Abstract
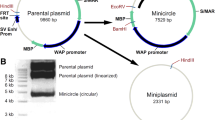
The presence of a bacterial backbone in conventional eukaryotic expression plasmids may cause undesirable effects by triggering the immune responses in mammals and repression of episomal transgene expression. To avoid these problems, researchers have proposed the use of minicircle DNAs which are episomal vectors that have lost their bacterial backbone using a site-specific recombinase mediated recombination. In the present study, we have constructed a new minicircle DNA vector that carries an enhanced green florescent protein (EGFP) reporter gene using phage ΦC31 integrase-mediated recombination and homing endonuclease ISceI-mediated purification in E. coli. ΦC31 integrase expression was under the control of the araBAD promoter, whereas ISceI endonuclease was controlled by the tac promoter. This vector was transfected into CHO-K1 cells, which showed transient expression of EGFP up to 14 generations. Similar results were obtained upon transient transfection into HEK cells. In addition, PCR results on genomic DNA, demonstrated the EGFP-minicircle was episomal and did not integrate into the host genome. Our constructed parental plasmid expresses EGFP and could be used for the generation of episomal minicircle DNA with intent to carry out transient transfection of interested DNA fragments into the eukaryotic cells for various purposes.











Similar content being viewed by others
References
Gill D, Pringle I, Hyde S (2009) Progress and prospects: the design and production of plasmid vectors. Gene Ther 16:165–171
Klinman DM (2004) Immunotherapeutic uses of CpG oligodeoxynucleotides. Nat Rev Immunol 4:249–259
Chen Z, He C, Meuse L, Kay M (2004) Silencing of episomal transgene expression by plasmid bacterial DNA elements in vivo. Gene Ther 11:856–864
Chen ZY, He CY, Ehrhardt A, Kay MA (2003) Minicircle DNA vectors devoid of bacterial DNA result in persistent and high-level transgene expression in vivo. Mol Ther 8:495–500
Chen ZY, Riu E, He CY, Xu H, Kay MA (2008) Silencing of episomal transgene expression in liver by plasmid bacterial backbone DNA is independent of CpG methylation. Mol Ther 16:548–556
Mayrhofer P, Schleef M, Jechlinger W (2009) Use of minicircle plasmids for gene therapy. Methods Mol Biol 542:87–104
Nehlsen K, Broll S, Bode J (2006) Replicating minicircles: generation of nonviral episomes for the efficient modification of dividing cells. Gene Ther Mol Biol 10:233–244
Darquet A, Cameron B, Wils P (1997) A new DNA vehicle for nonviral gene delivery: supercoiled minicircle. Gene Ther 4:1341–1349
Bigger BW, Tolmachov O, Collombet JM, Fragkos M, Palaszewski I, Coutelle C (2001) An araC-controlled bacterial Cre expression system to produce DNA minicircle vectors for nuclear and mitochondrial gene therapy. J Biol Chem 276:23018–23027
Jechlinger W, Azimpour Tabrizi C, Lubitz W, Mayrhofer P (2004) Minicircle DNA immobilized in bacterial ghosts: in vivo production of safe non-viral DNA delivery vehicles. J Mol Microbiol Biotechnol 8:222–231
Kay MA, He CY, Chen ZY (2010) A robust system for production of minicircle DNA vectors. Nat Biotechnol 28:1287–1289
Chen ZY, He CY, Kay MA (2005) Improved production and purification of minicircle DNA vector free of plasmid bacterial sequences and capable of persistent transgene expression in vivo. Hum Gene Ther 16:126–131
Jia F, Wilson KD, Sun N, Gupta DM, Huang M, Li Z, Panetta NJ, Chen ZY, Robbins RC, Kay MA, Longaker MT, Wu JC (2010) A nonviral minicircle vector for deriving human iPS cells. Nat Methods 7:197–199
Siegele DA, Hu JC (1997) Gene expression from plasmids containing the araBAD promoter at subsaturating inducer concentrations represents mixed populations. Proc Natl Acad Sci 94:8168–8172
Baneyx F (1999) Recombinant protein expression in Escherichia coli. Curr Opin Biotechnol 10:411–421
Xu X, Nagarajan H, Lewis NE et al (2011) The genomic sequence of the Chinese hamster ovary (CHO)-K1 cell line. Nat Biotechnol 29:735–741
Kobelt D, Schleef M, Schmeer M, Aumann J, Schlag PM, Walther W (2013) Performance of high quality minicircle DNA for in vitro and in vivo gene transfer. Mol Biotechnol 53:80–89
Dong Y, Aied A, Li J, Wang Q, Hu X, Wang W (2013) An in vitro approach for production of non-scar minicircle DNA vectors. J Biotechnol 166:84–87
Trichas G, Begbie J, Srinivas S (2008) Use of the viral 2A peptide for bicistronic expression in transgenic mice. BMC Biol 6:40
Acknowledgments
This study was funded by a Grant-in-aid of research from Royan Institute for Biotechnology in support of Nafiseh Sanei Ata-Abadi for obtaining her M.Sc. degree from the University of Isfahan.
Conflict of interest
None of the authors has any conflicts of interest to disclose and all authors support submission to this journal.
Author information
Authors and Affiliations
Corresponding authors
Electronic supplementary material
Below is the link to the electronic supplementary material.
11033_2015_3864_MOESM1_ESM.doc
Supplementary Fig. 3 Dot blot of flow cytometry to measure the EGFP intensity.Flow cytometry was carried out to compare the rate of green fluorescence in transfected mycoplasma free HEK293 cells with semi-purified minicircle product (a) and in vitro completely purified minicircle (b).Supplementary material 1 (DOC 29 kb)
11033_2015_3864_MOESM2_ESM.tif
Supplementary Fig. 1 Dot blot of flow cytometry to measure the EGFP intensity.Flow cytometry was carried out to determine the rate of transfected CHO cells expressing EGFP. Please see materials and methods section for further elucidationSupplementary material 2 (TIFF 52 kb)
11033_2015_3864_MOESM3_ESM.tif
Supplementary Fig. 2 Dot blot of flow cytometry to measure the EGFP intensity.Flow cytometry was carried out to determine the rate of green fluorescence in transfected HEK cells different days post transfection as described in Materials and MethodsSupplementary material 3 (TIFF 588 kb)
Rights and permissions
About this article
Cite this article
Sanei Ata-Abadi, N., Dormiani, K., Khazaie, Y. et al. Construction of a new minicircle DNA carrying an enhanced green florescent protein reporter gene for efficient expression into mammalian cell lines. Mol Biol Rep 42, 1175–1185 (2015). https://doi.org/10.1007/s11033-015-3864-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11033-015-3864-z