Abstract
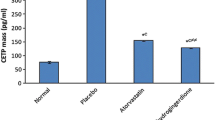
10-Dehydrogingerdione (10-DHGD) was previously reported to possess a hypolipidemic, anti-inflammatory and anti-oxidant properties in hyperlipidemic rabbit model. In this study, we investigated a possible new role for 10-DHGD in modulating atherogenic lipid profile by targeting proprotein convertase subtilisin kexin-9 (PCSK-9). Cholesterol (0.2% w/w)-fed rabbits received either atorvastatin (20 mg/kg) or 10-DHGD (10 mg/kg) for 12 weeks along with cholesterol feeding (HCD). Lipid profile, serum PCSK-9 and macrophage migration inhibitory factor (MIF), and aorta level of tumor necrosis factor-alpha (TNF-α) and glycosaminoglycans (GAGs) were measured. HCD-fed rabbits revealed an atherogenic lipid profile along with increased serum level of PCSK-9 (p < 0.001) and increased serum MIF and aortic TNF-α and GAGs (p < 0.001). 10-DHGD administration to HCD-fed rabbits prevented this atheogenicity by modulating the release of PCSK-9, inflammation extent (serum MIF and aortic TNF-α) and GAGs. These results provide new insights on the hypolipidemic potential of 10-DHGD. The effects of 10-DHGD was superior to that of atorvastatin in most studied parameters modulating atherogenicity. 10-DHGD is found to be able to suppress the release of PCSK-9, decrease aortic expression of GAGs in cholesterol-fed rabbits and halt the inflammation extent. These effects may provide new insights on the hypolipidemic potential of 10-DHGD.



Similar content being viewed by others
Refrences
WHO Raised cholesterol: Situation and trends. https://www.who.int/gho/ncd/risk_factors/cholesterol_text/en/?fbclid=IwAR3X8tDaOrbwGCO4VsKNWQh6oTfFITnL5UQlnEjTvocjOadQNlAPEJP2Dlo
Jeong SM, Choi S, Kim K, Kim SM, Lee G, Park SY, Kim YY, Son JS, Yun JM, Park SM (2018) Effect of change in total cholesterol levels on cardiovascular disease among young adults. J Am Heart Assoc 7(12):e008819
Cannon CP, Steinberg BA, Murphy SA, Mega JL, Braunwald E (2006) Meta-analysis of cardiovascular outcomes trials comparing intensive versus moderate statin therapy. J Am Coll Cardiol 48(3):438–445. https://doi.org/10.1016/j.jacc.2006.04.070
Cholesterol Treatment Trialists C, Baigent C, Blackwell L, Emberson J, Holland LE, Reith C, Bhala N, Peto R, Barnes EH, Keech A, Simes J, Collins R (2010) Efficacy and safety of more intensive lowering of LDL cholesterol: a meta-analysis of data from 170,000 participants in 26 randomised trials. Lancet 376(9753):1670–1681. https://doi.org/10.1016/s0140-6736(10)61350-5
Stancu C, Sima A (2001) Statins: mechanism of action and effects. J Cell Mol Med 5(4):378–387
Davignon J (2004) Beneficial cardiovascular pleiotropic effects of statins. Circulation 109(23 Suppl 1):39–43. https://doi.org/10.1161/01.cir.0000131517.20177.5a
Guo S, Wang R, Yang Z, Li K, Wang Q (2012) Effects of atorvastatin on serum lipids, serum inflammation and plaque morphology in patients with stable atherosclerotic plaques. Exp Ther Med 4(6):1069–1074. https://doi.org/10.3892/etm.2012.722
Karlson BW, Wiklund O, Palmer MK, Nicholls SJ, Lundman P, Barter PJ (2016) Variability of low-density lipoprotein cholesterol response with different doses of atorvastatin, rosuvastatin, and simvastatin: results from VOYAGER. Eur Heart J Cardiovasc Pharmacother 2(4):212–217. https://doi.org/10.1093/ehjcvp/pvw006
