR-10b-5p reduced cell proliferation and colony formation in the U251 GBM cell line, suggesting that these miRNAs may act as tumor-suppressive and oncogenic miRNAs, respectively. The present study highlights the distinct epigenetic profiling of adult and pediatric GBMs and underscores the biological importance of mir-10b-5p and miR-630 for the pathobiology of these lethal tumors.





Similar content being viewed by others
Data availability
If the data used in this study are required, they can be available from the corresponding author.
References
Stupp R, Mason WP, van den Bent MJ et al (2005) Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 352(10):987–996
Stupp R, Taillibert S, Kanner A et al (2017) Effect of tumor-treating fields plus maintenance temozolomide vs maintenance temozolomide alone on survival in patients with glioblastoma: a randomized clinical trial. JAMA 318(23):2306–2316
Orringer D, Lau D, Khatri S et al (2012) Extent of resection in patients with glioblastoma: limiting factors, perception of resectability, and effect on survival. J Neurosurg 117(5):851–859
Louis DN, Perry A, Reifenberger G et al (2016) The 2016 World Health Organization classification of tumors of the central nervous system: a summary. Acta Neuropathol 131(6):803–820
Ostrom QT, Gittleman H, Liao P et al (2014) CBTRUS statistical report: primary brain and central nervous system tumors diagnosed in the United States in 2007–2011. Neuro Oncol 16(Suppl 4):iv1–iv63
Seystahl K, Wick W, Weller M (2016) Therapeutic options in recurrent glioblastoma—an update. Crit Rev Oncol Hematol 99:389–408
Anthiya S, Griveau A, Loussouarn C et al (2018) MicroRNA-based drugs for brain tumors. Trends Cancer 4(3):222–238
Areeb Z, Stylli SS, Koldej R et al (2015) MicroRNA as potential biomarkers in glioblastoma. J Neurooncol 125(2):237–248
Filippini G, Falcone C, Boiardi A et al (2008) Prognostic factors for survival in 676 consecutive patients with newly diagnosed primary glioblastoma. Neuro Oncol 10(1):79–87
Krex D, Klink B, Hartmann C et al (2007) Long-term survival with glioblastoma multiforme. Brain 130(Pt 10):2596–2606
Paugh BS, Qu C, Jones C et al (2010) Integrated molecular genetic profiling of pediatric high-grade gliomas reveals key differences with the adult disease. J Clin Oncol 28(18):3061–3068
Miele E, Buttarelli FR, Arcella A et al (2014) High-throughput microRNA profiling of pediatric high-grade gliomas. Neuro Oncol 16(2):228–240
Catania A, Maira F, Skarmoutsou E, D'Amico F, Abounader R, Mazzarino MC (2012) Insight into the role of microRNAs in brain tumors (review). Int J Oncol 40(3):605–624
Lu J, Getz G, Miska EA et al (2005) MicroRNA expression profiles classify human cancers. Nature 435(7043):834–838
Zhang B, Pan X, Cobb GP, Anderson TA (2007) microRNAs as oncogenes and tumor suppressors. Dev Biol 302(1):1–12
Kim TM, Huang W, Park R, Park PJ, Johnson MD (2011) A developmental taxonomy of glioblastoma defined and maintained by MicroRNAs. Cancer Res 71(9):3387–3399
Parker NR, Correia N, Crossley B, Buckland ME, Howell VM, Wheeler HR (2013) Correlation of microRNA 132 up-regulation with an unfavorable clinical outcome in patients with primary glioblastoma multiforme treated with radiotherapy plus concomitant and adjuvant temozolomide chemotherapy. Transl Oncol 6(6):742–748
Henriksen M, Johnsen KB, Olesen P, Pilgaard L, Duroux M (2014) MicroRNA expression signatures and their correlation with clinicopathological features in glioblastoma multiforme. Neuromolecular Med 16(3):565–577