Zhang H, Plutzky J, Skentzos S, Morrison F, Mar P, Shubina M, Turchin A (2013) Discontinuation of statins in routine care settings: a cohort study. Ann Intern Med 158(7):526–534. https://doi.org/10.7326/0003-4819-158-7-201304020-00004
Konrad RJ, Troutt JS, Cao G (2011) Effects of currently prescribed LDL-C-lowering drugs on PCSK9 and implications for the next generation of LDL-C-lowering agents. Lipids Health Dis 10:38. https://doi.org/10.1186/1476-511x-10-38
Stroes E, Colquhoun D, Sullivan D, Civeira F, Rosenson RS, Watts GF, Bruckert E, Cho L, Dent R, Knusel B, Xue A, Scott R, Wasserman SM, Rocco M, Investigators G- (2014) Anti-PCSK9 antibody effectively lowers cholesterol in patients with statin intolerance: the GAUSS-2 randomized, placebo-controlled phase 3 clinical trial of evolocumab. J Am Coll Cardiol 63(23):2541–2548. https://doi.org/10.1016/j.jacc.2014.03.019
Tavori H, Giunzioni I, Predazzi IM, Plubell D, Shivinsky A, Miles J, Devay RM, Liang H, Rashid S, Linton MF, Fazio S (2016) Human PCSK9 promotes hepatic lipogenesis and atherosclerosis development via apoE- and LDLR-mediated mechanisms. Cardiovasc Res 110(2):268–278. https://doi.org/10.1093/cvr/cvw053
Ooi TC, Abujrad H (2016) PCSK9 as a biomarker of cardiovascular disease. In: Patel VB, Preedy VR (eds) Biomarkers in cardiovascular disease. Springer Refrences, the Netherlands
Sabatine MS, Giugliano RP, Keech AC, Honarpour N, Wiviott SD, Murphy SA, Kuder JF, Wang H, Liu T, Wasserman SM, Sever PS, Pedersen TR, Investigator FSC (2017) Evolocumab and clinical outcomes in patients with cardiovascular disease. New Engl J Med 376(18):1713–1722. https://doi.org/10.1056/nejmoa1615664
Robinson JG, Farnier M, Krempf M, Bergeron J, Luc G, Averna M, Stroes ES, Langslet G, Raal FJ, El Shahawy M, Koren MJ, Lepor NE, Lorenzato C, Pordy R, Chaudhari U, Kastelein JJP, Investigators OLT (2015) Efficacy and safety of alirocumab in reducing lipids and cardiovascular events. New Engl J Med 372(16):1489–1499. https://doi.org/10.1056/nejmoa1501031
Elseweidy MM, Abdallah FR, Younis NN, Aldohmy S, Kassem HM (2013) 10-Dehydrogingerdione raises HDL-cholesterol through a CETP inhibition and wards off oxidation and inflammation in dyslipidemic rabbits. Atherosclerosis 231(2):334–340. https://doi.org/10.1016/j.atherosclerosis.2013.09.024
El-Seweidy MM, Asker Mel S, Eldahmy SI, Atteia HH, Abdallah MA (2015) Haemostatic risk factors in dyslipidemic rabbits: role of 10-dehydrogingerdione as a new hypolipemic agent. J Thromb Thrombolysis 39(2):196–202. https://doi.org/10.1007/s11239-014-1150-x
Madhumathi BG, Venkataranganna MV, Gopumadhavan S, Rafiq M, Mitra SK (2006) Induction and evaluation of atherosclerosis in New Zealand white rabbits. Indian J Exp Biol 44(3):203–208
Sabatine MS, Giugliano RP, Wiviott SD, Raal FJ, Blom DJ, Robinson J, Ballantyne CM, Somaratne R, Legg J, Wasserman SM, Scott R, Koren MJ, Stein EA, Open-Label Study of Long-Term Evaluation against LDLCI (2015) Efficacy and safety of evolocumab in reducing lipids and cardiovascular events. N Engl J Med 372(16):1500–1509. https://doi.org/10.1056/nejmoa1500858
Feingold KR, Moser AH, Shigenaga JK, Patzek SM, Grunfeld C (2008) Inflammation stimulates the expression of PCSK9. Biochem Biophy Res Commun 374(2):341–344. https://doi.org/10.1016/j.bbrc.2008.07.023