Malzkorn B, Wolter M, Liesenberg F et al (2010) Identification and functional characterization of microRNAs involved in the malignant progression of gliomas. Brain Pathol 20(3):539–550
Low SY, Ho YK, Too HP, Yap CT, Ng WH (2014) MicroRNA as potential modulators in chemoresistant high-grade gliomas. J Clin Neurosci 21(3):395–400
Huang SW, Ali ND, Zhong L, Shi J (2018) MicroRNAs as biomarkers for human glioblastoma: progress and potential. Acta Pharmacol Sin 39(9):1405–1413
Meyer SU, Kaiser S, Wagner C, Thirion C, Pfaffl MW (2012) Profound effect of profiling platform and normalization strategy on detection of differentially expressed microRNAs—a comparative study. PLoS ONE 7(6):e38946
Vlachos IS, Zagganas K, Paraskevopoulou MD et al (2015) DIANA-miRPath v3.0: deciphering microRNA function with experimental support. Nucleic Acids Res 43(W1):W460–466
Livak KJ, Schmittgen TD (2001) Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) method. Methods 25(4):402–408
Wang Y, Jiang T (2013) Understanding high grade glioma: molecular mechanism, therapy and comprehensive management. Cancer Lett 331(2):139–146
Lee RC, Feinbaum RL, Ambros V (1993) The C. elegans heterochronic gene lin-4 encodes small RNAs with antisense complementarity to lin-14. Cell 75(5):843–854
Hummel R, Maurer J, Haier J (2011) MicroRNAs in brain tumors: a new diagnostic and therapeutic perspective? Mol Neurobiol 44(3):223–234
Ciafrè SA, Galardi S, Mangiola A et al (2005) Extensive modulation of a set of microRNAs in primary glioblastoma. Biochem Biophys Res Commun 334(4):1351–1358
Rao SA, Santosh V, Somasundaram K (2010) Genome-wide expression profiling identifies deregulated miRNAs in malignant astrocytoma. Mod Pathol 23(10):1404–1417
Chen W, Yu Q, Chen B, Lu X, Li Q (2016) The prognostic value of a seven-microRNA classifier as a novel biomarker for the prediction and detection of recurrence in glioma patients. Oncotarget 7(33):53392–53413
Jha P, Agrawal R, Pathak P et al (2015) Genome-wide small noncoding RNA profiling of pediatric high-grade gliomas reveals deregulation of several miRNAs, identifies downregulation of snoRNA cluster HBII-52 and delineates H3F3A and TP53 mutant-specific miRNAs and snoRNAs. Int J Cancer 137(10):2343–2353
Liang ML, Hsieh TH, Ng KH et al (2016) Downregulation of miR-137 and miR-6500-3p promotes cell proliferation in pediatric high-grade gliomas. Oncotarget 7(15):19723–19737
Zhu XP, Mou KJ, Xu QF et al (2015) Microarray analysis of the aberrant microRNA expression pattern in gliomas of different grades. Oncol Rep 34(1):318–324
Zhao JJ, Chen PJ, Duan RQ, Li KJ, Wang YZ, Li Y (2016) miR-630 functions as a tumor oncogene in renal cell carcinoma. Arch Med Sci 12(3):473–478
Chen WX, Zhang ZG, Ding ZY et al (2016) MicroRNA-630 suppresses tumor metastasis through the TGF-β- miR-630-Slug signaling pathway and correlates inversely with poor prognosis in hepatocellular carcinoma. Oncotarget 7(16):22674–22686
Chen MJ, Wu DW, Wang GC, Wang YC, Chen CY, Lee H (2018) MicroRNA-630 may confer favorable cisplatin-based chemotherapy and clinical outcomes in non-small cell lung cancer by targeting Bcl-2. Oncotarget 9(17):13758–13767
Chu D, Zhao Z, Li Y et al (2014) Increased microRNA-630 expression in gastric cancer is associated with poor overall survival. PLoS ONE 9(3):e90526
Chu D, Zheng J, Li J et al (2014) MicroRNA-630 is a prognostic marker for patients with colorectal cancer. Tumour Biol 35(10):9787–9792
Jin L, Yi J, Gao Y et al (2016) MiR-630 inhibits invasion and metastasis in esophageal squamous cell carcinoma. Acta Biochim Biophys Sin (Shanghai) 48(9):810–819
Zhou CX, Wang CL, Yu AL et al (2016) MiR-630 suppresses breast cancer progression by targeting metadherin. Oncotarget 7(2):1288–1299
Song YF, Hong JF, Liu DL, Lin QA, Lan XP, Lai GX (2015) miR-630 targets LMO3 to regulate cell growth and metastasis in lung cancer. Am J Transl Res 7(7):1271–1279
Zhang L, Wang C, Xue ZX (2017) Inhibition of miR-630 enhances the cell resistance to radiation by directly targeting CDC14A in human glioma. Am J Transl Res 9(3):1255–1265
Li D, Tian B, Jin X (2018) miR-630 inhibits epithelial-to-mesenchymal transition (EMT) by regulating the Wnt/β-catenin pathway in gastric cancer cells. Oncol Res 27(1):9–17
Corcoran C, Rani S, Breslin S et al (2014) miR-630 targets IGF1R to regulate response to HER-targeting drugs and overall cancer cell progression in HER2 over-expressing breast cancer. Mol Cancer 13:71
Farhana L, Dawson MI, Murshed F, Das JK, Rishi AK, Fontana JA (2013) Upregulation of miR-150* and miR-630 induces apoptosis in pancreatic cancer cells by targeting IGF-1R. PLoS ONE 8(5):e61015
Teplyuk NM, Uhlmann EJ, Wong AH et al (2015) MicroRNA-10b inhibition reduces E2F1-mediated transcription and miR-15/16 activity in glioblastoma. Oncotarget 6(6):3770–3783
Gabriely G, Yi M, Narayan RS et al (2011) Human glioma growth is controlled by microRNA-10b. Cancer Res 71(10):3563–3572
Sasayama T, Nishihara M, Kondoh T, Hosoda K, Kohmura E (2009) MicroRNA-10b is overexpressed in malignant glioma and associated with tumor invasive factors, uPAR and RhoC. Int J Cancer 125(6):1407–1413
Darrigo Júnior LG, Lira RCP, Fedatto PF et al (2019) MicroRNA profile of pediatric pilocytic astrocytomas identifies two tumor-specific signatures when compared to non-neoplastic white matter. J Neurooncol 141(2):373–382
Sun L, Yan W, Wang Y et al (2011) MicroRNA-10b induces glioma cell invasion by modulating MMP-14 and uPAR expression via HOXD10. Brain Res 1389:9–18
El Fatimy R, Subramanian S, Uhlmann EJ, Krichevsky AM (2017) Genome editing reveals glioblastoma addiction to microRNA-10b. Mol Ther 25(2):368–378
Funding
This study was supported by the Brazilian Public Research Agencies: Fundação de Amparo à Pesquisa do Estado de São Paulo (FAPESP, Grant Number: 2010/07020-9) and Conselho Nacional de Desenvolvimento Científico e Tecnológico (CNPq, Grant Number: 471885/2013-4).
Author information
Authors and Affiliations
Contributions
LGDJ, MB, LGT and CAS designed the project; LGDJ, MB, RCPL, ST, PFF, VSS, VKS, LCV, RAP and DSMA conducted the experiments and microarray analysis; LGDJ, MB, MB, JAY, SRB, SSA, LN, RSO, HRM, CGC, LGT, ETV, CAS were responsible for acquisition, analysis, or interpretation of data; LGDJ, MB, ETV and CAS wrote the manuscript. All authors revised and approved the final version.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that there are no conflicts of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Junior, L.G.D., Baroni, M., Lira, R.C.P. et al. High-throughput microRNA profile in adult and pediatric primary glioblastomas: the role of miR-10b-5p and miR-630 in the tumor aggressiveness. Mol Biol Rep 47, 6949–6959 (2020). https://doi.org/10.1007/s11033-020-05754-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-020-05754-3