Lakoski SG, Lagace TA, Cohen JC, Horton JD, Hobbs HH (2009) Genetic and metabolic determinants of plasma PCSK9 levels. J Clin Endocrinol Metab 94(7):2537–2543. https://doi.org/10.1210/jc.2009-0141
Denis M, Marcinkiewicz J, Zaid A, Gauthier D, Poirier S, Lazure C, Seidah NG, Prat A (2012) Gene inactivation of proprotein convertase subtilisin/kexin type 9 reduces atherosclerosis in mice. Circulation 125(7):894–901. https://doi.org/10.1161/circulationaha.111.057406
Poirier S, Mayer G, Benjannet S, Bergeron E, Marcinkiewicz J, Nassoury N, Mayer H, Nimpf J, Prat A, Seidah NG (2008) The proprotein convertase PCSK9 induces the degradation of low density lipoprotein receptor (LDLR) and its closest family members VLDLR and ApoER2. J Biol Chem 283(4):2363–2372
Ouguerram K, Chetiveaux M, Zair Y, Costet P, Abifadel M, Varret M, Boileau C, Magot T, Krempf M (2004) Apolipoprotein B100 metabolism in autosomal-dominant hypercholesterolemia related to mutations in PCSK9. Arterioscler Thromb Vasc Biol 24(8):1448–1453. https://doi.org/10.1161/01.atv.0000133684.77013.88
Blom DJ, Hala T, Bolognese M, Lillestol MJ, Toth PD, Burgess L, Ceska R, Roth E, Koren MJ, Ballantyne CM, Monsalvo ML, Tsirtsonis K, Kim JB, Scott R, Wasserman SM, Stein EA (2014) A 52-week placebo-controlled trial of evolocumab in hyperlipidemia. N Engl J Med 370(19):1809–1819. https://doi.org/10.1056/nejmoa1316222
Welder G, Zineh I, Pacanowski MA, Troutt JS, Cao G, Konrad RJ (2010) High-dose atorvastatin causes a rapid sustained increase in human serum PCSK9 and disrupts its correlation with LDL cholesterol. J Lipid Res 51(9):2714–2721. https://doi.org/10.1194/jlr.m008144
Careskey HE, Davis RA, Alborn WE, Troutt JS, Cao G, Konrad RJ (2008) Atorvastatin increases human serum levels of proprotein convertase subtilisin/kexin type 9. J Lipid Res 49(2):394–398. https://doi.org/10.1194/jlr.m700437-jlr200
Walley KR, Thain KR, Russell JA, Reilly MP, Meyer NJ, Ferguson JF, Christie JD, Nakada TA, Fjell CD, Thair SA, Cirstea MS, Boyd JH (2014) PCSK9 is a critical regulator of the innate immune response and septic shock outcome. Sci Transl Med 6(258):258ra143. https://doi.org/10.1126/scitranslmed.3008782
Landlinger C, Pouwer MG, Juno C, van der Hoorn JWA, Pieterman EJ, Jukema JW, Staffler G, Princen HMG, Galabova G (2017) The AT04A vaccine against proprotein convertase subtilisin/kexin type 9 reduces total cholesterol, vascular inflammation, and atherosclerosis in APOE*3Leiden.CETP mice. Eur Heart J 38(32):2499–2507. https://doi.org/10.1093/eurheartj/ehx260
Liberale L, Montecucco F, Camici GG, Dallegri F, Vecchie A, Carbone F, Bonaventura A (2017) Treatment with proprotein convertase subtilisin/kexin type 9 (PCSK9) inhibitors to reduce cardiovascular inflammation and outcomes. Curr Med Chem 24(14):1403–1416. https://doi.org/10.2174/0929867324666170303123734
Kalliolias GD, Ivashkiv LB (2016) TNF biology, pathogenic mechanisms and emerging therapeutic strategies. Nat Rev Rheumatol 12(1):49–62. https://doi.org/10.1038/nrrheum.2015.169
Bernhagen J, Krohn R, Lue H, Gregory JL, Zernecke A, Koenen RR, Dewor M, Georgiev I, Schober A, Leng L, Kooistra T, Fingerle-Rowson G, Ghezzi P, Kleemann R, McColl SR, Bucala R, Hickey MJ, Weber C (2007) MIF is a noncognate ligand of CXC chemokine receptors in inflammatory and atherogenic cell recruitment. Nat Med 13(5):587–596. https://doi.org/10.1038/nm1567
Ohta H, Wada H, Niwa T, Kirii H, Iwamoto N, Fujii H, Saito K, Sekikawa K, Seishima M (2005) Disruption of tumor necrosis factor-alpha gene diminishes the development of atherosclerosis in ApoE-deficient mice. Atherosclerosis 180(1):11–17. https://doi.org/10.1016/j.atherosclerosis.2004.11.016
Jacobsson LT, Turesson C, Gulfe A, Kapetanovic MC, Petersson IF, Saxne T, Geborek P (2005) Treatment with tumor necrosis factor blockers is associated with a lower incidence of first cardiovascular events in patients with rheumatoid arthritis. J Rheumatol 32(7):1213–1218
Lin SG, Yu XY, Chen YX, Huang XR, Metz C, Bucala R, Lau CP, Lan HY (2000) De novo expression of macrophage migration inhibitory factor in atherogenesis in rabbits. Circ Res 87(12):1202–1208
Burger-Kentischer A, Goebel H, Seiler R, Fraedrich G, Schaefer HE, Dimmeler S, Kleemann R, Bernhagen J, Ihling C (2002) Expression of macrophage migration inhibitory factor in different stages of human atherosclerosis. Circulation 105(13):1561–1566
Tang ZH, Peng J, Ren Z, Yang J, Li TT, Li TH, Wang Z, Wei DH, Liu LS, Zheng XL, Jiang ZS (2017) New role of PCSK9 in atherosclerotic inflammation promotion involving the TLR4/NF-kappaB pathway. Atherosclerosis 262:113–122. https://doi.org/10.1016/j.atherosclerosis.2017.04.023
Fatkhullina AR, Peshkova IO, Koltsova EK (2016) The role of cytokines in the development of atherosclerosis. Biochem Biokhimiia 81(11):1358–1370. https://doi.org/10.1134/s0006297916110134
Kong YZ, Huang XR, Ouyang X, Tan JJ, Fingerle-Rowson G, Bacher M, Mu W, Scher LA, Leng L, Bucala R, Lan HY (2005) Evidence for vascular macrophage migration inhibitory factor in destabilization of human atherosclerotic plaques. Cardiovasc Res 65(1):272–282. https://doi.org/10.1016/j.cardiores.2004.09.020
Huang Q, Qin L, Dai S, Zhang H, Pasula S, Zhou H, Chen H, Min W (2013) AIP1 suppresses atherosclerosis by limiting hyperlipidemia-induced inflammation and vascular endothelial dysfunction. Arterioscler Thromb Vasc Biol 33(4):795–804. https://doi.org/10.1161/atvbaha.113.301220
Nakashima Y, Fujii H, Sumiyoshi S, Wight TN, Sueishi K (2007) Early human atherosclerosis: accumulation of lipid and proteoglycans in intimal thickenings followed by macrophage infiltration. Arterioscler Thromb Vasc Biol 27(5):1159–1165. https://doi.org/10.1161/atvbaha.106.134080
Acknowledgements
Authors would like to acknowledge the support given by Faculty of Pharmacy, Zagazig University for using Biotechnology Research and Pathological Markers laboratory and animal unit facilities. This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
MME is the principle investigator suggested the research point. STA and NNY carried out the experiments, performed statistical analysis and wrote the manuscript. MME and SEE revised the results and the manuscript. All authors revised and approved the manuscript for submission.
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Elseweidy, M.M., Elswefy, S.E., Younis, N.N. et al. Contribution of aorta glycosaminoglycans and PCSK9 to hyperlipidemia in experimental rabbits: the role of 10-dehdrogingerdione as effective modulator. Mol Biol Rep 46, 3921–3928 (2019). https://doi.org/10.1007/s11033-019-04836-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-019-04836-1